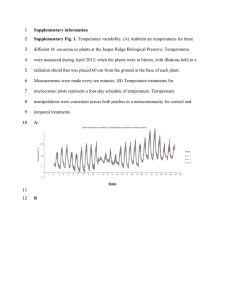
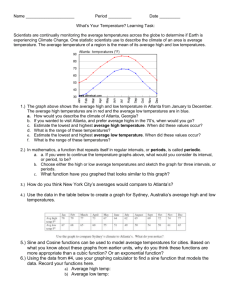
copy of Chapter 4 topics, key words and class notes.
advertisement

4.1 Describing Temperature (p. 110 – 116) Temperature describes how hot or cold an object is. Common temperatures include: 0 ⁰C – freezing/melting point of water 20 – 23 ⁰C – comfortable room temperature 37 ⁰C – normal body temperature of humans 100 ⁰C – boiling point of water. Body temperature is an important indicator of health. Human body temperature is about 37⁰C. Different mammals have different body temperatures (e.g. dogs are ~ 38⁰C and birds are ~ 41 ⁰C). Hypothermia occurs when a person’s body temperature drops. This causes the heart to slow down and organs will stop working properly. Hyperthermia occurs when a person’s body temperature rises. This indicates an infection or an illness. Bacteria and germs can be destroyed by an increase in temperature. Hypothermia and hyperthermia can be life threatening. Air temperature ranges from cold to hot temperatures. Air temperature depends on location, seasons, and time of day (night vs. day). Wind chill is the “feels like” temperature. It is a combination of air temperature + wind speed. When the wind blows, it moves a warmed layer of air away from your skin and replaces it with colder air. The colder air draws more heat from your skin making it feel even colder. The faster the wind, the colder the wind chill. 4.2 Measuring Temperature (p. 120 – 127) You can use your sense to gauge how hot or cold an object is. For example, smell the odour of something cooking see the glow of the coils in the oven feel the heat of an oven The first temperature sensing devices were developed in the 1500s. Thermoscope a device that shows if the temperature of an object is increasing or decreasing. DO NOT have scales so we are not able to use them to determine a precise (specific) temperature. Examples: (1) Air Thermoscope Galilei (1596) When the air temperature rises, the liquid drops. This is because warm air expands (needs more room) and as a result, the liquid is pushed down. When the air temperature drops, the liquid rises. This is because cold air contracts (takes up less room) and a result, the liquid is able to rise. (2) Liquid Thermoscope Similar to thermometers we use today. Liquid will expand (rise up) when heated and contract (drop) when cooled. Thermometer - a device that measures the specific temperature of an object. Includes a scale. The commonly used temperature scales are the Fahrenheit scale, the Celsius scale, and the Kelvin scale. Fahrenheit Scale Gabriel Fahrenheit (1686–1736) This was the first scale to be widely used. United States still uses this scale today. To create this scale they used two common reference points: o 1st reference point – 0⁰F. A mixture of salt/ice/water (coldest substance they could create in the lab) o 2nd reference point was ~ 100 ⁰F. This is the approximate temperature of the human body. Other common temperatures: o 32 ⁰F – freezing point of water o 98.6 ⁰F - actual human body temperature o 212 ⁰F - boiling point of water Celsius Scale Anders Celsius (1701–1744) Studied the freezing point and boiling point of water at different atmospheric pressures. o Lower atmospheric pressure (e.g. going up a mountain) – water boils at lower temperatures (e.g. at the top of Mount Everest, water boils at 69 ⁰C). o Sea-level atmospheric pressure – water boils at 100 ⁰C. Most commonly used temperature scale Common temperatures: o 0⁰C – freezing/melting point of water o 20 – 23 ⁰C – comfortable room temperature o 37 ⁰C – human body temperature o 100 ⁰C – boiling point of water Kelvin Scale William Thomson (Lord Kelvin; 1800’s) Studied the effects of changing temperatures on gasses Came up with the idea of absolute zero – the coldest temperature possible Absolute Zero – 0K (0 Kelvin) Thermometers must be calibrated by exposing them to substances at known temperatures. To calibrate an instrument means to accurately assign the numbers on a scale. Calibrating a thermoscope involves marking the level of the liquid at 0 ⁰C (freezing point of water) and 100 ⁰C (boiling point of water) and then using a scale to evenly mark the temperatures between these two points. Thermometers must contain a substance that changes as the temperature changes so that the change can be observed. Liquid Thermometer Can be filled with mercury or coloured alcohol. The liquid expands (takes up more room - rises up) when heated. The liquid contracts (takes up less room – goes down) when cooled. Bimetallic Strip Made of two different types of metals (e.g. copper and iron) that are fused together. The metals expand and contract at different rates causing the strip to bend at different temperatures. A thermostat uses a bimetallic strip to turn the furnace on and off! As the air temperature rises . . . o The bimetallic strip expands, which tilts the glass. o The mercury will flow away from the contacts, breaking the circuit, and turning off the furnace. When the temperature drops back down . . . o The bimetallic strip contracts, which tilts the glass (opposite direction). o The mercury will flow to the contacts, completing the circuit, and the furnace turns on. Thermocouple Made of two wires of different metals that are joined together at one end. When they are heated an electrical current is created. The higher the temperature the more current is produced. This current can turn on and off a switch or valve. Infrared Thermometer Converts infrared radiation into colors that can interpret a temperature difference. Can be used to detect heat loss in your home.