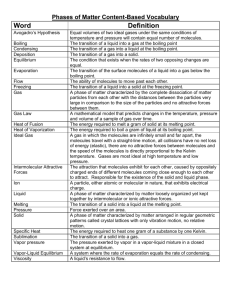
It is the change of matter from solid state to liquid state Melting
advertisement

1. Matter : It is anything that has a mass and a volume 2. Density : It is the mass of volume unit of matter 3. Melting point : It is the temperature at which the matter begins to change from solid state to liquid state 4. Boiling point : It is the temperature at which the matter begins to change from liquid state to gaseous state 5. Melting : It is the change of matter from solid state to liquid state 6. Boiling : It is the change of matter from liquid state to gaseous state 7. Molecule : It is the smallest part of matter which can exist freely and it has properties of matter 8. Element : It is the simplest pure form of matter which can be analyzed chemically into simpler form 9. Compound : It is a substance which is formed from combination of atoms of different elements with constant weight ratios The density of milk = 1.03 gm/cm3 The density of water = 1 gm/cm3 The melting point of ice = 0 c The boiling point of water = 100 c The mass of 1 cm3 of milk is 1.03 gm The mass of 1 cm 3 of water = 1 gm The ice begins to change into water at 0 c The water begins to boil and change into water vapour at 100 c 1. Color, taste and odour cannot be used to differentiate between water and oxygen gas. Because both of water and oxygen are colorless, tasteless and odourless 2. A piece of wood floats on water while a piece of lead sinks in it Because the density of wood is less than the density of water while the density of lead is more than the density of water 3. Ice floats on water surface although both are from one matter Because the density of ice is less than the density of water 4. Water is not used to put out petrol fires Because the density of petrol is less than the density of water so petrol floats on water surface 5. Balloons filled with hydrogen and helium rise up n air?Because the density of hydrogen and helium are less than the density of air 6. A piece of ice melts when it is left in air Because the melting point of ice is low 7. Hardness can be used to distinguish between rubber and gold Because the rubber is soft at room temperature while gold is not 8. Iron rods not copper rods are used to in building houses concrete Because the hardness of iron is more than the hardness of copper 9. Electric wires are made of copper or aluminum and they are covered by a plastic layer Because copper and aluminum are good conductors of electricity but plastic s a bad conductor of electricity 10.Screwdrivers are made of steel iron while their handles are made of wood or plastic Because steel iron is good conductor of electricity but wood and plastic are bad conductor are bad conductor of electricity 11.Cooking pans are made of aluminum Because the melting point of aluminum is high and it is good conductor of heat 12.Handles of cooking pans are made of wood or plastic Because each of them is bad conductor of heat 13.Potassium and sodium are kept under kerosene surface To prevent their reaction with oxygen in humid air 14.Steel bridges and the holders of light bulbs are painted from time to time Metallic spare parts of cars are covered with grease To protect them from rust and corrosion 15.Cooking pans made of aluminum are washed with a rough material To remove any layer formed on them as a result of their reaction with the oxygen of humid air 16.Silver and gold are used in making of jewels Because they are chemically poor active 17.Active metals such as sodium lose their metallic luster when they are exposed to moist air Because they react easily with atmospheric oxygen 18.When you leave the per fume bottle open you smell it all over the room Because the molecules of the perfume are in continuous motion and they keep the properties of the perfume 19.A drop of ink (potassium permanganate) spreads through water Because the molecules of ink or potassium permanganate are in continuous motion 20.The volume of a mixture of water and alcohol is less than the sum of their volume before mixing Because some molecules of alcohol occupy the intermolecular spaces among water molecule 21.It is very hard to fragmentize apiece of iron with your fingers while it is easy to divide an amount of water into small droplets Because intermolecular force among the iron molecules is very strong while that is among water molecules is weak 22.Solid substances have definite shape and volume Because the intermolecular spaces among its molecules are very narrow and the intermolecular force is very strong 23.The liquid matter takes the shapes and volumes Because the intermolecular spaces among its molecules are little big and the intermolecular force among its molecules is weak 24.Gases have indefinite shapes and volumes Because the intermolecular spaces among its molecules are large big and the intermolecular force among its molecules is very weak 25.The matter changes from liquid state into gaseous state by heating Because when a liquid substance is heated its molecules gain more energy and its speed increases and at boiling point some of them overcome the inter molecular force so they escape in the form of vapour 26.The matter changes from solid state into liquid state by heating Because when a solid substance is heated its molecules gain more energy and its speed increases and at melting point some of them overcome the inter molecular force and become more freely so it changes into liquid state 27.Oxygen is an element while table salt is a compound Because oxygen molecule is formed of two similar atoms while table salt molecule is formed of two different atoms Points of comparison Definition Element Compound It is the simplest pure form of matter which cannot be analyzed chemically into simpler form It is a substance which is formed from combination of atoms of two or more different elements with constant weight ratios Atoms Similar Different Examples Hydrogen, Oxygen, aluminum and sulphur Water, carbon dioxide, sodium chloride Points of The solid state comparison Motion of limited molecules Intermolecular Very small spaces (narrow) Intermolecular Very strong forces Volume Definite Shape Definite Examples Iron - aluminum The liquid state The gaseous state More free Completely free Intermediate Very large Intermediate Very weak Definite Indefinite Water – oil – alcohol Indefinite Indefinite Water vapour – oxygen 1. Calculate the mass of a piece of sulphur its volume is 5 cm3 knowing that the density of sulphur is 2.1 gm/cm3 2. In an experiment for determining the density of water the following results were recorded Mass of empty baker = 65 gm Mass of the beaker and water = 165 gm The volume of water = 100 cm3 Calculate the density of water