Accuracy and Precision Purpose: To gain an understanding of the
advertisement

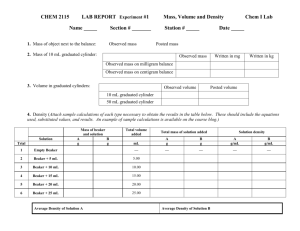
Accuracy and Precision Purpose: To gain an understanding of the relationships that exist between density, mass, and volume while calibrating laboratory glassware. In this experiment, water will be measured and delivered using a buret, a graduated cylinder, a beaker and a flask. Based on the results, the most accurate measuring device will be determined. Accuracy is a measure of how close a data point is to an accepted (true) value. Percent error is a measure of accuracy and is calculated using the following equation: % 𝐸𝑟𝑟𝑜𝑟 = |𝐴𝑐𝑐𝑒𝑝𝑡𝑒𝑑 𝑉𝑎𝑙𝑢𝑒 − 𝐸𝑥𝑝𝑒𝑟𝑖𝑚𝑒𝑛𝑡𝑎𝑙 𝑉𝑎𝑙𝑢𝑒| 𝑥 100% 𝐴𝑐𝑐𝑒𝑝𝑡𝑒𝑑 𝑉𝑎𝑙𝑢𝑒 The percent error is often expressed as a positive number, regardless of whether the measured value is larger or smaller than the accepted value. Precision reflects the uncertainty of reading an individual measurement. Procedure Part A: Determining the Accuracy of a 50 mL beaker A1. Weigh a clean, dry 50 mL beaker that has graduation marks on it. Record its mass on your data sheets. A2. Add tap water to the 20 mL mark, using a dropper pipette to get as close to this mark as possible. A3. Weigh the filled beaker. Record this value on your data sheets. A4. Empty and dry the beaker, and then repeat this measurement two more times. A5. Using the density of water at the lab temperature, calculate the volume of water delivered from the graduated cylinder. Part B: Determining the Accuracy of a Graduated Cylinder B1. Obtain a dry 100 mL graduated cylinder. B2. Add tap water to the 20 mL mark, using a dropper pipette to get as close to this mark as possible. B3. Dry your 50 mL beaker from part A and weigh it. Transfer the water from the graduated cylinder to the beaker and weigh the beaker. B4. Repeat steps B2 and B3 two more times. B5. Using the density of water at the lab temperature, calculate the volume of water delivered from the graduated cylinder. Part C: Determining the Accuracy of a Buret C1. Obtain a buret, and fill with tap water using a beaker and funnel. Remember that measurements must always be made at the bottom of the meniscus for aqueous solutions in glass. C2. Obtain a 50 mL beaker. Measure and record its mass. C3. Read and record the initial water level in the buret. Deliver approximately 20 mL of water to the pre-weighed beaker from the buret and read and record the water level in the buret again. C4. Measure the mass of the beaker and water. C5. Repeat steps C3 and C4 two more times. C6. Measure the temperature of your water, and use this measurement and the density table below to determine the density of the water. C7. Use the mass of water delivered and the known density of water to calculate the actual volume of water delivered. Reference Data Name: Lab Partner: These are EXAMPLES, not a KEY. Notice highlighted regions Data and Calculations Part A: Accuracy of a Beaker Box Trial 1 Trial 2 1 Mass of Empty Beaker (g) 38.12g 38.19g Trial 3 38.15g 2 Mass of Beaker with 20 mL Water (g) 57.33g 57.17g 59.85g 3 Mass of Water (g) Box 2-Box 1 19.21g 18.98g 20.70g 4 Volume of Water according to Beaker (mL)- “Experimental