Analysis of a Mixture of Carbonate and Bicarbonate
advertisement

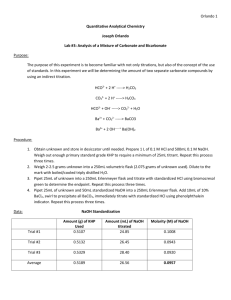
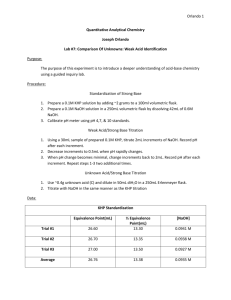
Matthew Marthaler Experiment Three Analysis of a mixture of Carbonate and Bicarbonate Purpose: In this lab, the students will learn the technique of titration. They will also learn the concepts of standards and how to use them. Lastly, they will determine the amount of the two different carbonate species via an indirect titration method. Procedure: 1) Obtain an unknown of carbonate and store it in the desiccator until it is needed. Standardize the acid and base now by preparing approximately 0.1 M HCl (1 L) and approximately 1 M NaOH (500 mL) using primary grade KHP which is drying in the oven. Accurately weigh enough KHP to require at least 25 mL of titrant. Repeat until three good trials are done. Now that the base is standardized, use this to standardize the acid. 2) Weigh 2.0-2.5g of unknown into a 250 mL volumetric flask. Dilute to mark with freshly boiled and cooled triply distilled water. 3) Pipet a 25.00 mL aliquot of unknown into a 250 mL Erlenmeyer and titrate with standardized HCl using bromocresol green to determine the endpoint. Repeat until at least three good trials are obtained. 4) Pipet 25.00 mL aliquot of unknown and a 50.00 mL aliquot of unknown into a 250 mL Erlenmeyer flask. Add 10 mL of 10% weight BaCl2, swirl to precipitate all BaCO3, then titrate with standard HCl using phenolphthalein indicator. Repeat until at least three good trials are done. 5) Report relative percent carbonate and bicarbonate into unknown with appropriate standard deviation values. Include all appropriate chemical equations into the lab notebook. Reactions: HCO3- + 2H+ H2CO3 CO32- + 2H+ H2CO3 HCO3- + OH- CO32- + H2O Ba2+ + CO32- BaCO3 Ba3+ + 2 OH- Ba(OH)2 Data for Unknown C: Standardization of NaOH using KHP KHP Trial Number KHP (g) 1 2 3 Average: 0.5108 0.5102 0.5102 0.5104 Volume of 0.1 M NaOH (mL) Trial One 26.62 26.09 21.90 24.87 Volume of 0.1 M NaOH (mL) Trial Two 26.62 26.63 26.62 26.62 M NaOH 0.09396 0.09381 0.09384 0.09387 Standardization of HCl using NaOH Trial Number 1 2 3 Average: Volume HCL (mL) 25.00 25.00 25.00 25.00 Volume NaOH (mL) 26.62 26.39 26.49 26.50 M HCl 0.10049 0.09903 0.09943 0.09965 Mass of Unknown C 1: 2.1035 g 2: 2.1135 g 3: 2.1134 g Titration of 25.00 mL Unknown C by HCl Trial Number (25.00 mL of unknown with the same number) 1 2 3 Average: Volume of HCl (mL) Bromocresol 29.13 29.01 28.31 28.82 Titration of 25.00 mL of Unknown C in 50.00 mL of NaOH and 10 mL of 10% weight BaCl2 by HCl Trial Number Volume of HCl (mL) First Trial Phenolphthalein 28.90 27.51 28.01 28.14 1 2 3 Average: Volume of HCl (mL) Second Trial Phenolphthalein 27.40 27.51 27.90 27.60 Calculations: Average mL for each titration set: Σxi n Ex (average mL for titration of HCl in NaOH): 26.62+26.39+26.49 3 Standard Deviation and Percent Relative Deviation: Ex (titration of HCl in NaOH): S= √(26.62−26.50)2 + (26.29−26.50)2 + (26.49+26.50)2 √2 % relative deviation: 0.115 26.50 x 100% = 0.444% Molarity of