pmic12245-sup-0001-suppmat
advertisement

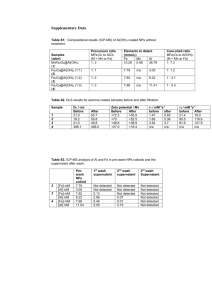
Supporting Information Designed Synthesis Mesoporous of Microspheres Fluorous-Functionalized for Specific Magnetic Enrichment of Phosphopeptides with Fluorous Derivatization Experimental section 1. Materials and reagents. Iron (Ⅲ) chloride hexahydrate (FeCl3·6H2O), ethylene glycol, ethanol, sodium acetate anhydrous (NaAc) and sodium hydroxide (NaOH) were purchased from Sinopharm Chemical Reagent Co., Ltd. (Shanghai, China). Hexadecyltrimethylammonium bromide was purchased from Sigma Chemical (St. Louis, MO, USA). Tetraethylsilicate (TEOS) was purchased from Shanghai Lingfeng Chemical Reagent Co., Ltd. (Shanghai, China). 1H,1H,2H,2H-perfluorodecyltriethoxysilane was purchased from Alfa Aesar (Tianjin, China). The NdFeB magnet was purchased from PCCW Ltd. (Beijing, China), 2 cm long, 2 cm wide, 1 cm high, with surface magnetic field strength of 1000 Gauss. Ammonium bicarbonate (NH4HCO3) was purchased from Chinasun Specialty Products Co., Ltd (Jiangsu, China). L-1-tosylamido-2-phenylethylchloromethyl ketone (TPCK) treated trypsin (from bovine pancreas), PNGase F (Genetimes Technology), bovine β-casein from bovine milk (95% PAGE), bovine serum albumin (BSA), chicken egg ovalbumin (OVA), 1H,1H,2H,2H-perfluorooctane-1-thiol, 2,5-dihydroxybenzoic acid (DHB) and trifluoroacetic acid (TFA) were purchased from Sigma Chemical (St. Louis, MO, USA). 1H,1H-perfluorooctylamine (PFOA) was purchased from J&K Chemical (Shanghai, China). The standard phosphorylated peptide (SADGQHAGGLVK, Mr = 1219.24 Da) was purchased from ChinaPeptides Co., Ltd. (Shanghai, China). Acetonitrile (ACN) was purchased from Merck (Darmstadt, Germany). Dimethyl sulfoxide (DMSO) and hydrogen peroxide (30%, w/w) were purchased from Shanghai Lingfeng Chemical Reagent Co., Ltd. (Shanghai, China). 1 Barium hydroxide (Ba(OH)2) was purchased from Sinopharm Chemical Reagent Co., Ltd. (Shanghai, China). The human serum sample originated from a hepatocellular carcinoma patient was acquired from Shanghai Zhongshan Hospital. Deionized water was purified by a Milli-Q system (Milford, MA, USA). All other chemicals and reagents are of the highest grade commercially available and used as received. 2. Synthesis of Fe3O4 microspheres The Fe3O4 microspheres were prepared via a hydrothermal reaction frequently used by our groups [1]. Briefly, 1.35 g of FeCl3·6H2O was dissolved in 75 mL of ethylene glycol under magnetic stirring. After complete dissolution, 3.60 g of NaAc was added. After the mixture was adequately stirred for another 1 h, the resulting solution was sealed in a Teflon-lined stainless-steel autoclave and heated at 200 ℃ for 16 h. The Fe3O4 microspheres obtained were washed with deionized water and ethanol several times in turn. 3. Characterizations and measurements Transmission electron microscope (TEM) images were taken on a JEM-2011 electron microscope (JEOL, Japan) operated at 200 kV. The sample was dispersed in ethanol in advance and collected for analysis by using carbon film-covered copper grids. Scanning electron microscope (SEM) images and the energy dispersive X-ray (EDX) spectrum were recorded on a Phenom Prox electron microscope (Phenom, Netherlands) operating at 15 kV. The sample was sprayed by a thin palladium film before the observation. Fourier transform infrared (FT-IR) spectra were collected on a Nexus 470 Fourier spectrophotometer (Nicolet, USA) using KBr pellets. Nitrogen adsorption-desorption isotherms were measured at 77 K with a Micromeritcs Tristar 3000 analyzer (Tristar, USA). The Brunauer-Emmett-Teller (BET) method was utilized to calculate the surface surface area and the Barrett-Joyner-Halenda (BJH) method was used to estimate the average pore size. The magnetic characterization was performed on a MPMS (SQUID) Vibrating Sample Magnetometer (Quantum Design, USA). 