CHE 2201, Section 101 Fall 2013 Exam IIIB Name: Honor Pledge: I

CHE 2201, Section 101 Fall 2013
Exam IIIB
Name: _____________________________________________________________________
Honor Pledge: I pledge on my honor that I have not violated the Appalachian State University
Academic Integrity Code. I will not discuss this exam with any students who have not yet taken it.
Signature: _________________________________________
Unsigned honor pledges will result in a grade of 0 points.
Page 1: _____/ 22 points
Page 2: _____/ 18 points
Page 3: _____/ 36 points
Page 4: _____/ 15 points
Page 5: _____/ 9 points
Total: _____/100 points
0
1.
A compound has molecular formula of C
7
H
13
ClO. a. (4 points) Calculate the degree of unsaturation for this molecule. b. (4 points) Draw a molecule with this molecular formula that is also a tertiaray alkyl chloride.
2. (4 points) Draw the structure of (Z)-3,6-dimethyl-2-octene.
3. (4 points) Rank the following carbocations by their stability (1 = most stable, 4 = least stable)
4. (6 points) Give an example (either name or structure) of a polar protic solvent and a polar aprotic solvent in the appropriate box.
/ 22 points
1
5. (4 points) Circle the structure in which the group in bold is a bad leaving group.
6. (4 points) Circle the definition of an organometallic coupling reaction. a.) It brings two organometallic reagents together to form a carbon-carbon bond. b.) It forms a carbon-carbon bond between an organometallic reagent and an organohalide. c.) It creates an organometallic reagent from an organohalide. d.) It forms two organometallic reagents from two organic compounds.
7. (6 points) Indicate whether the carbon in bold is oxidized, reduced, or unchanged in the reaction.
8. (4 points) You have discovered a new chemical species and you are not sure if it is a strong or weak nucleophile. You set up two substitution reactions, one in which the concentration of your new chemical species is 1.0 M and one in which the concentration is 2.0 M. You find that the two reactions have the same rate . Is your nucleophile strong or weak? Explain.
2
/ 18 points
9. (24 points) For each reaction below, draw the major organic products. For each product, specify the mechanism by which it was formed.
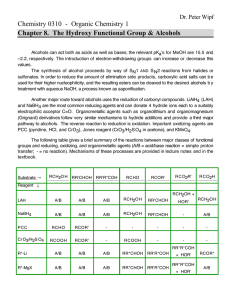
10. (16 points) Fill in the boxes with the missing reagents, reactants, or major products for each reaction below.
3
/ 36 points
(10 continued)
11. (5 points) Draw the mechanism for the following transformation.
12. (6 points) Draw the mechanism for the following transformation.
/ 15 points 4
13. (9 points) Devise a synthesis to the following target using both organic precursors from the chemical bank and any transformation we have learned .
/ 9 points
5