Rationale
advertisement

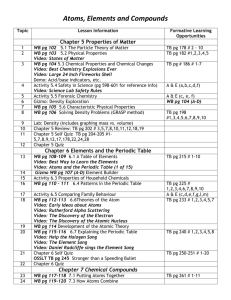
Monday Content Standard: SC3a discriminate between the size, charge, & position of protons, neutrons, and electrons SC3b use the orbital configuration of neutral atoms to explain chemical properties SC3c explain the relationship of the proton number to the element’s identity SC3d explain the relationship of isotopes to the relative abundance of atoms of a particular element. SC3f relate light emission and movement of electrons to element identification Essential Question: Have I mastered the standards of atomic structure? Musical Selection: RELAXATION: Pandora relaxation station Learning Activities: Summative Assessment: Unit Two Test Introduction to Unit Three Periodicity: Chem-To-Go Lesson 10 Rationale: As students finish the test at various times, each may move ahead individually. Summary: Students can watch the first video lesson of the new unit and complete the post-video online quiz. Differentiation Plan: NOTE: Today’s summative assessment hampers true classroom differentiation. Gathering data for tomorrow’s flexible grouping: Students will complete an online quiz after watching the video lesson, and teacher will use the results to form small groups for tomorrow’s lesson. Assessment Plan: Unit Two Test: Teacher will grade and record the test. Students will receive timely feedback. Online Quiz: Teacher will informally record the quiz grades and use them to create tomorrow’s groups. At the end of unit three, students may select this online quiz to go into Synergy. Homework: Watch Chem-To-Go Lesson 10, complete the Cornell notes, and take the post-video quiz. Tuesday Content Standard: SC4b: compare and contrast trend in the chemical and physical properties of elements and their placement on the Periodic Table. Essential Question: How are the elements arranged on the Periodic Table? Musical Selection: Twangy Tuesday Learning Activities: Activate Learning: Periodic Table Chalk Talk Rationale: Students often believe that they understand the arrangement of the periodic, but they struggle when asked to defend their answers using correct vocabulary and atomic structure details. Summary: Students will rotate through 6 learning stations to deepen their knowledge of the arrangement of the periodic table. Each station will ask the students to draw or write something in chalk on the blacktop table. Teacher will observe the answers of each group and clarify misconceptions. Partner Learning: Differentiated Tiered Activity Rationale: Students will be appropriately challenged to continue mastering the arrangement of the periodic table relative to chemical and physical properties. Summary: Students will be asked to self-report their understandings of today’s content from Level 1 to Level 2. Level One: Students will meet with teacher to discuss the basic anatomy of the periodic table. After answering questions and clarifying, they will be asked to construct a data table organizing their knowledge to use as a study tool for upcoming quizzes and tests. Level Two: Students will be asked to construct a periodic table using a set of cards with chemical and physical properties reported. Students will be challenged to select which properties they choose to highlight in their organization. No right answer exists. Differentiation Plan: Activate Learning: Periodic Table Chalk Talk o Flexible Grouping: Students are grouped based upon the data from last night’s online quiz. Teacher will monitor lower performing groups (on last night’s quiz) much more closely. Partner Learning: Differentiated Tiered Activity Rationale: Students will be appropriately challenged to continue mastering the arrangement of the periodic table relative to chemical and physical properties. Summary: Students will be asked to self-report their understandings of today’s content from Level 1 to Level 2. Level One: Students will meet with teacher to discuss the basic anatomy of the periodic table. After answering questions and clarifying, they will be asked to construct a data table organizing their knowledge to use as a study tool for upcoming quizzes and tests. Level Two: Students will be asked to construct a periodic table using a set of cards with chemical and physical properties reported. Students will be challenged to select which properties they choose to highlight in their organization. No right answer exists. Assessment Plan: Activate Learning: Periodic Table Chalk Talk o Flexible Grouping: Students are grouped based upon the data from last night’s online quiz. o Informal Assessment: Teacher will listen to student conversations and monitor student chalked answers to correct misconceptions and deepen understanding. Last night’s quiz data informs WHICH GROUPS to monitor most closely. Partner Learning: Differentiated Tiered Activity o Level One: Teacher will informally look over the student data tables and assist with corrections and questions. o Level Two: Teacher will informally monitor students’ reasoning for the organization of their periodic tables. Homework: Watch Chem-To-Go Lesson 11, complete the Cornell notes, and take the post-video quiz by midnight on Sunday. Wednesday Content Standard: SC4a: Use the periodic table to predict trends including atomic radii, ionic radii, ionization energy and electronegativity of various elements SC4b: Compare and contrast trends in the chemical and physical properties of elements and their placement on the Periodic Table Essential Question: How are elements arranged on the Periodic Table relative to atomic structure and properties? Musical Theme: Weird Science Wednesday Learning Activities: Active