Value” 20mL 20mL 20mL 5 Temperature of Water (ºC) 25o 25o 25o 6 Density of Water According to Table (g/mL) .9970479 g/ml .9970479 g/ml .9970479 g/ml 7 Volume of Water based on Density“Accepted Value” 𝒎𝒂𝒔𝒔 𝒗𝒐𝒍𝒖𝒎𝒆 = 𝒅𝒆𝒏𝒔𝒊𝒕𝒚 𝑩𝒐𝒙 𝟑 𝒗𝒐𝒍𝒖𝒎𝒆 = 𝑩𝒐𝒙 𝟔 19.267mL 19.036mL 20.761mL 8 Percent Error Try these calculations out, we’ll check in class. % 𝐸𝑟𝑟𝑜𝑟 |𝐴𝑐𝑐𝑒𝑝𝑡𝑒𝑑 𝑉𝑎𝑙𝑢𝑒 − 𝐸𝑥𝑝𝑒𝑟𝑖𝑚𝑒𝑛𝑡𝑎𝑙 𝑉𝑎𝑙𝑢𝑒| 𝑥 100% = 𝐴𝑐𝑐𝑒𝑝𝑡𝑒𝑑 𝑉𝑎𝑙𝑢𝑒 |𝐵𝑜𝑥 7 − 𝐵𝑜𝑥 4| 𝑥 100% % 𝐸𝑟𝑟𝑜𝑟 = 𝐵𝑜𝑥 7 Part B: Accuracy of a Graduated Cylinder Box 1 Mass of Empty Graduated Cylinder (g) Trial 1 128.12g Trial 2 128.52g Trial 3 127.97g 2 Mass of Graduated Cylinder with 20 mL Water (g) 147.98g 147.95g 147.81g 3 Mass of Water (g) Box 2- Box1 Volume of Water- Experimental Value (according to graduated cylinder) 19.86g 19.43g 19.84g 20mL 20mL 20mL 5 Temperature of Water (ºC) 25o 25o 25o 6 Density of Water According to Table (g/mL) .9970479 g/ml .9970479 g/ml .9970479 g/ml 7 Volume of Water based on density“Accepted Value” 𝒎𝒂𝒔𝒔 𝒗𝒐𝒍𝒖𝒎𝒆 = 𝒅𝒆𝒏𝒔𝒊𝒕𝒚 𝑩𝒐𝒙 𝟑 𝒗𝒐𝒍𝒖𝒎𝒆 = 𝑩𝒐𝒙 𝟔 19.919mL 19.488mL 19.899mL Percent Error Try these calculations out, we’ll check in class. 4 % 𝐸𝑟𝑟𝑜𝑟 |𝐴𝑐𝑐𝑒𝑝𝑡𝑒𝑑 𝑉𝑎𝑙𝑢𝑒 − 𝐸𝑥𝑝𝑒𝑟𝑖𝑚𝑒𝑛𝑡𝑎𝑙 𝑉𝑎𝑙𝑢𝑒| 𝑥 100% = 𝐴𝑐𝑐𝑒𝑝𝑡𝑒𝑑 𝑉𝑎𝑙𝑢𝑒 |𝐵𝑜𝑥 7 − 𝐵𝑜𝑥 4| 𝑥 100% % 𝐸𝑟𝑟𝑜𝑟 = 𝐵𝑜𝑥 7 Part C: Accuracy of a Buret Box 1 Initial mass of empty beaker (g) Trial 1 38.12g Trial 2 38.19g Trial 3 38.15g 2 Initial water level in buret (mL) 2.25mL 22.25mL 5.70mL 3 Final water level in buret (mL) 22.25mL 44.25mL 25.70mL 4 Volume of water delivered (mL)“Experimental Value” Box 3- Box 2 Final mass of water and beaker (g) 20mL 20mL 20mL 58.04g 58.18g 58.12g 6 Mass of water delivered (g) Box 5- Box 1 19.92g 19.99 19.97 7 Temperature of water (ºC) 25o 25o 25o 8 Density of water based on table (g/mL) .9970479 g/ml 19.978mL .9970479 g/ml 20.049mL .9970479 g/ml 20.029mL 5 9 Volume of Water based on density“Accepted Value” 𝒎𝒂𝒔𝒔 𝒗𝒐𝒍𝒖𝒎𝒆 = 𝒅𝒆𝒏𝒔𝒊𝒕𝒚 𝑩𝒐𝒙 𝟔 𝒗𝒐𝒍𝒖𝒎𝒆 = 𝑩𝒐𝒙 𝟖 10 Percent Error % 𝐸𝑟𝑟𝑜𝑟 |𝐴𝑐𝑐𝑒𝑝𝑡𝑒𝑑 𝑉𝑎𝑙𝑢𝑒 − 𝐸𝑥𝑝𝑒𝑟𝑖𝑚𝑒𝑛𝑡𝑎𝑙 𝑉𝑎𝑙𝑢𝑒| 𝑥 100% = 𝐴𝑐𝑐𝑒𝑝𝑡𝑒𝑑 𝑉𝑎𝑙𝑢𝑒 % 𝐸𝑟𝑟𝑜𝑟 = |𝐵𝑜𝑥 9 − 𝐵𝑜𝑥 4| 𝑥 100% 𝐵𝑜𝑥 9 Try these calculatio ns out, we’ll check in class. Analysis Questions 1. Based on your results, which piece of glassware is most accurate in delivering liquids? Which piece of glassware is least accurate? 2. Consider the definition of precision. Do you feel any of the glassware used in lab today were precise? What criteria are you using to make your decision? 3. Determine the most appropriate piece of equipment to use in the following situations: a. A lab procedure calls for 50 mL of an acid b. You are trying to cool down a piece of hot metal and need about 100 mL of room temperature water c. A lab procedure calls for 25 mL of a calcium chloride solution d. You need to know exactly how much acid, place, it took to neutralize 25.0 mL of a sample of base. You will be using the number to make a series of other important calculations. e. A lab procedure calls for a hot water bath created with 40-50 mL of water 4. A student performs a lab to experimentally determine the boiling point of rubbing alcohol. She determines it is 79.4ºC. After doing research, she finds the “accepted” value is 82.5ºC. Calculate the percent error for this situation.