Heart Rate Variability test
advertisement

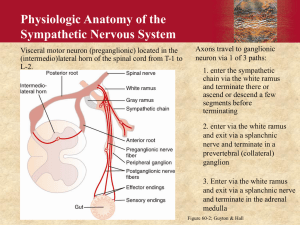
HEART RATE VARIABILITY TEST Introduction Heart Rate Variability (HRV) is defined as the beat to beat timing changes that occur between heart beats. In other words the time between heart beats changes from one beat to the next. Changes in beat intervals are measured in milliseconds or 1/1000th of a second. It is this timing change or rate change (variability) that gives us the term Heart Rate Variability. For most of us, the idea that the time between heart beats is variable, contradicts what we have commonly known. Most of us learned to take a radial, carotid, or popliteal pulse, and that the pulse rate equaled the heart rate, and that rate was fairly constant. This is not the case, and PULSE RATE ≠ HEART RATE. In short, heart rate variability is just what the name implies. It is the continuous (variable) changes in heart rate or heart beat frequency over a given period of time. To clarify, most of us assume that if we take our pulse, and the pulse rate is 80 beats in a 60 second period, than the heart is beating 80 times per minute at a fairly constant rate. This is not true. Pulse rate is the number times that the you feel blood surging through the artery that is being palpated. For the purpose of HRV, Heart Rate is the number of times the ventricles depolarize, or the number of electrical discharges that occur in the ventricles in a 60 second period. History of Heart Rate Variability Heart rate variability was invented in 1965 when Hon and Lee noted that fetal distress was preceded by alterations in interbeat intervals before any appreciable change occurred in the heart rate itself. Then Sayers and others focused attention on the existence of physiological rhythms imbedded in the beat-to-beat heart rate signal. During the 1970s, Ewing et al. devised a number of simple bedside tests of short-term heart beat differences to detect autonomic neuropathy in diabetic patients. The association of higher risk of post-infarction mortality with reduced HRV was first shown by Wolf et al. in 1972. In 1981, Akselrod et al. introduced power spectral analysis of heart rate fluctuations to quantitatively evaluate beat-to-beat cardiovascular control. These frequency–domain analyses contributed to the understanding of the autonomic nervous system's effect on RR interval fluctuations in the heart rate record. The clinical importance of HRV became apparent in the late 1980s when it was confirmed that HRV was a strong and independent predictor of mortality following an acute myocardial infarction. Physiological Basis of Heart Rate Variability The physiological basis for HRV rest in the fact that in addition to the normal heart cells responsible for contraction, there are also very specialized cells that are responsible for starting each contraction and for rapid and coordinated excitation of the heart muscle. These cells are collectively called the conduction system, which is comprised of the following structures: Sinoatrial node, atrioventricular node, atrioventricular bundle (Bundle of His), right and left bundle branches, and small conduction fibers called Purkinje fibers. The sinoatrial node (SA node) is often called the pacemaker of the heart, and depolarizes or fires to start the contraction of the heart muscle. It is located in the right superior aspect of the right atrium, and is under direct control of the autonomic nervous system. At rest it fires about 75-100 times in a 60second period. After the SA node fires the electrical signal travels to both atria and results in their simultaneous contraction. The electrical signal then passes through an area of nonconductive fibrous tissue via the AV node which slows down the speed of the electrical signal. This delay is what allows the ventricles to fill with blood before their collective contraction as discussed earlier. The signal then reaches the bundle of HIS and is divided into left and right bundle branches, which carry the electrical impulse to the bottom portions of the ventricles, causing the lower portion of the ventricles to contract first. This is followed by the middle and upper portion of the ventricles which therefore squeezes the blood out of the heart and off to the lungs and the