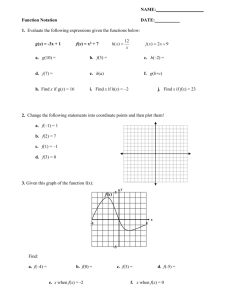
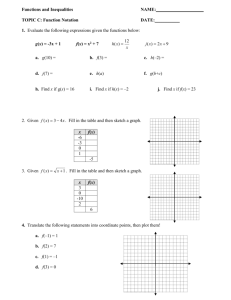
PCR - OIE
advertisement

OIE Reference Laboratory Reports Activities in 2011 Name of disease (or topic) for which you are a designated OIE Reference Laboratory: Address of laboratory: Swine influenza National Veterinary Services Laboratories USDA, APHIS, VS P.O. Box 844, 1920 Dayton Avenue Ames, Iowa 50010 UNITED STATES OF AMERICA Tel.: +1-515-337-7551 Fax: +1-515-337-7348 e-mail address: website: Name (including Title and Position) of Head of Laboratory (Responsible Official): Name(including Title and Position) of OIE Reference Expert: sabrina.l.swenson@aphis.usda.gov http://www.aphis.usda.gov/animal_health/lab_info_services/ Dr Elizabeth A. Lautner Director, National Veterinary Services Laboratories Dr Sabrina Swenson Head, Bovine, Porcine, and Aquaculture Viruses Section Name (including Title and Position) of writer of this report (if different from above): Annual reports of OIE Reference Centres, 2011 1 Swine Influenza Part I: Summary of general activities related to the disease 1. 2. 2 Test(s) in use/or available for the specified disease/topic at your laboratory Test For Specificity Total HI Antibody H1 54 HI Antibody H3 217 PCR Nucleic acid Matrix 59 PCR Nucleic acid Pandemic N1 59 PCR Nucleic acid H subtype 367 PCR Nucleic acid N subtype 367 Sequencing Genes 216 MDCK Virus isolation 42 Egg inoculation Virus isolation 0 Production and distribution of diagnostic reagents Type of reagent Amount supplied nationally (including for own use) Amount supplied to other countries H1 HI positive control serum 5 – 2 ml/vial 11 – 2 ml/vial H3 HI positive control serum 2 – 2 ml/vial H1 antiserum 9 – 2 ml/vial 2 – 2 ml/vial H3 antiserum 8 – 2 ml/vial 7 – 2 ml/vial H1 HI antigen 44 – 2 ml/vial H3 HI antigen 15 – 2 ml/vial H1 conjugate 5 – 1 ml/vial N1 RNA control for PCR 18 – 1.5 ml/vial 1 – 1 ml/vial Annual reports of OIE Reference Centres, 2011 Swine Influenza Part II: Activities specifically related to the mandate of OIE Reference Laboratories 3. International harmonisation and standardisation of methods for diagnostic testing or the production and testing of vaccines a) Establishment and maintenance of a network with other OIE Reference Laboratories designated for the same pathogen or disease and organisation of regular inter-laboratory proficiency testing to ensure comparability of results None. b) Organisation of inter-laboratory proficiency testing with laboratories other than OIE Reference Laboratories for the same pathogens and diseases to ensure equivalence of results None. 4. Preparation and supply of international reference standards for diagnostic tests or vaccines Reagents are available for serology, PCR, and FA. National repository of SIV isolates is being developed. 5. 6. Research and development of new procedures for diagnosis and control Evaluated subtyping PCRs for H1/H3 and N1/N2. Assays are currently undergoing licensing procedures in the United States. Evaluating the use of the ion torrent and illumina for rapid, whole genome sequencing. Collection, analysis and dissemination of epizootiological data relevant to international disease control The United States began a voluntary swine influenza surveillance project in May 2009 to generate information on swine influenza circulating in domestic swine in the United States. Diagnostic specimens (swabs, tissues, oral fluids) entering National Animal Health Laboratory Network (NAHLN) labs can voluntarily be placed into the surveillance stream and tested for the presence of SIV. Testing and epidemiologic data are accumulated, viruses are referred to the NVSL for development of a National repository and for appropriate confirmatory testing. Sequences from 3 genes (H, M, N) up to 8 genes are deposited into GenBank. Testing is standardized in the labs. NVSL participates with public health and other agricultural groups in epidemiologic investigations and testing (where applicable) when influenza virus is identified in an unexpected host. 7. Maintenance of a system of quality assurance, biosafety and biosecurity relevant to the pathogen and the disease concerned The National Veterinary Services Laboratories’ Quality Management System is accredited to ISO/IEC 17025 by an independent accreditation body recognized by the International Laboratory Accreditation Cooperation. Audits and surveillance visits are an ongoing part of maintaining ISO/IEC 17025 accreditation. Biosafety and biosecurity at the NVSL comply with the U. S. Department of Agriculture and U. S. Department of Health and Human Services Select Agent Program and as such meet or exceed guidelines established in “Biosafety in Microbiological and Biomedical Laboratories”. Annual reports of OIE Reference Centres, 2011 3 Swine Influenza 8. Provision of consultant expertise to OIE or to OIE Member Countries NVSL is a member of OFFLU and a swine specific subgroup of OFFLU. NVSL has participated in OFFLU meetings and teleconferences and has reviewed documents and provided opinion when requested. 