Name: ______________________________
Pd. _____
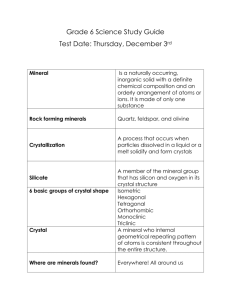
Unit 3A – The Lithosphere
CRYSTAL SYSTEMS ACTIVITY
47 pts.
MATERIALS
Crystal System Paper Models (A-F)
Scissors
Tape
Colored Pencils
Mineral Sample Box (#1-6)
ChemPlate
Marker
Stereo Microscope and Compound Microscope
Rubber Gloves
Safety Glasses
Sodium Chloride (NaCl)
Ammonium Di-Hydrogen Phosphate (NH4)H2PO4
Potassium Chromate (K2CrO4)
Sodium Nitrate (NaNO3)
Potassium Ferricyanide (K3Fe(CN)6)
Cupric Sulfate (CuSO4 · 5H20)
PART 1: PAPER CUTOUTS – 12 pts.
1. Carefully cut out all crystal system models (A-F). Each member of your group should assist in doing this.
2. Appropriately identify the crystal system associated with each cutout model listed below:
Model
Crystal System
A
B
C
D
E
F
PART 2: MINERAL EXAMPLES – 6 pts.
1. You have the following six minerals within your lab group, in no particular order:
Halite
Aragonite
Gypsum
Quartz
Plagioclase
Zircon
2.
Your goal is to identify the crystal system for each mineral using the following information:
Mineral Name
Crystal System
Halite
Isometric
Aragonite
Orthorhombic
Gypsum
Monoclinic
Quartz
Hexagonal (Trigonal)
Plagioclase
Triclinic
Zircon
Tetragonal
# 41
3.
Appropriately identify each mineral by completing the following table using your new knowledge of crystal systems:
Mineral #
Mineral Name
1
2
3
4
5
6
PART 3: MICROSCOPIC CRYSTAL SLIDES – 6 pts.
1. At the compound microscope set-up in the back of the lab, study the various slides containing the six different types of
crystal systems. Make sure each member of your group gets a chance to view the crystal slides. Draw what you see in
the appropriate spaces below:
ISOMETRIC
HEXAGONAL
MONOCLINIC
TETRAGONAL
ORTHORHOMBIC
TRICLINIC
PART 4: MICROCRYSTAL GROWTH – 18 pts.
1. Everyone must wear safety glasses during this part of the activity and the person handling the solutions must also wear
rubber gloves.
2.
Use the following information to label your ChemPlate with numbers 1 to 6 and the names of member of your group:
#1 – NaCl
#2 – (NH4)H2PO4
#3 – K2CrO4
#4 – NaNO3
#5 – K3Fe(CN)6
#6 – CuSO4 · 5H20
3.
Drop four drops of each chemical solution, located at the center lab table, into the correct spot plate well. Be certain to
match the number with the correct chemical as listed above.
4.
Allow the solutions to slowly evaporate overnight, placing them on the storage table.
5.
During the second day of the activity, retrieve your ChemPlate and study each crystalline spot under the Stereo
Microscope. Describe and draw the crystals in the appropriate space. Use the crystal systems worksheet to help in
determining the crystal system each salt belongs to. Try to find crystals that stand up on the ChemPlate so you can
check for angles between crystal faces that are not 90°. Think of crystal axes as unit cells or building blocks of crystals.
Spot #
Chemical Salt
1
NaCl
2
(NH4)H2PO4
3
K2CrO4
4
NaNO3
5
K3Fe(CN)6
6
CuSO4 · 5H20
Description
Drawing
Crystal System
ANALYSIS QUESTIONS – 5 pts.
1.
What are the five (5) characteristics defining a mineral?
______________________________________________________
______________________________________________________
______________________________________________________
______________________________________________________
______________________________________________________
2.
What is the basic building block of all silicate minerals? _______________________________________________________
3.
The hardest known mineral, according to Moh’s Hardness Scale is what? _________________________________________
4.
Describe rhombohedral cleavage.
_____________________________________________________________________________________________________
_____________________________________________________________________________________________________
5.
Why is color not the best property to use when identifying minerals?
_____________________________________________________________________________________________________
_____________________________________________________________________________________________________
 0
0


![ULEXITE [NaCaB5O6.8H2O]: An Extreme](http://s3.studylib.net/store/data/006902682_2-6ec8a0d1193ce61c1182d5c91126ae5a-300x300.png)