systemic circulation Additionally, the SA node is under direct control of the Autonomic Nervous System which is the part of the nervous system that non-voluntarily controls all organs and systems of the body. Remember that the human nervous system is divided in two ways, structurally, and functionally. Structurally, the nervous system is divided into two parts, the central nervous system (CNS) and the peripheral nervous system (PNS). The central nervous system is comprised of the brain and spinal cord. The peripheral nervous system is comprised of all of the other nerves that branch from the brain and spinal cord, including the cranial nerves, and spinal nerves. Functionally, the nervous system is divided into the autonomic nervous system and the voluntary nervous system. The voluntary nervous system is involved with actions or movements that are under our direct conscious control. The autonomic nervous system, or automatic nervous system, is the portion of the nervous system that controls all of the bodily functions that are not under direct conscious control i.e., heart rate, breathing, eye blinking, etc. It is this portion of the nervous system that we will be focusing on throughout the rest of this course The autonomic nervous system is divided into two parts or branches, the sympathetic branch and the parasympathetic branch. We will concentrate on the sympathetic branch first. In general the sympathetic branch is responsible for increasing or speeding up bodily functions, such as heart rate, respiratory rate, pupil dilation, and increase blood flow to the extremities. At the SA node, the sympathetic nervous system’s primary function is to increase SA node depolarization or firing, which in turn speeds up the heart rate. The parasympathetic branch is responsible for slowing down heart rate and respiratory rate, decreasing blood pressure, and shunting blood from the extremities to the digestive organs. The parasympathetic nerve supply to the heart is primarily derived form cranial nerve ten (X) also know as the vagus nerve. The vagus nerve sends preganlionic fibers directly to the terminal ganglia located on the heart muscle and then postganglionic fibers influence the SA node from there. Parasympathetic stimulation of the SA node decreases its depolarization rate, or firing rate, and thereby slows down the heart rate. At rest both sympathetic and parasympathetic systems are active with parasympathetic dominance. The actual balance between them is constantly changing in an attempt to achieve optimum homeostasis despite ever changing external and internal conditions. In other words the two branches of the ANS are constantly working against each other like an old married couple arguing. Who Can Benefit From HRV Testing Patient's with the following conditions may benefit from Autonomic Nervous System testing. Abnormal reflex Migraines and other headaches AIDS Depression or Bipolar Disease Anxiety Post-traumatic Stress Syndrome Attention Deficit Disorder Fibromyalgia Hypertension Post-MI Angina Atherosclerosis Mitral Valve Prolapse Syndrome Cardiomyopathy Cardiac Dysrhythmias Congestive Heart Failure Acquired Hypothyroidism Thyroid Disorders Premature Menopausal Symptoms Menopausal Syndromes Sleep Apnea Asthma COPD Sleep Disorders Manifestations of thiamine deficiency Paralysis agitans Cerebellardegeneration Spinocerebellar Amyotrophic diseases Syringomyelia Diseases of the spinal cord Idiopathic peripheral autonomic neuropathy Multiple sclerosis Disorders of Vagus Nerve. Mononeuritis Peripheral & Poly-Neuropathies Diabetes Mellitus Chronic Pain Orthostatic & Chronic Hypotension Neurogenic bladder Urogenital Dysfunctions Anhidrosis Sjögren’sdisease Syncope and collapse Chronic fatigue syndrome Tachycardia (postural) Methods of Measuring Heart Rate Variability Time Domain Measurement This method analyzes either the heart rate at any point in time, or the intervals between successive normal QRS complexes. In a continuous electrocardiographic (ECG) recording, each QRS complex is detected, and the so-called normal-to-normal (NN) intervals (that is all intervals between adjacent QRS complexes resulting from normal sinus node depolarizations), or the instantaneous heart rate is determined. The so called normal complexes are