Nucleic Acids
advertisement

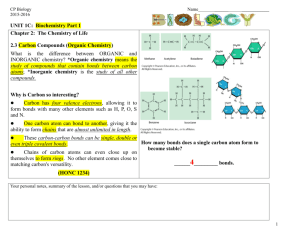
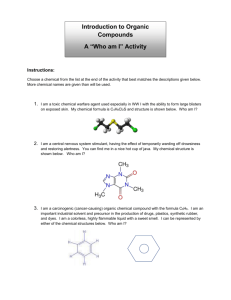
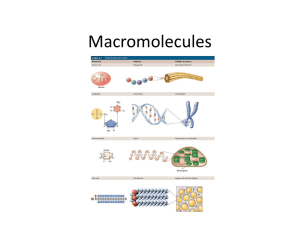
CP Biology 2015-2016 Name KEY______ ____________ UNIT 2B: Biochemistry Part 1 Chapter 2: The Chemistry of Life 2.3 Carbon Compounds (Organic Chemistry) What is the difference between ORGANIC and INORGANIC chemistry? *Organic chemistry means the study of compounds that contain bonds between carbon atoms; *Inorganic chemistry is the study of all other compounds. Why is Carbon so interesting? Carbon has four valence electrons, allowing it to form bonds with many other elements such as H, P, O, S and N. One carbon atom can bond to another, giving it the ability to form chains that are almost unlimited in length. These carbon-carbon bonds can be single, double or even triple covalent bonds. Chains of carbon atoms can even close up on themselves to form rings. No other element comes close to matching carbon's versatility. (HONC 1234) How many bonds does a single carbon atom form to become stable? ______4_________ bonds. Your personal notes, summary of the lesson, and/or questions that you may have: 1 *ORGANIC COMPOUNDS: Are usually defined as compounds which contain carbon with hydrogen. May contain additional elements such as O, N, P, K, S, Fe, Ca, Na, Cl and others.) Are produced only by living things (biotic). Range from simple to very complex. Contain strong, covalent bonds. examples: _CH4, C6H12O6, SUGARS, PROTEINS, FATS, OILS, DNA Identify whether each of the following compounds is organic or inorganic by placing a √ in the appropriate column. Substance Organic? Inorganic? 1. sodium chloride (table salt): NaCl 2. glucose: C6H12O6 X X 3. water: H2O *INORGANIC COMPOUNDS: Usually defined as compounds that do not contain carbon with hydrogen. (exceptions may contain just carbon.) They often can be formed in the non-living (abiotic) environment, but : Can also be made by/found in living things. examples: ____H2O, NaCl, O2, NH3, CaCO3, CO2__ X 4. heating oil: C14H30 X 5. chitin (a protein): C8H12NO5 X 6. thymine (a nitrogenous base): C5H5N2O2 X 7. sulfuric acid: H2SO4 X 8. oxygen gas: O2 X 9. ethanol: C2H5OH X 10. adenosine triphosphate (ATP): C10H16N5O13P3 X 11. carbon dioxide: CO2 X Your personal notes, summary of the lesson, and/or questions that you may have: 2 1 The General Structure of Macromolecules Many of the organic compounds produced by living things are so large that they are called *macromolecules which means giant molecules. Examples: Carbohydrates, lipids, proteins and nucleic acids. Most macromolecules are fomed through a process called *polymerization in which large compounds are built by joining smaller ones together. The smaller repeating units called *monomers may be identical or different from one another. The large compounds which are formed from the joining of many monomers are called *polymers. 2 Monomers combine to form one dimer Two monomers joined together are called a *dimer. The exception: Lipids are not composed of monomers and polymers. Instead, they take different forms which we will discuss later in this unit. The dimer, plus 3 more monomers combine to form a polymer Your personal notes, summary of the lesson, and/or questions that you may have 3 1 Four Types of Biologically Important Macromolecules What are the four major types of organic macromolecules and what are the building blocks of each type? Carbohydrates (monosaccahrides) Lipids (glycerol and 3 fatty acids) Nucleic Acids (nucleotides) Proteins (amino acids) (In this unit, we will be focusing primarily on the STRUCTURE or FORM of these molecules. We will discuss their FUNCTION at appropriate points throughout the course.) Most of the foods we eat are made from living things; therefore they are composed of a variety of different organic molecules. A well-balanced diet provides not only the energy needed to fuel life processes but also the materials needed for cells to build essential molecules and cell structures. GROUP Basic Building Blocks (Monomers) Polymer Carbohydrates monosaccharide polysaccharide Proteins Amino acid (or peptide) polypeptide Nucleic Acids nucleotide Nucleic acid Lipids Glycerol and 3 fatty acids Triglyceride Carbohydrates Foods in Breads, cereal, which pasta, fruit they are found Lipids Nucleic Acids Proteins Oils, butter, meat, dairy products, plant seeds All types of Meat, dairy foods derived products, from living beans things Organic molecules must be digested and converted by metabolic processes into molecules organisms can use. All organisms use chemical processes to BUILD UP and BREAK DOWN organic polymers. Remember, there is a continual cycling of matter throughout all ecosystems, while energy is not recycled Your personal notes, summary of the lesson, and/or