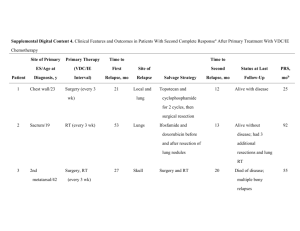
Therapeutic Discussion – Granulomatosis with

Therapeutic Discussion – Granulomatosis with Polyangitiis
Basic Pathophysiology
Small vessel vasculitis – necrotizing inflammation of the small vessels: arterioles, capillaries, and venules
90-95% positive for proteinase 3 (PR3) antineutrophil cytoplasmic Abs
TNF alpha increases adhesion of endothelial cells and primes neutrophils leading to migration of PR3
PR3-ANCA interacts with PR3 & Fcy-receptor on surface of neutrophil = neutrophil activation
Leads to change in neutrophil rheology, arrest in diapedesis (passage of blood cells through walls of capillaries, typically accompanying inflammation) & adhesion to endothelium
PR3-ANCA induces release of reactive O2 species & proteolytic enzymes resulting in damage of endothelium
Activated T-Helper 1 cells migrate to inflammatory sites, secrete TNF alpha & IFN gamma, and induce macrophage maturation, tissue destruction and granuloma formation
CC. HPI,
Subjective findings
What are the symptoms of Wegener’s?
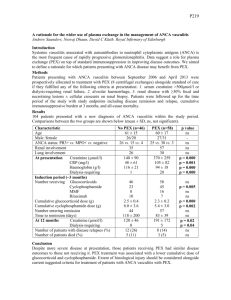
Flu-like symptoms – fever, polymyalgia, headache, malaise, anorexia, unintended weight loss
HEENT – hearing loss, bloody rhinorrhea, sinusitis**, nasal crusting, recurrent otitis media, nasal/oral ulcers, paresis of ocular motor nerves, ocular pain, diplopia, visual loss
RESP – severe/persistent rhinorrhea, recurrent epistaxis, cough, fever, wheezing
CNS – peripheral neuropathy
Meds PTA
Are there any medications that may cause Wegener’s?
Objective
?propylthiouracil, ?hydralazine
How do we diagnose Wegener’s?
> 2 of the following: o Abnormal urinary sediment (red cast cells or > 5 RBC/HPF) o Abnormal findings on CXR o Oral ulcers/nasal discharge o Granulomatous inflammation on biopsy
Renal biopsy – little/no deposition of immune complexes (pauci-immune) in vessel wall & segmental necrotizing & crescentic
ANCA may rule out ANCA-associated vasculitis but positive ANCA not associated with definitive diagnosis
Vitals
Physical Exam
Vitals: ↑ BP; ↑ RR
HEENT: purulent/bloody discharge from nose; nasal ulcers; saddle-nose deformity; oral ulcers; epistaxis
Resp: Pulmonary hemorrhage (10% of pts with ANCA GN); wheezing
GU: hematuria
DERM: black fingertips
Investigations Lab Proteinuria > 1-3 g/day with presence of dysmorphic red cells in urine and red cell casts
Dx
Declining GFR over days/weeks
+ve ANCA to neutrophil granule proteins myeloperoxidase (MPO) or proteinase 3 (PR3)
90% of pts
↑ ESR, ↑ CRP
CXR: bilateral infiltrates that don’t respond to abx; non-TB cavitating lesions
Echo: lesions attributed to GPA – regional wall abnormalities; LV systolic dysfxn w/ ↓ EF; pericardial effusion
U/S: granulomatosis mass in kidney
Goals of therapy
Achieve remission as soon as possible and prevent relapse
Prevent mortality and organ damage
Prevent adverse events
Treatment alternatives and duration
Induction:
Immunosuppressive therapy is indicated in all cases of ANCA vasculitis & GN
Rare exception: severe kidney-limited disease, in absence of extrarenal manifestations of small-vessel vasculitis o Those requiring dialysis, ?risks of therapy >? Likelihood of recovering kidney function
All patients with extrarenal manifestations should receive immunosuppressive therapy, regardless of degree of kidney dysfunction
Cyclophosphamide addition to CCS in induction therapy improved remission rates from 55% 85% & ↓ relapse rate three-fold
Pulse IV & daily PO = similar remission & relapse rates
IV pulse group receive ~1/2 the cumulative amount vs. PO daily
Pulse IV vs. daily PO associated with less leukopenia, infections (NSS), increased risk of relapse & trend toward increased number of pts requiring renal replacement therapy
Duration of continuous PO cyclophosphamide should be limited to 3 months with a maximum of 6 months o Pulse IV duration is inferred, but not tested o If still dialysis dependent after 3 months & no ongoing extrarenal manifestations of active vasculitis –
D/C cyclophosphamide therapy
Methylprednisolone pulse therapy for induction not directly tested
Pulse b/c rapid anti-inflammatory effect & a rapid reduction in ANCA-producing plasma cells
MEPEX Trial: 3 x 1g IV vs. plasmapheresis as adjunctive to PO CCS & PO cyclophosphamide o Pulse methylprednisone < efficacious than plasmapheresis in preserving kidney fxn
No data that 1000 mg > 500 mg lower dose used in practice
Rituximab
RITUXVAS Trial: randomized 3:1 to either rituximab 375 mg/m 2 weekly x 4 + cyclophosphamide 15 mg/kg IV 2 weeks apart x 2 doses OR cyclophosphamide 15 mg/kg IV Q 2 weeks x 3, then Q 3 weeks for max of 10 doses o Remission rates similar 76% R vs. 82% C
