Reassessment of the Validity of the Universal Definition for
advertisement

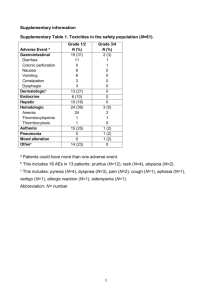
Electronic appendix Consideration of a New Definition of Clinically Relevant Myocardial Infarction after Coronary Revascularization An Expert Consensus Document from the Society for Cardiovascular Angiography and Interventions (SCAI) Issam D. Moussa, Lloyd W. Klein, Binita Shah, Roxana Mehran, Michael J. Mack, Emmanouil S. Brilakis, John P Reilly, Gilbert Zoghbi, Elizabeth Holper, Gregg W. Stone Table of Contents Page Practical Recommendations for Assessment of Cardiac Biomarkers after PCI 2 Post-PCI Cardiac Biomarker Determinations: Quality of Care Indicator 4 Impact of the “Clinically Relevant” Myocardial Infarction Definition on Clinical Trial Design Table 1. Contributing Factors to Cardiac Biomarker Elevations After Coronary 5 7 Revascularization Procedures Table 2. Prognostic Implications of Pre vs. Post-PCI Cardiac Biomarkers Elevations 8 Table 3A: Prognostic Implications of CK-MB Elevations After PCI 9 Table 3B: Prognostic Implications of Troponin Elevations After PCI 12 1 Practical Recommendations for Assessment of Cardiac Biomarkers after PCI The accurate assessment of post-PCI MI is dependent on knowledge of whether biomarker levels are elevated at baseline, both in patients with ACS and those undergoing elective PCI, in whom cTn may be falsely elevated in 10% or more of patients due to crossreacting heterophilic antibodies (36), interference with the immunoassay by rheumatoid factor (37), the presence of cTn T in diseased or regenerating skeletal muscle (38), cardiac conditions not related to CAD (1), analytic errors and other reasons. Thus, all patients should preferably have measurement of cardiac biomarkers prior to PCI. Ideally, cardiac biomarkers should be tested at least twice within 24 hours post-PCI. However, most interventionalists currently do not routinely measure cardiac biomarkers after an uncomplicated PCI (39), either because of cost considerations, planned early discharge, or lack of evidence that the knowledge of elevated biomarkers after an uncomplicated PCI affects prognosis or should change clinical management. In the most recent ACCF/AHA/SCAI PCI guidelines, routine assessment of cardiac biomarker levels after PCI is supported with a class I recommendation in the patient with angiographic complications during PCI or clinical symptoms or ECG changes after PCI, but with only a class IIb recommendation after an uncomplicated PCI (40). In the absence of routine testing we recommend the following: If cardiac biomarker levels were not elevated (or were not measured) pre-PCI, the PCI procedure is uncomplicated, and the patient is pain-free post procedure, a 12-lead electrocardiogram should be obtained within 1-4 hours post-PCI. If the ECG is normal cardiac biomarkers do not need to be drawn. If either clinical symptoms are present post-PCI, and/or transient or sustained angiographic complications occurred, and/or the post-PCI ECG shows new ST-segment depression or 2 elevation, then CK-MB (or cTn in hospitals where CK-MB is not available) should be measured 8-12 hours post the procedure. If post-PCI cardiac biomarker elevation >ULN is noted, then serial levels should be drawn q8-12 hours until they are falling. An ECG should also be repeated in 24 hours or earlier if symptoms develop. If a clinically relevant MI is diagnosed, an echocardiogram may be considered to assess left ventricular function, and other routine post-MI medical measures may be initiated, including beta-blockers and angiotensin converting enzyme