NaOH: Ex: 0.5108 g KHP x 1 mol KHP 204.2212 g KHP x 0.02662 Molarity of HCl : Ex : = 0.09396 M M NaOH x V NaOH V HCl 0.09396 x 0.02662 0.02500 =0.100049 M Mass of NaOH (used in step one): 0.1 mols NaOH x 0.5 L x 39.997 g KHP 1 L NaOH x 1 mol NaOH = 1.999 g NaOH Mass of KHP (used in step one): 0.1 mol HCl x 0.025 L x 204.2212 g KHP 1 L HCl x 1 mol KHP = 0.5106 g KHP = 0.115 = 26.50 mL Moles of Carbonates (CO32-) and Bicarbonates (HCO3-): Ex: 0.1000486 M HCl x 0.02913 L HCl (bromocresol) = 0.00291 moles Moles of NaOH initial: Ex: 0.09396 M NaOH x 0.0500 L NaOH (added) = 0.00470 mol NaOH initial Moles of NaOH excess: Ex: 0.1000486 M HCl x 0.02740 L HCl (Phenolphthalein) x 1 mol NaOH 1 mol HCl = 0.00274 mol NaOH Moles of NaOH reacted with HCO3-: Ex: 0.00470 mol initial – 0.00274 mol excess = 0.00196 mol NaOH reacted Moles of Bicarbonate (HCO3-): Ex: 0.00196 moles NaOH x 1 mol Bicarbonate = 1 mol NaOH 0.00196 mol Bicarbonate Moles of Carbonate (CO32-): Ex: 0.00291 mol carbonate/bicarbonate – 0.00196 mol bicarbonate = 0.00095 mol carbonate Relative Percent of Carbonate: Ex: 0.00095 mol carbonate x 60.007 g Carbonate x 1 1 mol carbonate x 2.1035 g Unknown C x 100% = 2.71 % Average: 2.59% Relative Percent Bicarbonate: Ex: 0.00196 mol bicarbonate x 61.014 g bicarbonat x 1 1 mol bicarbonate x 2.10365 g Unknown C x 100% = 5.69% Average: 5.65% Standard deviation of percent Carbonate: √ (0..0271− .0259)2 + (0.0253−0.0259)2 + (0.0253−0.0259)2 2 = 0.00104 Standard deviation of percent Bicarbonate √ (0.0569−0.0565)2 + (0.0572−0.0565)2 + (0.0554−0.0565)2 2 = 0.000964 Conclusion: The purpose of this lab was to determine the amount of carbonate and bicarbonate in unknown C. Upon the completion of this lab, the calculations show an average of 5.65% bicarbonate and 2.59% carbonate. The standard deviation of the percent carbonate was 0.00104 and the standard deviation of the percent bicarbonate was 0.000964. This shows that our calculations are precise, because the standard deviation is very small, so we did not make any huge errors that would make our trials differ significantly. I have no idea, however, if those numbers are accurate. Post-Lab Questions: 1) A primary standard is a reagent that is extremely pure, stable, has no waters of hydration, and has a high molecular weight. I got this definition from the Department of Chemistry at Adelnide University’s website. This primary standard also can calibrate other standards. 2) A secondary solution is a standard that has been titrated against a primary standard. A standard solution is a secondary standard. I got this definition from chemistry-Dictionary.com. 3) An indirect titration is the process of determining the concentration of an analyte by reacting it with a known number of moles of excess reagent. The excess reagent is then titrated with a second reagent. I got this definition from Chemistry-Dictionary.com. 4) Titrant is a solution of known concentration which is added (titrated) to another solution to determine the concentration of a second chemical species. I got this definition from Chemistry.about.com.