Unit 4 Take Home Test

Unit 4 Take Home Test: The Nucleus and Electrons DUE Monday, 10/6/14
Directions: Answer each question fully, use complete sentences. IF you are solving a problem, you must show all your work, and answers must be in correct significant figures and have units.
1.
Compare and contrast the three types of radioactive decay using the following properties: a.
Type of particle b.
How fast the particles move c.
How harmful the particles are in terms of what can stop the particle.
2.
What part of the atom undergoes change during radioactive decay?
3.
Explain what happens when atoms undergo fission. Give 2 examples where fission occurs.
4.
Explain what happens when atoms undergo fusion. Give 2 examples where fusion occurs.
5.
Explain how a nuclear fission reactor works to create electricity.
6.
Fusion reactors can produce much more energy than fission reactors can, yet as of right now; fusion reactors are not used to generate electricity. Elaborate on why this is the case.
7.
Draw and label/explain the Bohr model of the atom. Draw and label/explain the quantum mechanical model of the atom.
8.
How many energy levels are possible in an atom?
9.
Describe the 4 types of sublevels possible within the energy levels of an atom?
(shape of the simplest two sublevels, how many electrons can exist within each sublevel, etc.)
10.
Write electron configurations for the following elements. You can write them long hand or using the shorter Noble Gas configurations. a.
Carbon, C b.
Scandium, Sc c.
Niobium, Nb d.
Tin, Sn e.
Samarium, Sm
11.
According to quantum mechanics, an electron acts as both a particle and a wave
(or wave function). Explain in detail.
12.
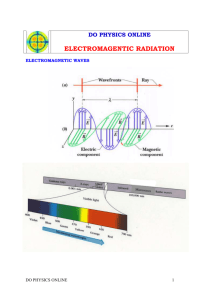
Draw and label a wave with a large wavelength and low frequency. Draw and label a wave with a small wavelength and a high frequency.
13.
Which color of visible light has the largest wavelength: red, yellow, or blue?
14.
Using the three colors in #13, red, yellow, and blue; which color of light has the highest frequency?
15.
Explain the relationship between wavelength and frequency.
16.
Calculate the wavelength of the yellow light emitted by a sodium vapor lamp if the frequency of the radiation is 5.10 × 10
14
1/s.
17.
A mercury lamp emits light with a wavelength of 4.36 × 10
-7 m. What is the frequency of the light?
18.
Relate the energy in a photon released by excited electrons in an atom to the frequency of the colored light produced.
19.
Explain how you can identify an element using the movement of electrons and the color of light emitted by an element.
20.
What is the energy of a photon of green light emitted by a copper atom with a frequency of 5.80 × 10
14
1/s?
21.
Long range radio waves have been sent out into space by SETI with the hope that another civilization will recognize them and respond that we are not alone in the universe. The frequency of the radio waves is 3.00 × 10
6
1/s. How much energy is being sent into space?
22.
How many protons, electrons, and neutrons are in an atom of each isotope? a.
59
26
Fe b.
c.
235
92
U
52
24
Cr
23.
The density of gold is 19.3g/cm
3
. What is the mass, in grams, of a cube of gold that measures 2.0cm on each side?
24.
Arrange these elements in order of decreasing atomic size: sulfur, chlorine, aluminum, and sodium.
25.
Classify each of the following as a physical or chemical change: a.
Bending a piece of wire b.
Burning coal c.
Cooking a steak d.
Cutting grass