27 - Colligative Properties - 1 - Chemistry

Colligative Properties
Electrolytes
Substance whose aqueous solution conducts electricity.
Ions carry the electric current, MUST HAVE IONS.
Non-electrolytes – do not conduct electricity, do not break into ions.
Colloids
“Gluelike” Elmer’s glue is a colloid (do not always need a glue however)
Mixture of 2 phases of matter: continuous and dispersed phases
Between solution and suspension
Has subcategories: o Aerosols – liquid or solid in a gas
Fog (liquid), smoke (solid) o Foams – gases in liquids or solids
Shaving cream, whipped cream (liquids), marshmallows (solid) o Emulsions – liquids in liquids
Mayonsaise, milk o Sols – solids in liquids
Paints, gelatin, clay, jellies (w/ out preserves)
Tyndall Effect: the scattering of light by colloidal or suspended particles
Brownian Motion: the random movement of colloidal particles due to their bombardment
(bumping into collisions) by the molecules of the dispersing medium.
Ex: hitting a balloon to keep it from touching the ground
Suspension
Heterogeneous mixture that will settle out
Can’t see through it
Will settle
Questions to ask:
1) Can you see through it? a.
Yes: solution b.
No: colloid OR suspension
2) Will it settle? a.
Yes: suspension b.
No: Colloid
Type
Solution
Colloid
Suspension
Colligative Properties
Effect on light
Does not scatter
Scatters
Scatters
Particle size
Less than 1 nm
1 – 100 nm
Greater than 100 nm
Permanence
Permanent
Permanent
Settles out
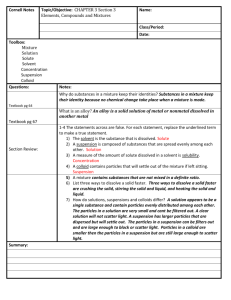
Properties determined by the # of particles in solution (rather than the type)
*# of particles of solute in solvent will cause the following* o Vapor pressure decreases o Boiling point increases o Freezing point decreases
Higher vapor pressure pure solvent
More solute = lower vapor pressure
**type of solute is note important, what is important is how many particles the solute contributes .
Some particles create more particles than other upon dissolving o Creates more ions more temperature change
NaCl (s) Na + (aq) + Cl
-
(aq) 2 ions formed
CaCl
2
(s) Ca 2+ (aq) + 2 Cl
-
(aq) 3 ions formed
C
6
H
12
O
6
(s) C
6
H
12
O
6
(aq) 1 molecules formed
The change in vapor pressure, freezing and boiling point is directly proportional to the number of particles dissolved.
Distillation
Separates substances with different boiling points.