H2O Lecture Notes Note: The image below has been flipped and
advertisement

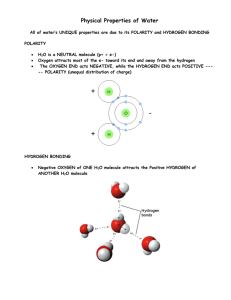
H2O Lecture Notes Note: The image below has been flipped and everything reads backwards, in order to have the Hydrogen “ears” be at the top of the molecule. For your test or notes, please write things in the conventional way, such as “1 P” for one proton, 8N for the 8 neutrons of Oxygen, and “bond” for the Hydrogen bond between each ear and the Oxygen atom. Be sure to show the areas of negative and positive charges as shown below. Area of positive charges since each Hydrogen electron “hangs out” a lot with the Oxygen atom. + + + + + + + + + + + + + + + + + + + _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ Area of negative charge thanks to the electrons of Oxygen Because the area near the H “ears” have a positive charge, and the area at the opposite end, or “pole” of H2O has a negative charge, the H2O molecule is called a POLAR MOLECULE. This polarity explains several things: Miscibility with other polar molecules. Miscibility means it mixes with other polar liquids, like vinegar. Immiscibility means it doesn’t mix, such as with vegetable oil and motor oil, or with other NON-POLAR liquids. Some materials are so non-polar that they hate being around the H2O molecule; they are called hydrophobic. Polar molecules attracted to the H2O molecule are called hydrophilic. Because the majority of substances are polar in nature, many of them dissolve in water. For example, sugar and salt stirred into water will dissolve to make salty or sweet water. Water dissolves so many things, the solids being called solutes, that water is called the universal solvent. Polarity also causes H2O molecules to be attracted to each other, and stick together. This is called cohesion. When H2O molecules stick to polar molecules that are other than H2O molecules, such as aluminum foil, that is called adhesion. Another feature of liquid water is its strong surface tension, which is a strong barrier at its surface thanks to the bonds between H2O molecules. This feature is what lets water striders walk on water, and a mound of H2O molecules build up on a penny. Capillary action is another result of water’s polarity. Capillary action is when a liquid climbs through or up a tube, despite the pull of gravity, to include the stems of plants, the bristles of a paint brush, even the small fibers that make up paper towel. The adhesion of the H2O molecules to the sides of the tube can be stronger than the pull of gravity, and the cohesion of the H2O molecules to each other help to pull up a column of water. Liquid H2O has its molecules in constant contact, with no special structure. They are in constant motion and constant contact. One result of being in constant contact is that water is not very compressible. Even at the bottom of the ocean, it’s density is close to the density at the surface. The density of fresh water is about 1 gm/mL, so anything denser than that will sink. However, when temperatures get lower and lower, almost to freezing, the molecules start to form a circle of 6 molecules, with their hydrogen ears facing out. At freezing, that circle of 6 becomes a rigid crystal. Since there is nothing at the center of the molecule, this explains why water EXPANDS when it freezes. Its volume expands. Since its mass stays the same, it is less dense than liquid water, by about 9%, and will float. Viscosity refers to the rate at which water will flow. The more viscous a liquid is, the slower it flows. Water is more viscous than most liquids. For example, it is 80 times more viscous than olive oil. Specific heat refers to water’s VERY HIGH ability to store heat and hold on to it. Water is second only to ammonia in its ability to store heat. For example, it holds heat much better than alcohol. This ability of water helps to explain why temperatures are more MODERATE close to the ocean. This means that in summer, areas near the ocean are not as hot as areas inland. During the winter, areas near the ocean are not as cold. Finally, the bond between the Hydrogen “ears” and the Oxygen are sometimes called “hydrogen bonds”, and help to explain why H2O is a universal solvent.