inhibitors absent contraindications. Repeat cardiac catheterization might be considered if there were no overt angiographic complications to explain this level of myonecrosis and left ventricular function is depressed. Conversely, if lesser levels of CK-MB elevation occur and/or Q-waves have not developed, and the patient is ambulating without symptoms or signs of ischemia or heart failure, the patient may be discharged on the day after the procedure. For classification purposes this might be considered “myonecrosis without clinically relevant MI”. Of note, the recent proliferation of “high-sensitivity” cTn assays offers the capability to detect substantially lower levels of cTn. Compared to standard cTn assays, these tests offer improved sensitivity and diagnostic accuracy for detection of myonecrosis, at the cost of poorer specificity (41). However, while these tests may enhance the ability to identify patients presenting with true ACS from non-cardiac conditions, their prognostic utility in assessing postPCI myonecrosis has not been evaluated. In addition, low levels of troponin elevation may be detected in a high proportion of a healthy control population (42), and in patients with stable CAD (43). While such elevations may identify a cohort of patients at increased risk of death and MI (regardless of treatment) (44), their use cannot presently be recommended for assessment of post-PCI (or CABG) myonecrosis. 3 Post-PCI Cardiac Biomarker Determinations: Quality of Care Indicator? Because of a lowering of the threshold for its diagnosis (and switch from CK-MB to cTn as the “preferred” biomarker), the incidence of type 4a MI has been increasing over the past decade (39), despite an overall improvement in PCI outcomes in terms of survival and other measures (45-47). When evaluated by the National Cardiovascular Data Registry, institutions having excellent outcomes in the highest risk cases appear to also have the highest incidence of type 4a MI (39). The authors concluded that this observation could be attributed to the fact that the best institutions routinely measure cardiac biomarkers post-PCI and, hence, are more likely to find them elevated. Also, as previously discussed, post-procedural biomarker elevations may be a stronger marker of high-risk patients than of prognostically important procedural complications (8). In this regard, most large administrative databases do not adjust for the extent of coronary atherosclerosis. Thus, the use of post-PCI biomarker elevations to evaluate operators or programs is fraught with inconsistencies due to ascertainment bias, inconsistent measure of pre-PCI biomarker levels, and use of different biomarkers and assays, and is further confounded by difficulty in adjusting for clinical and angiographic risk factors for peri-procedural myonecrosis. Unless cardiac biomarkers are routinely determined at baseline and post procedure in all procedures and in all programs and appropriately adjusted for patient and lesion risk, its use as a quality measure should be discouraged. 4 Impact of the “Clinically Relevant” Myocardial Infarction Definition on Clinical Trial Design An important role for a standardized definition of MI is to allow for accurate comparisons of the frequency of MI and the impact of pharmacologic and other invasive treatments across different populations and trials. The Universal Definition criteria are relatively sensitive, and will increase the number of patients who are classified as having MI compared to more specific criteria. Caution is warranted with an overly sensitive MI diagnosis in clinical