Learning: Creative Problem-Solving Extraterrestrial Chemistry Rationale: o ENRICH THE CONTENT: Teacher enriches periodicity content by making connections to the biochemistry of the human body and activates recall of concepts learned in 9th grade honors biology. o ENGAGE CRITICAL AND CREATIVE THINKING: Students engage in critical and creative thinking as they use the periodic table to create the new species by selecting elements similar in reactivity to those of similar function in the human body. o DIFFERENTIATE THE PRODUCT WITH RESEARCH-BASED STRATEGY: The Body Biography strategy developed by Underwood in 1987 is used to bring novelty to the content. The strategy is highlighted in Brunner’s 2012 book, “Now I Get It: Differentiate, Engage, and Read for Meaning.” Summary of Creative Problem-Solving: o Prior Instructional Delivery: APPROPRIATE INSTRUCTIONAL TECHNOLOGY: Teacher created video lesson using Camtasia screencasting software teaching the basic arrangement of the periodic table relative to atomic structure. Teacher posted the video, and students watched the video online on while completing guided Cornell notes. Teacher created short video understanding quiz, and students take the short quiz to inform self-evaluation and teacher preparation for Friday’s class. DEVELOP CRITICAL THINKING THROUGH QUESTIONING: Students worked in small homogeneous groups to interact with inquiry stations in class yesterday and completed a summarizing graphic organizer before leaving for the long weekend. o Procedure for Creative Problem-Solving: Read the information provided about the human species and its biochemistry. Use your knowledge of the periodic table and properties of elements to create an extraterrestrial species that is not carbon-based. Construct a body biography of a specimen of your extraterrestrial species. The body biography should communicate the biochemistry of the being with labels, illustrations, and symbols. o Body Biography Instructions: Draw the body of your ET. You may choose to trace the outline of a group member. Address the following anatomy/physiology with labels, creative illustrations, and symbols: Skeletal make-up Primary brain biochemistry Blood components Dietary needs: food and drink Breathing intake and output Components of the ET atmosphere Hygiene biochemistry: bathing and brushing teeth Components of digestive fluid Composition of perspiration Primary ions necessary for carrying electrical pulses Differentiation Plan: Appropriate challenge: Students will be grouped homogeneously by their current grade in the class. o Highest achievers: Students will receive instructions that invite them to research all of the components listed in the “summary” above. o Middle/Low achievers: Students will be invited to research more concrete and fewer components of human biochemistry. Assessment Plan: INFORMAL: Informal observations/conversations with students in individual and small group settings about the properties of elements on the periodic table and their choices as they develop their ETs; provide constructive feedback to groups as they work Homework: Review the anatomy of the periodic table. Be sure that you can complete the families checklist. Thursday Content Standard: SC4a: Use the periodic table to predict trends including atomic radii, ionic radii, ionization energy and electronegativity of various elements SC4b: Compare and contrast trends in the chemical and physical properties of elements and their placement on the Periodic Table Essential Question: How can I predict the relative atomic radius and ionic radius of an atom using the periodic table? Why does this pattern happen? Musical Theme: Throwback Thursday Learning Activities: HOOK: Periodic trend video clip – enrich the basic lab experiences available to our advanced learners Active, Inquiry Learning: Graphing Element Properties & Discovering Patterns [45 min] Rationale: o ACTIVE ENGAGEMENT & ACADEMIC RIGOR: Students actively engage in discovering the patterns of atomic radius and ionization energy by graphing given values for several elements. o DEVELOP CRITICAL THINKING THROUGH QUESTIONING: Higher level thinking skills will be enhanced as follow-up questions prompt students to draw connections between the graphs and elements’ locations on the periodic table. o RESEARCH-BASED STRATEGIES: Ritchhart, Church, and Morrison’s 2011 book “Making Thinking Visible” explains that making thought processes visible by asking students to diagram and illustration highlights misconceptions for teachers to address and ultimately increases student achievement. Active Learning & Communicating Scientific Concepts: Using Academic Language to Describe Periodic Trends Discovered in Graphing Lab [20 min] Rationale: o Summary: Period and group trends and why (using appropriate vocab) with same topic; Anchor Activity: Sample Multiple Choice Periodic Trends Questions Summary: Copies of sample questions are laminated and place under student desks. As a summarizing activity, students can pull out the sample questions to practice interpreting multiple questions. Differentiation Plan: Flexible, purposeful grouping: Students will be grouped homogeneously by postvideo quiz results. Active listening and attending to students’ needs and responses: Teacher will speak with each group about specific questions missed on the online quiz. Diagnostic assessment: Teacher will use exit questions to prepare for flexible grouping tomorrow. Assessment Plan: INFORMAL: Teacher will informally observe and question students during atomic and ionization energy inquiry graphing. Teacher will provide constructive suggestions for