Applied Notes: Unit 5A—Counting Atoms
advertisement

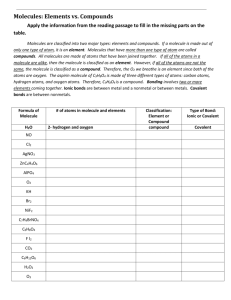
Name:___________________________________________ Period_________ Date_________________ Applied Notes: Unit 5A—Counting Atoms Read each note slide to find the answers to the questions. The questions go in the order of the slides. 1. What is a “subscript” and what does it tell you in chemistry? To help you remember what subscript means, remember that a submarine is something that goes below the surface of the water. Look at an example of a subscript and notice how we write the number below the surface of the line on which it is written. 2. What is a “coefficient” and what does it tell you in chemistry? 3. In each of the following examples, underline the coefficients and circle the subscripts: H2SO4 Ba(OH)2 4Cu(NO3)2 4K2SO4 2KNO4 3CaCl2 (NH4)PO4 2Ca3(PO4)2 2H2O 4. In H2O, there are ____# of Hydrogens and ______# of Oxygens. 5. When a chemical formula has parentheses, what do you do with the subscript inside the parentheses and the subscript outside the parentheses (what kind of math)? 6. In the formula Cu(NO3)2 there are _____# of Copper atoms, ______# of Nitrogen atoms and _____# of Oxygen atoms. 7. In question 6, how is it that you calculated more Oxygen atoms than Copper atoms? 8. In the formula 3CaCl2 there are _______# of molecules of CaCl2 and there are _______# of atoms ( remember there is a difference between molecules and atoms!) 9. Use the formula 4Cu(NO3)2 to identify the following: How many Cu in one molecule?____ How many N in one molecule?_____ How many O in one molecule?_____ How many total atoms in this molecule?_____ 10. Use the formula H2SO4 to identify the following: How many H in one molecule?____ How many S in one molecule?_____ How many O in one molecule?_____ How many total atoms in this molecule?_____ 11. Use the formula 4K2SO4 to identify the following: How many K in one molecule?____ How many S in one molecule?_____ How many O in one molecule?_____ How many total atoms in this molecule?_____ 12. Use the formula (NH4)PO4 to identify the following: How many N in one molecule?____ How many H in one molecule?_____ How many P in one molecule?_____ How many O in one molecule?_____ How many total atoms in this molecule?_____ 13. Use the formula Ba(OH)2 to identify the following: How many Ba in one molecule?____ How many O in one molecule?_____ How many H in one molecule?_____ How many total atoms in this molecule?_____ 14. Use the formula 2KNO4 to identify the following: How many K in one molecule?____ How many N in one molecule?_____ How many O in one molecule?_____ How many total atoms in this molecule?_____ 15. Use the formula 2Ca3(PO4)2 to identify the following: How many Ca in one molecule?____ How many P in one molecule?_____ How many O in one molecule?_____ How many total atoms in this molecule?_____