trials, as any gain in power from greater event accrual may be attenuated by misclassification errors and noise (66). In this regard cTn elevations are observed not infrequently in otherwise stable patients, and from non-coronary related conditions such as pericarditis and myocarditis (67,68). Including such patients in clinical trials risks over-diagnosing type 4a MI if the baseline value wasn’t assessed. Moreover, including non-clinically relevant MI as part of the primary composite endpoint in device and pharmacologic clinical trials may lead to improper assessment of treatment effects. In some cases one therapy may be deemed superior to another on the basis of clinically nonrelevant biomarker elevations. In other cases use of overly sensitive biomarkers resulting in high rates of peri-procedural MI may add noise, thereby obscuring the detection of clinically important differences between therapies. For example, in the COURAGE trial, the strategy of routine PCI plus optimal medical therapy (OMT) was non-inferior for the occurrence of death or MI compared to OMT plus provisional PCI in patients with stable CAD (69). However, the point estimates favored the PCI group for the endpoints of spontaneous MI (which has been strongly related to late mortality in numerous studies) as well as death. Conversely, the point estimate favored type 4a MI for the OMT arm, driven by small biomarker elevations post PCI. Had those “MI” endpoints, most of 5 which have not been shown to be of prognostic importance, been excluded, the interpretation of the COURAGE trial may have been quite different. In support of this concept, a recent metaanalysis of 12 randomized trials of PCI vs. medical therapy in stable CAD with 37,548 patientyears of follow-up has demonstrated a significant 24% reduction in spontaneous MI, which parallelled a trend toward a 30% reduction in cardiovascular mortality with PCI, despite a significant increase in peri-procedural MI with PCI (70). We therefore recommend that future clinical trials utilize the clinically relevant definition of post revascularization MI introduced in this document, rather than a more sensitive and less prognostically relevant definition. Importantly, the level of biomarker elevations post-CABG vs. post-PCI that reflect equivalent levels of myonecrosis have not been determined. As such, biomarkers should not be used to compare the rates of MI between PCI and CABG procedures (except perhaps for the occurrence of new Q-wave MI). In addition, patients and caregivers in the post-CABG and postPCI settings have different thresholds (or patient ability, if intubated) for reporting chest pain, different rates of routine ECG assessment, different thresholds to perform repeat catheterization, etc. The lack of uniformity in classification, assessment and understanding of the prognostic relevance of post-CABG and post-PCI MIs (as currently defined) represents a major limitation of past and recent studies that have attempted to compare these revascularization modalities. 6 Table 1. Contributing Factors to Cardiac Biomarker Elevations After Coronary Revascularization Procedures Percutaneous coronary intervention Distal embolization (inherent or device- Coronary artery bypass graft surgery Systemic inflammatory response due to related) cardiopulmonary bypass - Platelet-fibrin aggregates Global ischemia due to aortic cross-clamping - Atheromatous debris Direct trauma - Calcified plaque Atheroembolism - Thrombus (pre-existing or iatrogenic) Coronary dissection Side-branch occlusion (small or large) Reperfusion injury Transient abrupt closure or