questions that you may have: 4 Synthesis and Breakdown of Macromolecules Two major chemical processes (metabolic reactions) occur to build up or break down all four types of macromolecules into larger or smaller units. *Dehydration Synthesis is the combination of smaller organic subunits by the removal of water. This chemical reaction involves removing an –H from one subunit and an –OH from the other subunit to allow the subunits to bond together. *Hydrolysis is the breakdown of macromolecules into their subunits by adding water. This chemical reaction involves breaking a bond and adding an –H and an – OH to adjacent subunits so the molecules can exist separately. NOTE: Water is required as a reactant A ___polymer (large molecule)____ and _water (H2O)_______ are products of the reaction Dehydration Synthesis. Water is a (reactant / product) of the reaction, Hydrolysis. Smaller molecules are the (reactants / products) of the reaction, Hydrolysis. These reactions occur in both directions, depending upon the needs of the organism. Your personal notes, summary of the lesson, and/or questions that you may have: 5 1 Examples (Dehydration Synthesis): 1) Carbohydrate Organic Compound 2) Lipid Dehydration Synthesis Reactant(s) Product(s) carbohydrate Monosaccharide Polysaccharide +water Lipid glycerol + 3 fatty acids Triglyceride + water protein Amino acid *Polypeptide + water nucleic acid Nucleotide DNA or RNA + water (triglyceride) 3) Protein Your personal notes, summary of the lesson, and/or questions that you may have: 6 1 Examples (Hydrolysis): 1) Carbohydrate Organic Compound Hydrolysis Reactant(s) Product(s) carbohydrate Polysaccharide + water monosaccahrides Lipid Triglyceride + water glycerol + 3 fatty acids protein *polypeptide + water Amino acids + water nucleic acid DNA or RNA + water nucleotides 2) Lipid (triglyceride) 3) Protein Your personal notes, summary of the lesson, and/or questions that you may have: 7 1 CARBOHYDRATES Monosaccharides are important for energy and cell structure. Sometimes called ‘hydrated’ carbons. They contain atoms of *CARBON, HYDROGEN and OXYGEN, usually in a ratio of *1: 2: 1. Carbohydrates differ in structural makeup. They range from small, monosaccharides (*simple sugars) to intermediate molecules such as disaccharides, to large polysaccharides (complex carbohydrates). *GLUCOSE Chemical Formula C6H12O6 *FRUCTOSE Chemical Formula C6H12O6 *GALACTOSE Chemical Formula C6H12O6 *RIBOSE Chemical Formula C6H12O6 *Monosaccharides: single sugar molecules, the simplest (monomer) unit of carbohydrates. *Disaccharides: a compound made by joining two monosaccharides together Examples *Sucrose - (table sugar) - made by joining Glucose and Fructose *Lactose - (milk sugar) - made by joining Galactose and Glucose Disaccharides Chemical Formula: C12H10O11 Your personal notes, summary of the lesson, and/or questions that you may have: ISOMERS- Same (iso) parts (mer) Glucose , Fructose and Galactose are isomers 8 1 *Polysaccharides: large molecules formed from monosaccharides. Aka: complex carbohydrates CELLULOSE GLYCOGEN STARCH *What differences and/or similarities do you notice about the polysaccharides above? They are all composed of ring-like structures of simple sugars but the bonds that connect them are drawn differently. Some are branched and some are formed in a diagonal manner. (Remember form leads to function, so these differences in form mean they function differently) Your personal notes, summary of the lesson, and/or questions that you may have: 9 1 Important Polysaccharides Synthesized by Animals and Plants: Animals consume carbohydrates in food, digest them (hydrolysis) and then store the excess sugar by synthesizing glycogen. *Glycogen is a polysaccharide that many animals make to store excess sugar, sometimes referred to as "animal starch". When the level of glucose in your blood runs low, glycogen stored in the liver and muscles is broken down into glucose which is released into the blood so it can be delivered to cells. Plants produce glucose during photosynthesis and use it to synthesize starch. *Starch is a polysaccharide that plants make to store excess sugar. In addition, *Cellulose is another polyssacharide made by plants. It forms tough flexible fibers that give plants much of their strength and rigidity. Cellulose is a major component of the cell walls of plant cells. Wood and paper are made largely of cellulose! Humans generally can't digest cellulose ("fiber"), but it helps regulate the elimination of your solid wastes. Examine the picture to the right. What do starch, glycogen and cellulose have in common? 1) all made of glucose monomers 2) all are polysaccharides 3) all contain only C, H and O Your personal notes, summary of the lesson, and/or questions that you may have: 10 1