considered to be normal sinus rhythm beats of the heart. This excludes any premature beats, late beats, or ectopic beats that are collectively know as cardiac arrhythmia. Simple time domain variables that can be calculated include the mean or average NN interval, the mean heart rate, the difference between the longest and shortest NN interval, the difference between night and day heart rate, etc (only in a 24 hour recording). Other time domain measurements that can be used are variations in instantaneous heart rate secondary to respiration, 90° passive tilt, Valsalva maneuver, spinal manipulation, acupuncture therapy, and injection or oral administration of various medications. The simplest variable to calculate is the standard deviation of the NN interval (SDNN), which is the square root of variance. Variance is mathematically equal to total power of spectral analysis (discussed below), therefore SDNN reflects all the cyclic components responsible for variability in the period of recording. To put it another way SDNN represents both the sympathetic and parasympathetic influences on heart rate. SDNN can be calculated over a 24 hour period or a short term five minute period. As discussed further in this course, short-term 5-min recordings and 24 hour long-term recordings both seem to be appropriate options for the accurate analysis of the autonomic nervous system. Other commonly used statistical variables calculated from segments of the total monitoring period include SDANN, which is the standard deviation of the average NN interval calculated over short periods, usually 5 min, which is an estimate of the changes in heart rate due to cycles longer than 5 min, and the SDNN index, the mean of the 5-minute standard deviation of the NN interval calculated over 24 hours, which measures the variability due to cycles shorter than 5 min. However, the most commonly used measures derived from interval differences include rMSSD, the square root of the mean squared differences of successive NN intervals, NN50, the number of interval differences of successive NN intervals greater than 50 ms, and pNN50 the proportion derived by dividing NN50 by the total number of NN intervals. All of which you will see in most HRV analysis programs. Frequency Domain Measurement Another method of measuring heart rate variability is spectral analysis or power spectral density (PSD). Power spectral density shows how the total power, or variance, is distributed as a function of the various frequencies that affect the heart. In other words, PSD is the mathematical calculation of the overall strength of the influence of both the parasympathetic and sympathetic nervous systems on the heart. Frequency domaina analysis is traditionally performed by means of Fast Fourier Transformation (FFT). This method is simple to calculate and yields all of the following spectral frequencies in a graphical display. In other words, power spectral analysis yields four frequencies that occur during HRV assessment. These frequencies are defined as the ultra low frequency (ULF) 0.0001-0.003Hz, very low frequency (VLF) 0.003-0.04Hz, low frequency (LF) 0.04-0.15Hz, and high frequency (HF) 0.15-0.4Hz. The HF power spectrum reflects parasympathetic (vagal) tone and fluctuations caused by spontaneous respiration known as respiratory sinus arrhythmia. The LF power spectrum is evaluated in the range from 0.04 to 0.15 Hz. This band reflects sympathetic nervous system activity . The VLF power spectrum represents the thermoregulatory activity of the ANS on the body. Another frequency domain measurment is the total power (TP).The TP is a net effect of all possible physiological mechanisms contributing in heart rate variability that can be detected in 5-min recordings. In other words the total power represents the overall variability of the nervous system, and highly correlates to the SDNN measurement. The LF/HF Ratio is used to indicate balance between sympathetic and parasympathetic tone. A decrease in this score might indicate either increase in parasympathetic output, or a decrease in the sympathetic output. In other words the LF/HF ratio is the ratio between the LF and HF power. A higher number indicates increased sympathetic activity or reduced parasympathetic activity. This ratio can be used to help quantify the overall balance between the sympathetic and parasympathetic systems. References 5. Robertson R P. Halter J P, Porte D A. A role for alphaadrenergic nerves in abnormal insulin secretion in diabetes mellitus. J Clin Invest 57: 791-803, 1976. 