4. Sample preparation 2 Bovine β-casein and BSA proteins were dissolved in 25 mM NH4HCO3 buffer (pH 8.3) respectively and digested with trypsin (2.5%, w/w) at 37 ℃ for 16 h. The tryptic digests could be diluted to lower concentrations with 25 mM NH4HCO3 solution. To prepare the DHB matrix solution, we dissolved 20 mg of 2,5-dihydroxybenzoic acid in 1 mL of 50% ACN (v/v) aqueous solution containing H3PO4 (1%, v/v). Before investigating the enrichment efficiency of Fe3O4@mSiO2-C8F17 for fluorous labelled peptides, we suspended the magnetic microspheres in deionized water to prepare a dispersion (10 mg/mL). After vibration with the help of ultrasonication, a clear and homogeneous solution was obtained. Under a magnetic field, the Fe3O4@mSiO2-C8F17 microspheres were immediately attracted from the solution to the sidewall of the vial within 2 s, which saved much time for the enrichment procedures. 5. Enrichment of fluorous derivatized phosphopeptides To enrich fluorous derivatized phosphopeptides from the β-casein tryptic digest, 20 μL of Fe3O4@mSiO2-C8F17 suspension was added to 200 μL of the derivatized digest (2 μL of fluorous derivatized β-casein tryptic digest spiked into 198 μL of deionized water) in a 0.6 mL centrifuge tube. Then, the mixture was vibrated in a vortex at 37 ℃ for 30 min. The magnetic microspheres were isolated from the supernatant under a magnetic field. Next, the magnetic microspheres were rinsed with deionized water three times and eluted with 10 μL of acetonitrile at 37 ℃ for 15 min. The eluent was deposited on a MALDI sample target (Applied Biosystems/MDS SCIEX, Foster City, CA, USA) and dried at room temperature. The dilution without enrichment and the supernatant of equal volume were also deposited on the same sample target for comparison. Later on, 0.8 μL of DHB matrix was pipetted on the sample spots. Four replicate spots were taken for every sample. After the evaporation of the solvent, the samples were submitted to MALDI-TOF MS for identification. For the fluorous derivatized mixtures of β-casein tryptic digest and BSA tryptic digest, and for the fluorous derivatized human serum, similar dilution, enrichment, washing, elution and spotting steps were followed. The adducts of samples and DHB matrix were analyzed by MALDI-TOF MS. However, after the fluorous labelled mixtures of β-casein tryptic digest and BSA tryptic digest were enriched by Fe3O4@mSiO2-C8F17, the adsorbents were washed with 3 0.2 M ammonium to remove the interfering non-fluorous peptides. 6. Fluorous derivatization of glycans An OVA solution at the concentration of 1 μg/μL was treated with PNGase F to release the glycans, and the enzymatic deglycosylation reaction was conducted at 37 ℃ for 18 h. The resultant glycans were dissolved in 50% ACN/5% acetic acid (v/v) buffer and were allowed to react with 1H,1H-perfluorooctylamine (the molar ratio of derivatization reagent to glycans was 1:10) at 65 ℃ for 2h, with NaBH3CN as the reducing reagent. 4 Scheme S1. The synthetic route to Fe3O4@mSiO2-C8F17 microspheres. 5 Figure S1. (a) The SEM image of the spot chosen for the energy dispersive X-ray (EDX) analysis and (b) The EDX spectrum of the Fe3O4@mSiO2-C8F17 microspheres. 6 Figure S2. The magnetization curve of Fe3O4@mSiO2-C8F17. 