balloon- Perioperative coronary vasospasm induced ischemia Stenosis or occlusion of bypass graft Coronary dissection - Loss of small branches - Compromised distal flow Coronary vasoconstriction or vasospasm Air embolism Endothelial dysfunction Plaque characteristics Soft core with thin cap Marked eccentricity Ulcerated lesion surface Large plaque volume 7 Table 2. Prognostic Implications of Pre vs. Post-PCI Cardiac Biomarkers Elevations Study Gustavsson71 Population 327 patients Biomarkers cTn I >1x ULN CK-MB >1x ULN Study findings Patients with normal baseline cTn: cTn, compared to CK-MB, more often resulted in the diagnosis of type 4a myocardial infarction Patients with elevated baseline cTn: The degree in increase in baseline cTn correlated with post-PCI CK-MB levels rather than post-PCI cTn levels. (The association between post-PCI cardiac biomarker elevation and the risk of subsequent events was not assessed) Miller72 2352 patients cTn T and CK-MB Patients with normal baseline cTn: Post-PCI cTn elevation did not lead to increased longterm mortality (median follow-up 11.9 months). Patients with elevated baseline cTn: Further elevation in post-PCI cTn level was not associated with a increase in long-term events (Only baseline cTn has prognostic significance; death, myocardial infarction: HR 1.14 (95% CI 1.07-1.22) 8 Table 3A: Prognostic Implications of CK-MB Elevations After PCI Study Patient population Biomarkers Incidence Follow-up Long-term mortality Kini73 Year Published 1999 1,675 patients CK-MB normal CK-MB 3-5x ULN CK-MB >5x ULN 81.3% 3.5% 2.4% 13 ± 3 months 1.3% 1.7% 7.5% (p<0.05 vs. other groups) Hong74 1999 1,693 patients: CK-MB normal CK-MB 1-5x ULN CK-MB >5x ULN* 52.7% 32.1% 15.2% 1 year 4.8% 6.5% 11.7% (OR 3.3) Tardiff75 1999 2,341 patients CK-MB normal CK-MB 3-5x ULN CK-MB 5-10x ULN CK-MB >10x ULN 76.0% 3.6% 3.7% 2.9% 6 month Post-PCI CK-MB associated with 6-month MACE (r2=32.56, p<0.0001) Stone24 2001 7,147 patients: 69% with ACS CK-MB normal CK-MB 3-5x ULN CK-MB 5-8x ULN CK-MB >8x ULN* Q-wave MI* 62.1% 6.1% 4.1% 7.7% 0.6% 1.4 ± 1.2 years 7.0% 4.3% 6.3% 14.5% (OR 2.2) 38.6% (OR 9.9) Brener25 2002 3,478 patients 64% with ACS CK-MB normal CK-MB 3-5x ULN CK-MB 5-10x ULN CK-MB >10x ULN* 76% 4.5% 2.9% 2.4% 15±15 months Death or MI: 9.4% 13.5% 16.7% 30.5% (OR 1.89) (OR 1.78 if > 3 x ULN) Dangas76 2002 4,085 patients: 67% with ACS CK-MB >5x ULN* 12.8% 1 year OR 1.5 9 Ellis77 2002 8,409 patients 63% with ACS CK-MB normal CK-MB 1-5x ULN CK-MB >5x ULN 82.8% 13.6% 3.6% 4 years 15.1% 16.9% 19.4% Ioannidis78 (meta-analysis) 2003 23,230 patients CK-MB 1-5x ULN CK-MB >5x ULN 19% 6% 6 to 34 months Risk ratio: 1.5 Ajani79 2004 1,326 patients 85.4% with ACS CK-MB normal CK-MB >3x ULN* 55% 20% 1 year 3.4% 9.3% (OR 1.57 for death or MI) Jeremias14 (meta-analysis) 2004 5,850 patients CK-MB normal CK-MB 3-8x ULN CK-MB >8x ULN or Q-wave MI 78.6% 6.1% 2.6% 1 year 1.9% 2.6% 4.7% Jang80 2008 1,807 patients 40.9% with ACS CK-MB normal CK-MB 1-5x ULN CK-MB >5x ULN 79.1% 14.6% 6.4% 13 ± 7 months MACE: 5.0% 6.1% 10.4% Andron81 2008 3,864 patients 30.4% with ACS CK-MB normal CK-MB 3-5x ULN CK-MB >5x ULN* 70.6% 4.4% 5.1% 22 months 2.4% 7.2% 7.6% (HR 2.26) Lindsey26 2011 6,347 patients 100% elective PCI CK-MB <5 ng/mL CK-MB 5-15 ng/mL CK-MB 15-25 ng/mL CK-MB 25-50 ng/mL CK-MB >50 ng/mL 74.5% 17.6% 3.9% 2.5% 1.5% 1 year 2% 3% 3% 4% 12% (adjusted HR 4.87) 10 3.1 Bonaca19 2012 13,608 patients 100% with ACS CK-MB >3x URL with 2 samples or >5x URL with 1 sample 4.4% 180 days Adjusted HR 2.4 for cardiovascular death Damman28 (meta-analysis) 2012 5,467 patients 100% with ACS FRISC II: CK-MB >1.5x ULN ICTUS: CK-MB >1x ULN RITA 3: CK-MB >2x ULN 3.9% 5 years No association between procedural-related MI and 5-year cardiovascular mortality * Predictor of outcomes by multivariable analysis. 