6. Frohman L A. CNS peptides and glucoregulation. Ann Rev Physiol 45: 95-107. 1983. 7. Shimazu T. Central nervous system regulation of liver and adipose tissue metabolism. Diabetologia 20: 343- 356. 1981. 8. Bray G A. Nutrient balance: New insights into obesity. Int J Obesity ll(Suppl 3): 83-95, 1987. 9. Bergmanson J P G. Neural control of intraocular pressure. Am J Optom Physiol Opt 59: 94-98, 1982. 10. Korner P I. Central nervous control of autonomic cardiovascular function. pp 691-739 in Handbook of Physiology: The Cardiovascular System, sect 2, Vol I. (RM Berne, N Sperelakis eds) Am Physiol Sot, Bethesda, MD, 1979. 11. Sandin B, Chorot P. Changes in skin. salivary, and urinary pH as indicators of anxiety level in humans. Psychophysiology 22: 226-230. 1985. 12. Morse D R, Schacterle G R, Furst M L, et al. The effect of stress or meditation on salivary protein and bacteria: A review and pilot study. J Human Stress 8(4): 31-39. 1982. 13. Benetos A, Gavras I, Gavras H. Norepinephrine applied in the paraventricular hypothalamic nucleus stimulates vasonressin release. Brain Res 381: 322-326. 1986. 14. Lowknstein 0. Lowenfeld I. The pupil. pp 255-337 in The Eye, Vol. III. (Davson H. ed) Academic Press, New York, 1969. 15. Ashworth B. The pupil. Q J Med 243: 549-555. 1987. 16. Rogers M P, Dubey D. Reich P. The influence of the psyche and the brain on immunity and disease susceptibility: A critical review. Psychosom Med 41: 147-164. 1979. 17. Sanders V M, Munson A E. Norepinephrine and the antibody response. Pharm Rev 37: 229-248. 1985. 18. Livnat S, Madden K S, Felten D L, Felten S Y. Regulation of the immune system by sympathetic neural mechanism. Prog Neuropharm Biol Psychiat 11: 145-152, 1987. 19. Bulloch K, Pomerantz W. Autonomic nervous system innervation of thymic related lymphoid tissue in wildtype and nude mice. J Comp Neurol 228: 57-68. 1984. 20. Kissin I. McDanal J, Brown P J, Xavier A V. Bradley E L. Sympathetic blockade increases tactile sensitivity. Anaesth Analg 66: 1251-1255, 1987. 21. Asherman J G. Etiology of ectopic pregnancy: A new concept. Obstet Gvnecol 6: 619-624. 1955. 22. Bell G. Autonomic nervous control of reproduction: Circulatory and other factors, Pharm Rev 24: 657736. 1972. 23. Aguado I. Ojeda S R. Prepubertal ovarian function is finely regulated by direct adrenergic influences: Role of noradrenergic innervation. Endocrinology 114: 1845- 1853. 1984. 24. Bahr J. Kao L. Nalbandov A V. The role of catecholamines and nerves in ovulation. Biol Reprod 10: 273-290. 1974. 25. Ishii M, Igarashi M, Pate1 S, et al. Autonomic effects on R-R variations on the heart rate in the squirrel monkey: 26. An indicator of autonomic imbalance in conflict sickness. Am J Otolaryngol 8: 144-148. 1987. 27. Lichtor T, Davis H R. Johns L, et al. The sympathetic nervous system and atherosclerosis. J Neurosurg 67: 906- 914, 1987 28. Lathers C M, Schraeder P L. Weiner F L. Synchronization of cardiac autonomic neural discharge with epileptogenic activity: the lockstep phenomenon. EEG Clin Neuroohvsiol 67: 247-259. 1987. 29. Rothe C F: Reflex control of veins and vascular capacitance. Physiol Rev 63: 1281-1342, 1983. 30. Williams M E, Spokes K, Silva P. Role of alpha-adrenergic hormones in potassium homeostasis in the rat. J Lab Clin Med 110: 245-249. 1987. 31. Angel1 James J E. de Burgh Daly M. Nasal reflexes. Proc Roy Sot Med 162: 1287-1293. 1969. 32. Adlington J, Campeau S, Ciaramitaro K, Drennen T, Oliver A. Pre- and Post Treatment and Heart Rate Variability Readings of Subjects Receiving Logan Basic Technique Apex Contact Versus Table Position. Senior Research Publications Logan College of Chiropractic Library. Spring, 2001 edition. 33. Zhang J, Dean D: “Effect of chiropractic care on heart rate variability and pain in a multisite clinical Study.” J Manipulative Physiol Ther. 2006 ;29:267-274. 34. Budgell B, Polus B. The Effects of Thoracic Manipulation on Heart Rate Variability: A Controlled Crossover Trial. J Manipulative Physiol Ther. 2006 ;29 603-610.