Eval Efficacy Safety Tolerability of Tenofovir
advertisement

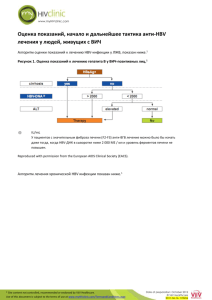
Phoenix Children’s Hospital Research Institute (PCRI) Website Clinical Trial Listing ** Please complete and return to Shy Walker at swalker@phoenixchildrens.com Study Title: Evaluating the Efficacy, Safety and Tolerability of Tenofovir DF in Pediatric Patients With Chronic Hepatitis B Infection Study Purpose: This placebo-controlled study evaluates the efficacy, safety and tolerability of tenofovir disoproxil fumarate (TDF) in participants 2 to < 12 years old with chronic Hepatitis B infection. Study Summary: While studies have shown significant virologic response in adults and adolescents, the effect in children is not well established. This study will provide valuable data that can help establish the efficacy and safety profiles of TDF in children. The study will consist of 72 weeks of blinded randomized treatment, after which participants will switch to open-label TDF treatment for an additional 120 weeks. Basic Eligibility Criteria: Inclusion Criteria: Male or Female, 2 to < 12 years of age Weight ≥ 10kg Chronic HBV infection ≥ 6 months HBeAg-positive or HBeAg-negative HBV Viral Load ≥ 100,000 copies/mL Alanine aminotransferase (ALT) ≥ 1.5 x the upper limit of the normal range (ULN) at screening Creatinine Clearance ≥ 80 mL/min Absolute neutrophil count (ANC) ≥ 1,500/mm^3, hemoglobin ≥ 10g/dL Negative pregnancy test at screening No prior tenofovir DF therapy (subjects may have received prior interferon-alfa and/or other oral anti-HBV nucleoside/nucleotide therapy; subjects must have discontinued interferon-alfa therapy ≥ 6 months prior to screening; subjects experienced on other anti-HBV nucleoside/nucleotide therapy must have discontinued therapy ≥ 16 weeks prior to screening to avoid flare if randomized to the placebo arm) Exclusion Criteria: Pregnant or lactating Decompensated liver disease Received interferon therapy within 6 months of Screening Received anti-HBV nucleoside/nucleotide therapy within 16 weeks of Screening Alpha-fetoprotein levels > 50 ng/mL Evidence of hepatocellular carcinoma (HCC) Co-infection with HIV, acute hepatitis A virus (HAV), hepatitis C virus (HCV), or hepatitis D virus (HDV) Chronic liver disease not due to HBV History of significant renal, cardiovascular, pulmonary, neurological or bone disease Long term non-steroidal, anti-inflammatory drug therapy Study Location(s): Phoenix Children’s Hospital Study Contact(s): Michael Johnson, Research Coordinator mjohnson7@phoenixchildrens.com