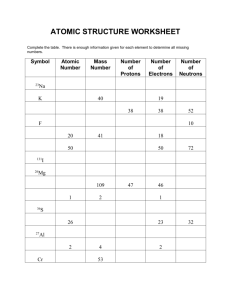
Molecular/Formula Mass The molecular mass is the atomic mass of
advertisement

Molecular/Formula Mass The molecular mass is the atomic mass of all atoms within a molecule. The formula mass is specific to ionic compounds. We will be using this molecular mass when talking about the mole in later sections. To find the molecular mass, (or formula mass in the case of ionic compounds..) we simply take the amount of atoms within the compound for each element, and multiply that amount by the atomic mass of that element. Example: HC2H3O2 (acetic acid) To calculate the molecular mass, we break the molecule into the individual elements to see how many atoms of each element we have. H-4 C-2 O-2 We then multiply that amount by the atomic mass of that element. You do not need to use the entire atomic mass from the periodic table, the general rule is going two past the decimal. H-4(1.01amu)= 4.04 amu C-2(12.01 amu) = 24.02 amu O-2 (16.00 amu)= 32.00 amu Now that we have the atomic mass of each individual part, we can add all parts together to get the atomic mass of the molecule. 4.04amu + 24.02 amu + 32.00 amu = 60.06 amu This value gives us the molecular mass for acetic acid. When we talk about this in terms of the mole, we will call this value the molar mass. The molar mass of a molecule is simply its total atomic mass, but instead of using the atomic mass unit, we use the unit of grams to equate this value to a mole. Use this example and the material covered in class Tuesday January 13th and Wednesday January 14th to complete this blizzard bag assignment.