Covalent Bonding: Chapter 8 pg 212 8.1 Molecular Compounds
advertisement

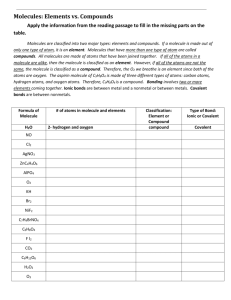
Covalent Bonding: Chapter 8 pg 212 8.1 Molecular Compounds Remember: When atoms join together, they form either _______________________ or ______________________. Ionic compound—Compound made of positive and negative _____, joined by ionic bonds. Molecule – A neutral group of atoms joined by ________________ bonds. Covalent bond -- A bond formed by _____________ of electrons between atoms. Many elements found in nature are in the form of molecules. Remember, molecules can be made of the same or different element(s). If the elements bonded together are different, we call it a _____________ _______________. If a diatomic molecule only has 2 atoms (whether they are the same element or not) we call it a ___________________ _________________. For example, O2 or HF. Atoms Elements Pure Substance Molecules (same element) Molecules (different element) Compound Ionic compounds Definitions to Review… Element- The simplest form of matter that has a unique set of properties; cannot be broken down any further into simpler substances; Can be present as a single atom or molecule. Binary Compound- Two different elements joined with either covalent of ionic bonds( so, either a molecule or an ionic compound). Compound- A substance that contains two or more different elements chemically combined in a fixed proportion; The atoms are joined by either covalent bonds (molecules) or by ionic bonds(ionic compounds). Molecular compound- A compound composed of 2 or more different elements covalently bonded. Molecular compounds tend to have relatively _______ melting and boiling points than ionic compounds. o Many molecular compounds are gases or liquids at room temperature. For example, H2O is a liquid at room temperature and CO is a gas at room temperature. Molecular Formulas A molecular formula is the ___________ formula of a _______________ compound. It shows how many _______ of each element a molecule contains. For example, H2O is the molecular formula for water. The _____________ written after the symbol indicates the ____________ __ _______ of each element in the molecule. For example: One molecule of ethane is C2H6. One molecule of ethane has 2 carbon atoms and 6 hydrogen atoms. A molecular formula reflects the ____________ number of atoms of each element in each molecule. This is not necessarily the lowest whole number ratio. * Remember when you did ionic compounds you always wrote the lowest whole number ratio of atoms. A molecular formula does not tell you the _____________ of the molecule. It doesn’t tell you how the atoms are arranged in _______, nor which atoms are covalently bonded to one another. So we use ______________ formulas, space filling models, _______________ drawings, or ______ _____ _______ models. 8.2 The Nature of Covalent Bonding. Remember in forming ionic compounds electrons tend to be transferred so each ion acquires a ______ _____ configuration. In forming covalent bonds, electron _____________ usually occurs so that atoms attain the electron configurations of noble gases. For example: Hydrogen atoms have one electron. But a pair of hydrogen atoms _________ these two electrons when they form a covalent bond in the hydrogen molecule. Each hydrogen atom thus attains the electron configuration of ____________ a noble gas with two electrons. Combinations of atoms of the nonmetallic elements in groups ____, ____, ____, and ____ of the periodic table are likely to form ____________ bonds. In this case the atoms usually acquire a total of ___ electrons (an octet) by _____________ sharing electrons , so the octet rule applies. Single Covalent Bonds The hydrogen atoms in a hydrogen molecule are held together mainly by the attraction of the shared electrons to the _______________ ________________. Two atoms held together by sharing a pair of electrons are joined by a ___________covalent bond. For example: H• + •H _______ Practice: Draw electron configurations for the following elements Carbon Nitrogen Oxygen Fluorine Neon Now, how many electrons would these elements need to share in order to achieve the same electron configuration as Neon? An electron dot structure such as _______ represents the shared pair of electrons of the covalent bond by two dots. The pair of shared electrons forming the covalent bond is also represented as a ________. For example, for hydrogen, _________.