strengthening student explanations. SELF-EVALUATION: Students will be encouraged to revise and review their illustrations and answers tonight and score their work against teacher-provided models in class tomorrow. Homework: Watch Chem-To-Go Lesson 11, complete the Cornell notes, and take the post-video quiz. Friday Content Standard: SC4a: Use the periodic table to predict trends including atomic radii, ionic radii, ionization energy and electronegativity of various elements. SC4b: Compare and contrast trends in the chemical and physical properties of elements and their placement on the Periodic Table. Essential Question: How can I predict the relative atomic radius and ionic radius of an atom using the periodic table? Why does this pattern or trend occur? Musical Selection: Free Play Friday. Learning Activities: Assessment: Anatomy of Periodic Table Quiz 15 question multiple choice quiz Hook: Periodic Trends Rap Song: Video of AP chemistry student created rap song on the periodic trends. Students will watch and write down one of the rhymes used to describe one of the periodic trends of atomic/ionic radius, ionization energy, and electronegativity. After watching a few students will share what they wrote down - at least one rhyme for each of the trends. Kinesthetic Social Learning: Rationale: Teacher facilitates discussion about shielding of electrons from the effects of positive nuclear charge. Desks are used to visualize energy levels of electrons and front work bench will be symbolic of the nucleus. Laboratory Items will be added to work bench to visualize the addition of protons (to form different atoms) to the nucleus. Shielding across a period does not change but nuclear effective charge will increase pulling in the electrons to make a smaller radius for the atoms/ions as they move to the right in the periodic table. Active Learning: Atomic Radius and Ionic Radius Chalk Talk Learning Stations Rationale: ACTIVE ENGAGEMENT & ACADEMIC RIGOR: Students actively engage in learning the concepts of atomic radius and ionic radius by visiting stations that build upon their existing knowledge and skills. Many of the stations’ activities are designed to mimic questions and examples in the AP chemistry textbook. DEVELOP CRITICAL THINKING THROUGH QUESTIONING: Higher level thinking skills will be enhanced as stations prompt students to answer inquiry-style questions. RESEARCH-BASED STRATEGIES: Ritchhart, Church, and Morrison’s 2011 book “Making Thinking Visible” explains that making thought processes visible by asking students to diagram and illustration highlights misconceptions for teachers to address and ultimately increases student achievement. Summary: Prior Instructional Delivery [Flipped Classroom Model]: APPROPRIATE INSTRUCTIONAL TECHNOLOGY: Teacher created video lesson using Camtasia screencasting software teaching the basic arrangement of the periodic table relative to atomic structure. Teacher posted the video, and students watched the video online while completing guided Cornell notes. Teacher created short video understanding quiz, and students took the short quiz to inform self-evaluation and teacher preparation for today’s class. Chalk Talk Station Prompts: Draw an illustration explaining WHY elements on the left side of the periodic table are larger than elements in the same row to the right. Draw an illustration explaining WHY elements on the top of the periodic table are smaller than elements in the same column on the bottom. Draw an illustration explaining HOW and WHY the radius (size) of a neutral atom changes as the atom forms a cation. Draw an illustration explaining HOW and WHY the radius (size) of a neutral atom changes as the atom forms an anion. Use a periodic table to answer the sample multiple choice questions. Diagnostic Assessment: Collect formative, diagnostic data using five atomic radius/ionic radius exit questions. Data will inform tomorrow’s instruction and flexible groups. Differentiation Plan: V.A.R.K Lesson Plan: Visual, Aural, Read/Write, and Kinesthetic-Students have choice of which method to use to learn the material. Students will hear the content through a song while writing a rhyme to assist in remembering, kinesthetic demonstration and discussion using desks and students, and visual representation and wring during Chalk Talk. Flexible, purposeful grouping: Students will be grouped homogeneously by postvideo quiz from yesterday’s Chem-to-Go Lesson 11 results. Active listening and attending to students’ needs and responses: Teacher will speak with each group about specific questions missed on the online quiz. Diagnostic assessment: Teacher will use exit questions to prepare for flexible grouping tomorrow. Assessment Plan: Summative Assessment: Anatomy of periodic table quiz. Data will be used to determine which students need additional support and in which specific area. Formative Assessment: Chem-to-Go lesson 10 online quiz is used to determine grouping for today’s groups and required individual/small group support. Teacher will informally observe and question students during lab activity. Formative, Diagnostic Checkpoint: Students will answer 5 atomic radius/ionic radius periodic trend and calculation exit questions that range from very basic to advanced application of today’s learning. o Diagnostic: The results will be used to form small groups for tomorrow’s lesson. o Formative: Students will use the feedback to select appropriate topics and levels on tomorrow’s student-directed tiered assignment. Homework: Watch Chem-to-Go Lesson 12 and Cornell Notes: Ionization Energy and Electronegativity Lesson 12 video online quiz