Stoichiometry - Fulton County Schools

Department: _______Science_______________ Teacher: ___Davis___________________
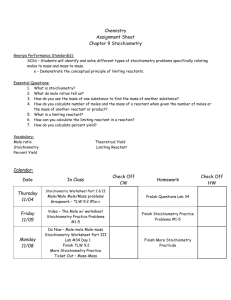
Topic / Theme: ____Stoichiometry____________________________________________
Essential Question:
What is the relationship between the quantities of reactants and products in a chemical reaction?
Course: ____Honors Chemistry______________________
____ Duration:___2-3 weeks________________________
Key Questions:
How do you solve problems relating mass, moles, number of representative particles or liters of gas at STP between any two substances involved in a chemical reaction?
How do you determine the limiting and excess reactant of a reaction given the starting amounts of each reactant?
How do you determine the theoretical yield of a reaction given the starting amount of each reactant?
How do you determine the percent yield of a reaction given the actual yield and starting amount of each reactant?
How do you determine the amount of excess reactant leftover after a reaction ?
How is the empirical formula of a hydrocarbon determined based on the amount of products produced?
Determine the purity of a mineral based on the products produced in a reaction with a known quantity of another substance (multi-step stoichiometry problem)
Essential Vocabulary:
Mole, Avogadro’s number, hydrate, molar mass,
Empirical formula, molecular formula, percent composition, molar volume, standard temperature and pressure, atom, molecule, formula unit, stoichiometry, limiting reactant (reagent), excess reactant (reagent), mole ratio, conservation of mass, theoretical yield, actual yield, percent yield, hydrocarbon
Standards (List the primary standard and the key verbs from the elements)
SC2 Students will relate how the Law of Conservation of Matter is used to determine chemical composition in compounds and chemical reactions. d. Identify and solve different types of stoichiometry problems, specifically relating mass to moles, mass to mass and mass to volume. e. Demonstrate the conceptual principle of limiting reactants.
Higher-Order Student Engagement
(Specific activities/tasks that will actively engage students in higher order learning)
Call on specific students with content specific questions every day.
Homework Problems.
Unit Review Homework/Study Guide.
Practice Problems (Worksheet)
Laboratory Activities.
Unit Test
(honors chemistry tasks require students to apply knowledge to solve problems)
Assessment of Learning Goals : Formative and Summative
(How do you know if your students have learned?)
Quizzes on non-limiting reactant stoichiometry
Quizzes on limiting reactant stoichiometry
Quizzes on conservation of mass problems
Call on specific students with content specific questions every day.
Nightly Homework Problems.
Unit Review Homework/Study Guide.
Laboratory Activities.
Unit Test
Laboratories
Conservation of mass lab
Gas Stoichiometry (Mg + HCl) at STP
PA – Stoichiometry lab (Fe + CuSO
4
)
We empower students to discover, think, and succeed.
GPS Standards (Optional)
SC2 Students will relate how the Law of Conservation of Matter is used to determine chemical composition in compounds and chemical reactions. d. Identify and solve different types of stoichiometry problems, specifically relating mass to moles, mass to mass and mass to volume. e. Demonstrate the conceptual principle of limiting reactants.