7 8 Figure S3. MALDI-TOF mass spectrum for the dilution of fluorous derivatized β-casein tryptic digest after treatment with Fe3O4@mSiO2-C8F17 (a); and the corresponding MALDI-TOF/TOF tandem mass spectra: precursor ion m/z 2343.0 (b), precursor ion m/z 2838.0 (c) and precursor ion m/z 3404.0 (d). Fluorous phosphopeptides identified are marked with capital F. 9 Figure S4. MALDI-TOF mass spectra for the dilution of fluorous derivatized standard phosphopeptide. The original phosphopeptide is marked with capital P and the fluorous derivatized phosphopeptide is marked with capital F. 10 Figure S5. MALDI-TOF mass spectra for the N-glycans derived from fluorous derivatized ovalbumin digests (1 μg/μL): (a) before and (b) after enrichment with Fe3O4@mSiO2-C8F17. [M + H]+ signals are denoted as capital F and [M + Na]+ signals are denoted as capital Na. 11 Figure S6. MALDI-TOF mass spectra for the dilution of fluorous derivatized human serum: (a) before and (b) after enrichment with Fe3O4@mSiO2-C8F17. Fluorous derivatized phosphopeptides identified are marked with capital F. 12 Table S1. The EDX analysis results of Fe3O4@mSiO2-C8F17 Element Symbol Element Name Weight Concentration Error O Oxygen 38.7 0.0 Fe Iron 32.6 0.2 F Fluorine 23.9 0.4 Si Silicon 4.0 0.1 C Carbon 0.8 3.6 Table S2. Detailed information for the fluorous derivatized phosphopeptides identified from the fluorous derivatized β-casein tryptic digests after enrichment with Fe3O4@mSiO2-C8F17 Amino acid sequence of Observed Mr (expt) Mr (calc) derivatized phosphopeptides RELEELNVPGEIVECLSS 3403.3557 3404.5331 3404.3246 SEESITR+F13-Alk(C) FQCEEQQQTEDELQDKI 2836.9656 2838.3280 2838.1713 HPF+F13-Alk(C) FQCEEQQQTEDELQDK+ 2342.9160 2344.0408 2343.9301 F13-Alk(C) 2209.3047 2210.2879 2209.3993a DIGCESTEDQAMETIK+F 13-Alk(C) 2115.4172 2115.1776 2114.27396a YLGEYLIVPNCAEER+F1 3-Alk(C) 1942.0817 1942.8778 1942.8459a VPQLEIVPNCAEER+F13Alk(C) 1843.2764 1844.2309 1844.1891a EQLCTSEENSKK+F13-Al k(C) 1748.2822 1748.7685 1748.7320a TVDMECTEVFTK+F13-A lk(C) 13 FQCEEQQQTEDELQDKI 1562.2347 1561.1010 1561.0187 HPF+F13-Alk(C) a The phosphopeptides derived from α-casein. Table S3. Detailed information of derivatized N-glycans released from OVA glycoproteins enriched by Fe3O4@mSiO2-C8F17 (N-linked glycans were released by PNGase F digestion, and glycan structures were searched with Glycoworkbench through the molecular weight of oligosaccharide.) ■ (blue) represents N-acetylglucosamine, ● (green) represents mannose, ● (yellow) represents galactose, and PFOA represents 1H,1H-perfluorooctylamine. Observed Mr (expt) Mr (calc) 1315.49 1316.28 1316.47 1495.52 1496.39 1496.43 1618.55 1617.47 1617.55 1659.59 1658.39 1658.46 1721.65 1722.45 1722.57 1801.57 1802.48 1802.58 1842.67 1843.52 1843.69 Structure 14 1903.83 1902.59 1902.65 2065.76 2064.61 2064.71 2127.78 2128.58 2128.60 2248.84 2249.70 2249.83 2266.70 2267.67 2267.79 2307.82 2308.79 2308.89 2412.82 2411.78 2411.88 2471.88 2470.68 2470.89 2535.84 2534.74 2534.88 2654.89 2655.75 2655.85 15 Table S4. Detailed information for the fluorous phosphopeptides identified from the fluorous derivatized human serum samples after enrichment with Fe3O4@mSiO2-C8F17 m/z of fluorous derivatized m/z of corresponding phosphopeptides phosphopeptides 1671 1389 D[pS]GEGDFLAEGGGV 1742 1460 AD[pS]GEGDFLAEGGGV 1827 1545 D[pS]GEGDFLAEGGGVR 1898 1616 AD[pS]GEGDFLAEGGGVR 1489 1206 RG[pS]FSSENTWRK 1591 1309 K[pS]LPVSPVWGFKE Amino acid sequence Reference [1] Xu, X., Deng, C., Gao, M., Yu, W., Yang, P., Zhang, X., Synthesis of magnetic microspheres with immobilized metal ions for enrichment and direct determination of phosphopeptides by matrix-assisted laser desorption ionization mass spectrometry. Adv. Mater. 2006, 18, 3289-3293. 16