11 Table 3B: Prognostic Implications of Troponin Elevation After PCI Study Garbarz82 Year Published Patient population 1999 109 patients 46% with ACS Biomarkers cTn I >1x ULN Incidence 27% Follow-up Outcomes 8±3 Not predictive of MACE months Bertinchant83 1999 cTn I >1x URL 18% 19 ± 10 months Not predictive of MACE Fuchs84 2000 cTn I >3x ULN* 15.4% 8 months Predictor of major in-hospital complications (OR 2.1, 95% CI 1.2-3.9) 105 patients (baseline cTn normal) 1126 patients: 70.9% with ACS Not predictive of long-term death Cantor85 Herrmann86 Nallamothu87 2002 481 patients: 100% with ACS 2002 2002 278 patients: 3,5739.7% patients with ACS 2003 cTn I >1x ULN 48% 3 months Not predictive of MACE cTn T >1x ULN* CK-MB normal CK-MB 3-5x ULN CK-MB 5-10x ULN CK-MB >10x ULN* 17.3% 62% 6% 6% 5% 7.8 ± 5.3 months 3 years Predictor of MACE (HR 7.0% 3.27, 95% CI 1.14-9.41) 14.7% 14.5% 17.7% (HR 1.1) 1157 patients: 36.5% with ACS cTn I 1-3x ULN cTn I 3-5x ULN cTn I 5-8x ULN cTn I ≥ 8x ULN* Q-wave MI* 16.0% 4.6% 2.0% 6.5% 0.3% 11 ± 7 months Predictors of long-term mortality: cTn >8xULN: HR 2.4, 95% CI 1.2-5.0 Q-wave MI: HR 8.9, 95% CI 1.3-62.6 Kizer88 2003 212 patients: 84% with ACS cTn T >1x ULN* 11% 5.6 years Predictor of MACE at 1 year (HR 2.39, 95% CI 1.09-5.26) Natarajan89 2004 1,128 patients cTn I ≥5x ULN* 9.1% 1 year Predictor of in-hospital (not 1-year) MACE (OR 3.8, 95% CI 1.1-12.6) 12 Kini90 2004 2,873 patients (normal baseline cTn) cTn I 3-5x ULN cTn I >5x ULN CK-MB 3-5x ULN CK-MB >5xULN* 8.4% 14.1 2.3% 1.6% 12 ± 6 months CK-MB >5x ULN (not cTn) predictor of long-term mortality (HR 6.7, 95% CI 1.9-22.9) Cavallini91 2004 3,494 patients : 50.8% with ACS cTn I >1x ULN CK-MB >1x ULN* 44.2% 16% 2 years Nageh92 2005 316 patients: cTn I >1x ULN* 31% 18 months CK-MB (not cTn) predictor of long-term mortality (OR per unit 1.04, 95% CI 1.01-1.07) Predictor of MACE on follow-up (OR 18.9, 95% CI 9.7-37) 2,352 patients (31% with elevated baseline cTn) 1,949 patients: 47.9% with ACS cTn T >1x ULN 62.8% 12 months Pre-PCI, not post-PCI, cTn predictor of death or MI cTn T >1x ULN* 19.6% 3 years Double post-PCI TnT associated with a partial HR 1.20 (95% CI 1.02-1.40) (22% with angiographic complications) Miller72 2006 Prasad93 2006 Prasad17 2008 5,487 patients (37% with elevated baseline cTn) cTn T >1x ULN --- 28 months Pre-PCI , but not post-PCI, troponin predictor of long-term mortality Nienhuis94 (meta-analysis) 2008 cTn >1x ULN* 32.9% 16.3 months Predictor of composite mortality or MI OR 1.59 (95% CI 1.29–1.95) Hubacek95 2009 15,581 patients (baseline cTn normal) 1,208 patients 64% with ACS cTn T >3x ULN 19.7% 1 year Not predictive of MACE De Labriolle96 2009 3,200 patients 100% elective PCI cTn I >3x ULN 23.4% 1 year Not predictive of MACE Feldman97 2009 1,601 patients 43.3% with ACS cTn I ≥5x ULN* 19.0% 25 ± 8 months Predictor of long-term mortality (HR 1.81, 95% CI 1.02-3.21) 13 Cavallini98 2010 2,362 patients: 45.1% with ACS Feldman99 (meta-analysis) 2011 Novack22 2012 cTn I >3x URL 19.8% 2 years Not predictive of MACE 22,353 patients cTn >1x ULN* (baseline Tn normal) --- 17.7 months Predictor of long-term all-cause mortality OR 1.45 (95% CI 1.22-1.72) 4,930 patients 100% elective PCI 7.2% 24.3% 7.0% 1 year Mortality: 5.8% (Adjusted HR 2.5) 4.5% (Adjusted HR 1.7) 6.8% (Adjusted HR 2.6) CK-MB >3x ULN cTn >3x ULN cTn >20x ULN *p < 0.05 for multivariable analysis 14