key terms
advertisement

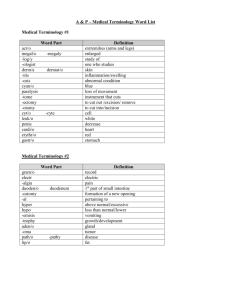
UNIT- Chemical Reactions As students look critically at their local environment they should become aware of the millions of substances that surround them. Some, like water and sand, occur naturally; some, like sugar and blood, are produced by living things; while others, like plastic and paper, are produced in factories. Regardless of their origin, the one thing that all of these substances have in common is that they all contain the basic building block of matter – the atom. The Chemical Reactions unit aims to: Extend students’ understanding of the Particle Model to the level of basic Atomic Theory Introduce the Periodic Table and its role in organising the elements Familiarise students with chemical symbols and formulae – the language of Chemistry Conduct a series of practical activity that demonstrate common chemical reactions Begin to write chemical equations Investigate the role and impact of industrial chemistry in the students’ every day life Key Competencies KEY SKILLS The Key Skills that students will improve during the completion of this unit are: Defining and using designated chemical terminology appropriately Using the theory learnt in the unit to solve problems Recording and interpreting experimental results Designing and evaluating a practical investigation Conducting an extended practical investigation Time management and meeting deadlines KEY KNOWLEDGE The Key Knowledge that students will gain from this unit include: basic structure of an atom differences between elements, compounds and mixtures symbols/formulae of designated elements/compounds characteristics that distinguish a physical change from a chemical change characteristics of several different types of chemical reactions guidelines for writing simple chemical equations using either words or symbols factors that influence the rate of a chemical reaction role of chemical reactions in common industrial processes KEY TERMS The Key Terms that students will become familiar with the definition and spelling of during this unit are: KEY TERMINOLOGY I Atom Symbol Neutron Periodic Table Nucleus Mass Number Proton Electron shell Element KEY TERMINOLOGY II Mixture Atomic Number Electron Compound Chemical formula Molecule KEY TERMINOLOGY III Physical change Chemical change Reactant Aqueous solution Rate of reaction Word equation States of matter Chemical reaction Product Insoluble Combination reaction Decomposition reaction Combustion reaction Precipitation reaction Formula equation Diagrammatic equation Unit Resources “Chemical Reactions” Resource Folder e-textbooks and Teacher Resources (should be loaded onto Laptop from external hard-drive) Hardcopy textbooks including- Science Alive 2, Science Links 2, Science Dimensions 2 Science Alive 1 and 2 Teacher Resource POSSIBLE ASSESSMENT TASKS INCLUDE Practical Report- “Is it an acid?” Practical Report- “The Hydrogen ‘pop’ test” Student designed practical investigation- “Properties of Elements” Topic test