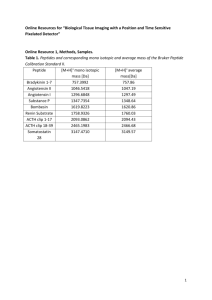
4-3 The Peptide Group
advertisement

4-3 THE PEPTIDE GROUP The Peptide Group Contains the two atoms involved in the peptide __________ along with their four substituents (the carbonyl __________ atom, the amide __________ atom, and the two adjacent __-__________ atoms). The peptide group can be represented by multiple Lewis structures, making them __________ structures. Figure A represents the common structure, Figure B represents the other contributing resonance structure, and the last is a hybrid representation, which best represents the resonance within the peptide group. Figure A Figure B Figure C This _______________ of electrons makes the C to O bond and the C to N bond behave like __________ bonds, restricting any _______________ around the atoms in the peptide group. The bonds between the carbonyl C and the -carbon, and between the N and the -carbon can still rotate __________. Restrictions on free rotation within the peptide group ultimately determine the __-__ _______________ of a protein. The double-bond nature of the peptide bond restricts the peptide group to either the _____ conformation or the _____ conformation. These conformations arise during protein _______________, and they cannot be interconverted around the peptide bond once it has formed. The _____ conformation is favored because there is less _______________ between the side chains in this conformation. Specific enzymes called peptidyl prolyl cis/trans isomerases can catalyze the interconversion of cis and trans conformations at _______________ residues. Human peptidyl prolyl cis/trans isomerase is the target of the immunosuppressive drug cyclosporin A, which is used with transplant patients to prevent the _______________ of the donated organ. Although nearly all peptide groups are in the trans conformation, interference can still occur with the rotation of the N–C and the C–C bonds, as shown in the following diagram: The letters (_____) and (_____) represent the angle of rotation allowed, which is restricted by the _______________ on the carbonyl group and by the __________ __________. Read pages 91 (3rd full paragraph) through page 92 (end of section) to learn how Ramachandran plots are used to determine which and angles are permissible in a polypeptide chain.