9. Provision of scientific and technical training to personnel from other OIE Member Countries NVSL has collaborated with Central American countries (through FAO) to train personnel, provide reagents and protocols, and to provide confirmatory testing. Training was completed in 2010; however, additional reagents were provided in 2011, and NVSL is available for consultation, as needed. NVSL is in early discussions with a Caribbean country to provide protocols, reagents, and confirmatory testing. 10. Provision of diagnostic testing facilities to other OIE Member Countries See above. 11. Organisation of international scientific meetings on behalf of OIE or other international bodies None 12. Participation in international scientific collaborative studies None 13. Publication and dissemination of information relevant to the work of OIE (including list of scientific publications, internet publishing activities, presentations at international conferences) 4 Presentations at international conferences and meetings Swenson S, Korslund J, Pyburn D, Vincent A. 2011. USDA SIV Surveillance. U.S. An Health Assoc Public Health and Rabies Com. Buffalo, NY. Swenson S. 2011. Swine Influenza Surveillance in the United States. OFFLU. Paris, France. Lorusso A, Vincent AL, Harland M, Alt D, Bayles DO, Swenson SL, Gramer MR, Russel CA, Smith DJ, Lager KM, Lewis NS. 2011. Genetic and Antigenic Charactierization of H1 Influenza Viruses from U.S. Swine Prior to the Emergence of the 2009 pandemic H1N1. 45 th US-Japan cooperative Program in Natural Resources Panel of Animal and Animal Health Mtg. Ames, IA. Irwin CK, Zimmerman J, Anderson J, Jenkins-Moore M, Leslie-Steen P, Patnayak D, Vincent A, Zhang J. June 2011. Detection of influenza A virus in oral fluid ring test samples by virus isolation. Proc 6th International Symposium on Emerging and Re-emerging Pig Diseases. Barcelona, Spain, p. 283. Irwin CK, Zimmerman J, Christopher-Hennings J, Harmon K, Kitikoon P, McGill J, Otterson T, Rauh R, Shah R, Skarbek K. June 2011. Detection of influenza A virus in oral fluid ring test samples by RT-PCR. Proc 6th International Symposium on Emerging and Re-emerging Pig Diseases. Barcelona, Spain, p. 284. Irwin C, Zimmerman J, Wang C, Hesse R, Harmon K, Christopher-Hennings J, Otterson T, McGill J, Vincent A, Shah R, Rauh R. October 2011. Real-Time rt-PCR detection of influenza A Virus in oral fluid using a check test. Proc. 54th Annual Conference American Association of Veterinary Laboratory Diagnosticians. Buffalo, NY, p. 153. Annual reports of OIE Reference Centres, 2011 Swine Influenza Scientific publications in peer-reviewed journals Dawood F, Dong L, Liu F, Blau D, Peebles P, Lu X, Wagers L, Oakland B, Zielenski M, Daly R, Horan V, Swenson S, Schmitt B, Hancock K, Katz J, Bridges C, Kightlinger L, Finelli L. A pre-pandemic outbreak of triple-reassortant swine influenza virus infection among university students, South Dakota, 2008.Journal of Infectious Disease 204 (8), (2011), 1165-1171. Chen LM, Rivailler P, Hossain J, Carney P, Balish A, Perry I, Davis CT, Garten R, Shu B, Xu X, Klimov A, Paulson JC, Cox NJ, Swenson S, Stevens J, Vincent A, Gramer M, Donis RO. (2011). Receptor specificity of subtype H1 influenza A viruses isolated from swine and humans in the United States. Virology. Apr 10; 412(2):401-10. Campagnolo E, Rankin J, Daverio S, Hunt E, Lute J, Tewari D, Acland H, Ostrowski S, Moll M, Urdaneta V, Ostroff S. (2011). Fatal Pandemic (H1N1) 2009 Influenza A Virus Infection in a Pennsylvania Domestic Cat. Zoonoses and Public Health. Nov; 58(7):500-7. (acknowledged for the virus isolation work NVSL did) Other communications Swenson S. April 2011. SIV Typing Assay Evaluations at the NVSL. NAHLN Technical Methods Working Group. Ames, IA. Swenson S. September 2011. SIV Typing Assay Evaluations at the NVSL. NAHLN Technical Methods Working Group. Ames, IA. _______________ Annual reports of OIE Reference Centres, 2011 5