Ch2quiz1summer-1.munoz.ANSWERS
advertisement

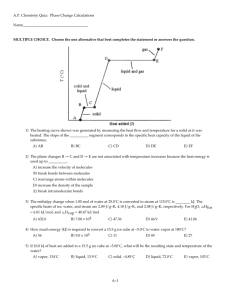
Chemistry Summer School Chapter 2 Quiz 1 2014 1. Describe the differences between a compound and an element. Is water a compound or an element? Explain your answer. Compound – combination / multiple types atoms or elements Element – pure substance / single type of atom or element 2. Ice at 0° Celsius is placed in a beaker and heated, at a uniform rate, on a hot plate. Heating continues until the boiling point of water is reached, and the water is allowed to boil for several minutes. Sketch a “heating curve” for this situation, and describe what is happening with regard to temperature, phase (state), kinetic energy and potential energy in each section. Describe: 1st Temperature will REMAIN CONSTANT and thermal energy will be used to break the forces between the SOLID H2O “ICE” molecules and MELTS at 0 DEGREES CELSIUS and becomes LIQUID H2O “WATER” molecules. 2nd Temperature will RISE due to the absorption of heat, the thermal energy is converted into INCREASED KINETIC ENERGY of each molecule. 3rd Temp will REMAIN CONSTANT as the energy breaks the molecules of the WATER apart and BOILS at 100 DEGREES CELSIUS and becomes VAPOR/GAS H2O “STEAM” OR “WATER VAPOR”. 3. Describe the process (steps) by which you could determine whether a liquid mixture was a solution, a suspension or a colloid. SEE WIKI ESSENTIAL QUESTION 2-3???