RAVE Trial: randomized to rituximab 375 mg/m 2 infusions weekly x 4 weeks OR cyclophosphamide 2 mg/kg/day
PO for months 1-3, then azathioprine 2 mg/kg/day PO for months 4-6 o NSS in rates of complete remission at 6 months, adverse events, or relapse rates o Excluded patients with severe alveolar hemorrhage or SrCr > 354 umol/L
Long term safety still unknown & more expensive than cyclophosphamide
Plasmapheresis is the removal, treatment and return of blood plasma from blood circulation
Indicated for SrCr > 500 umol/L or those with diffuse alveolar hemorrhage
7 treatments associated with significantly higher rate of kidney recovery at 3 months vs. 3 doses of IV methylprednisolone
In SrCr < 500 umol/L & as adjunctive therapy no benefit seen, but studies underpowered
Steroid taper: reduce to 10-20 mg/day by 12 weeks with continuing tapering until discontinuation
Remission = absence of manifestations of vasculitis and GN disease activity (absence of microscopic hematuria and a stable/improved proteinuria & GFR)
Maintenance:
Maintenance therapy > 18 months in patients who remain in complete remission (low evidence)
Required in those at high risk of relapse or who have received less than 6 months induction treatment with cyclophosphamide o High risk = persistence of PR3-ANCA (vs. MPO-ANCA), hx of URT/LRT disease
1 of 3 risk factors = 1.7-fold increased risk of relapse
All 3 risk factors = 4.7-fold increased risk of relapse
No maintenance therapy in dialysis-dependent patients with no extrarenal manifestations
Relapse rates are 60% lower in patients with ESRD & infections are twice as frequent
Azathioprine 1-2 mg/kg/day PO x 6-18 months
Preferred agent
As effective as cyclophosphamide but with a more favorable adverse-effect profile
Vs. MMF = superior
Mycophenolate mofetil 1 g PO BID – if allergic/intolerant to AZA
Septra – adjunct to maintenance therapy in those with URT disease
Decreased rate of upper-airway relapse
No impact on rate of relapse in other organs
Methotrexate 0.3 mg/kg/week to max of 25 mg/week – if intolerant to AZA & MMF & GFR > 60 ml/min/1.73m
2
12 month maintenance therapy compared to AZA 2 mg/kg/day after induction of remission with cyclophosphamide & CCS; NOT designed to test superiority, but safety o Rates of relapse were not significantly different between groups o Methotrexate wasn’t associated with higher rate of adverse events, but severity of events was greater
Not recommended in GFR < 30; dose adjustments in GFR < 60
Relapse
Relapse = occurrence of increased disease activity after a period of partial/complete remission
Associated with increased risk of ESRD and severe/life-threatening extrarenal damage
4.7 x more likely to progress to ESRD with relapse
Respond to immunosuppression with CCS & cyclophosphamide with similar response rates as initial disease
Take into account the cumulative dose of cyclophosphamide previously received o Risk of malignancy increases with cumulative doses of cyclophosphamide > 36 g
Severe relapses = cyclophosphamide + CCS + plasmapheresis when indicated
Rituximab more effective than cyclophosphamide in treating patients with relapsing ANCA vasculitis
Resistance
Resistance = persistence of/appearance of kidney and/or systemic manifestations of vasculitis, while receiving tx equal in intensity to initial immunosuppressive therapy
IV immunoglobulin 2 g/kg
More rapid decline in disease activity & CRP at 1 & 3 months, but NSS after 3 months wrt disease activity/frequency of relapse
Rituximab 375 mg/m 2 IV weekly x 4 or 500 mg IV weekly x 4 + CCS – remission in majority of patients
Monitoring Parameters: Efficacy and toxicity
Toxicity:
Corticosteroids
Vitals: ↑ BP
CNS: insomnia, shakiness, mood changes (euphoria, depression, personality changes, frank psychoses)
HEENT: moon face, cataracts, oral candidiasis
CVS: edema from Na retention, CHF
GI: bloated abdomen, ↑ appetite, N/V
ENDO: weight gain, menstrual irregularities, hyperglycemia
MSK/SKIN: muscle wasting (especially thigh), delayed wound healing, easy bruising, osteoporosis, tendon rupture
↑ infections
Cyclophosphamide
HEENT: alopecia
RESP: pulmonary fibrosis (long & high doses)
GI: anorexia, N/V, diarrhea,
GU: bladder toxicity and cancer; hemorrhagic cystitis (MESNA – antidote cystitis)
o Hydration is key!
HEME: leukopenia (nadir 8-15 days, then recover 17-28 days after 1 dose), thrombocytopenia (nadir 10-15 days), anemia
ENDO: fertility issues
↑ infections, ↓ wound healing
Rituximab
Azathioprine