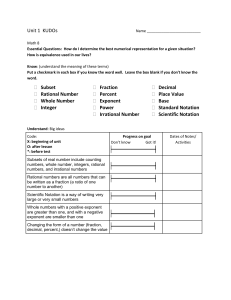
Scientific Notation is simply a method for expressing and working
advertisement

Scientific Notation is simply a method for expressing and working with very large or very small numbers. It is a short hand method for writing numbers. Numbers in scientific notation are made up for three parts: the coefficient, the base and the exponent. Observe the example on the right. This is the scientific notation for the standard number 21 600 000. Scientific Notation is simply a method for expressing and working with very large or very small numbers. It is a short hand method for writing numbers. Numbers in scientific notation are made up for three parts: the coefficient, the base and the exponent. Observe the example on the right. This is the scientific notation for the standard number 21 600 000. In order for a number to be in correct scientific notation, the following conditions must be true: 1. The coefficient must be greater than or equal to one and less than 10. In other words, there must be one and only one (nonzero) digit to the left of the decimal point. 2. The base must be 10. 3. The exponent must show the number of decimal places that the decimal needs to be moved to change the number to standard notation. In order for a number to be in correct scientific notation, the following conditions must be true: 4. The coefficient must be greater than or equal to one and less than 10. In other words, there must be one and only one (nonzero) digit to the left of the decimal point. 5. The base must be 10. 6. The exponent must show the number of decimal places that the decimal needs to be moved to change the number to standard notation. Changing numbers from Standard (decimal) Notation to Scientific Notation: Change 536 200 000 000 into scientific notation. Changing numbers from Standard (decimal) Notation to Scientific Notation: Change 536 200 000 000 into scientific notation. The decimal point must move to behind the 5 to make the coefficient have only one digit to the left of the decimal point. That will make the coefficient be 5.362. The decimal point moved 11 palces to the left, making the exponent 11. Therefore, 536 200 000 000 = 5.362 x 1011. The decimal point must move to behind the 5 to make the coefficient have only one digit to the left of the decimal point. That will make the coefficient be 5.362. The decimal point moved 11 palces to the left, making the exponent 11. Therefore, 536 200 000 000 = 5.362 x 1011. Here’s another way of looking at it. The original number is very large, and it’s being changed into a relatively small number (5.362). To make sure the number has the same value in decimal notation and scientific notation, you would have to multiply the smaller coefficient by a larger number, hence the positive power of ten. Here’s another way of looking at it. The original number is very large, and it’s being changed into a relatively small number (5.362). To make sure the number has the same value in decimal notation and scientific notation, you would have to multiply the smaller coefficient by a larger number, hence the positive power of ten. Change 0.000 000 032 8 into scientific notation. Change 0.000 000 032 8 into scientific notation. The decimal point must move behind the 3 to satisfy condition #1. This will make the coefficient 3.28. The decimal moves 8 places to the right, making the exponent -8. Therefore, 0.000 000 032 8 = 3.28 x 10-8. The decimal point must move behind the 3 to satisfy condition #1. This will make the coefficient 3.28. The decimal moves 8 places to the right, making the exponent -8. Therefore, 0.000 000 032 8 = 3.28 x 10-8. In this case, the original number is very small, and it’s being changed into a relatively larger number (3.28). To make sure the number has the same value in decimal notation and scientific notation, you would have to multiple the larger coefficient by a small number, hence the negative power of ten. In this case, the original number is very small, and it’s being changed into a relatively larger number (3.28). To make sure the number has the same value in decimal notation and scientific notation, you would have to multiple the larger coefficient by a small number, hence the negative power of ten. Scientific Notation Practice Name: _____________________________ Period: _________ Date: _______________ Complete the chart: Name: _____________________________ Period: _________ Date: _______________ Complete the chart: Standard Notation Speed of light in a vacuum Half-life of Uranium235 Melting point of Tungsten Temperature at which atomic fusion occurs Lowest Possible Temperature Radius of hydrogen energy level Scientific Notation Practice Scientific Notation 299293000 m/s 710000000 years 3410 C 1.5 x 107 C -2.73 x 102 C 5.3 x 10-11 m Perform the following operations, expressing the answer with the correct number of significant figures. 1. 1.35 m + 2.467 m ____________ Standard Notation Speed of light in a vacuum Half-life of Uranium235 Melting point of Tungsten Temperature at which atomic fusion occurs Lowest Possible Temperature Radius of hydrogen energy level Scientific Notation 299293000 m/s 710000000 years 3410 C 1.5 x 107 C -2.73 x 102 C 5.3 x 10-11 m Perform the following operations, expressing the answer with the correct number of significant figures. 1. 1.35 m + 2.467 m ____________ 2. 1 035 m2 ÷ 42 m ____________ 2. 1 035 m2 ÷ 42 m ____________ 3. 12.01 mL + 35.2 mL + 6 mL ____________ 3. 12.01 mL + 35.2 mL + 6 mL ____________ 4. 55.46 g – 28.9 g ____________ 4. 55.46 g – 28.9 g ____________ 5. 0.021 cm 3.2 cm 100.1 cm ____________ 5. 0.021 cm 3.2 cm 100.1 cm ____________ 6. 0.15 cm + 1.15 cm + 2.051 cm ____________ 6. 0.15 cm + 1.15 cm + 2.051 cm ____________ 7. 150 cm3 ÷ 4 cm ____________ 7. 150 cm3 ÷ 4 cm ____________ 8. 505 kg - 450.25 kg ____________ 8. 505 kg - 450.25 kg ____________ 9. 1.252 mm 0.115 mm 0.012 mm ___________ 9. 1.252 mm 0.115 mm 0.012 mm ___________ 10. 1.278 x 103 m2 ÷ 1.4267 x 102 m ____________ 10. 1.278 x 103 m2 ÷ 1.4267 x 102 m ____________ 11. 5.29 x 104 m 4.43 x 109 m ____________ 11. 5.29 x 104 m 4.43 x 109 m ____________ 12. 3.00 x 10-6 mm 5.21 x 1013 mm ____________ 12. 3.00 x 10-6 mm 5.21 x 1013 mm ____________