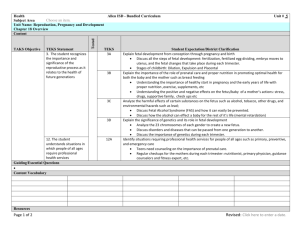
Supplementary Table 3. Overview of observational studies on
advertisement

Supplementary Table 3. Overview of observational studies on associations of triptan exposure during pregnancy and adverse pregnancy outcomes Country Data source Time period Multinational Pregnancy registry (1996–2012) Norway Prescription data base and medical birth registry (2004–2007) Study design Size Exposure Comparison group CH n = 904 T1: Sumatriptan, n = 533 Naratriptan, n = 52 Any triptan, n = 1,465 T1: Sumatriptan, n = 415 Rizatriptan, n = 310 Eletriptan, n = 189 Zolmitriptan, n = 144 Almotriptan, n = 67 Naratriptan, n = 16 None Congenital malformations: NS 1 Disease comparison group (n = 1,095) 2 Any triptan§, n = 139: Naratriptan Rizatriptan Sumatriptan Zolmitriptan Rizatriptan: Any trimester, n = 71 T1, n = 40 Health controls, differed in size for each outcome (n > 47,000) Any triptan, T1, n = 2,742 T2/3, n = 1,215: Sumatriptan Zolmitriptan Rizatriptan Naratriptan Eletriptan Almotriptan Unexposed to antimigraine drugs (n > 1,200,000) Congenital malformations: Disease comparison group (compared to population comparison group): aOR 1.48; 95% CI 1.11–1.97. Miscarriage/stillbirth: NS Maternal postpartum haemorrhage: For any triptan in 2nd trimester: aOR 1.57; 95% CI 1.19–2.07. Prematurity: Disease comparison group (compared to population comparison group): aOR 1.30; 95% CI 1.06–1.60. Apgar score: NS Low birth weight: Disease comparison group (compared to population comparison group): aOR 1.39; 95% CI 1.08–1.81. Spontaneous abortion: For any triptan: OR 2.65; 95% CI 1.57–4.48 Congenital malformations: NS Prematurity: NS Low birth weight: NS Congenital malformations: NS Spontaneous abortion: NS Elective abortion: NS Fetal death: NS Congenital malformation (any): Eletriptan in 1st trimester: RR 5.17; 95% CI 1.07–15.1 Preterm birth: For any triptan in 2nd and/or 3rd trimester: RR 1.31; 95% CI 1.03–1.67 Pregnancy duration: NS Pregnancy complications: NS Neonatal morbidity and mortality: NS Birth weight: NS CH n = 181,125 Canada Prescription data base and medical birth registry (1998–2007) Nested CC n = 59,707 USA Pregnancy registry (1998 to 2012) CH n = 161 Sweden Medical birth registry (1995–2008) and prescription data base (2005–2008) CH n = 1,211,670 Population comparison group (n = 178,565) None Outcome Main findings Ref. 3 4 5 Norway Norwegian Mother and Child Cohort Study database and medical birth registry (1999–2008) CH n = 69,929 Any triptan, n = 1,535 T1: Sumatriptan, n = 653 Rizatriptan, n = 328 Zolmitriptan, n = 243 Eletriptan, n = 179 Naratriptan, n = 31 Almotriptan, n = 29 Disease comparison group (n = 373) Sweden Registry of congenital malformations and medical birth registry (1995–1999) Denmark Prescription data base and medical birth registry (1991–1996) CH n = 912 T1: Sumatriptan, n = 658 Infants exposed in utero to other anti-migraine agents than triptans (n = 254) CH n = 35,950 Sumatriptan†, n = 34 Healthy controls (n = 15,955) USA Clinic visits, telephone interviews (Time period not specified) CH n = 12,339 T1: Sumatriptan, n = 76 Sumatriptan users prior to conception only (n = 92 Multinational Teratology Information Services database (Time period not specified) CH n = 288 Sumatriptan: Any trimester, n = 96 T1, n = 95 Disease-matched controls (n = 96) Health controls (n = 68,021) Migraine controls (n = 89 Congenital malformations: NS Miscarriage/stillbirth: NS Perinatal death: NS Low birth weight: NS Prematurity: NS Apgar score: NS Atonic uterus: Exposure during 2nd and/or 3rd trimester: aOR 2.0; 95% CI 1.3–3.1 Congenital malformations: NS 6, 7 Congenital malformations: not reported in exposed group. Still birth: not reported in exposed group. Prematurity: For exposed group (compared with migraine controls): OR 6.3; 95% CI 1.2–32.0 For exposed group (compared with healthy controls): OR 3.3; 95% CI 1.3–8.5 Low birth weight: NS Congenital malformations: not reported in either group. Still births: not reported in either group. Spontaneous abortion: NS Elective abortion: NS Congenital malformations: NS Miscarriage: NS Elective abortion: NS Gestational age: NS Birth weight: NS 9 8 10 11 Non-teratogen controls (n = 96 Abbreviations: CH, cohort; CC, case–control; T1, 1st trimester; T2, 2nd trimester; T3, 3rd trimester; NS, no excess risk (statistically not significant); aOR = adjusted OR. §Numbers not specified by triptan; †Trimester not specified. 1. 2. 3. Ephross, S.A. & Sinclair, S.M. Final results from the 16-year sumatriptan, naratriptan, and treximet pregnancy registry. Headache 54, 1158-1172 (2014). Nezvalová-Henriksen, K., Spigset, O. & Nordeng, H. Triptan safety during pregnancy: a Norwegian population registry study. Eur J Epidemiol 28, 759-769 (2013). Berard, A. & Kori, S. Dihydroergotamine (DHE) use during gestation and the risk of adverse pregnancy outcomes. Headache 52, 1085-1093 (2012). 4. 5. 6. 7. 8. 9. 10. 11. Merck & Co. Thirteenth/fourteenth annual report from the Merck Pregnancy Registry for Maxalt (rizatriptan benzoate): covering the period from approval (June 1998) through June 12, 2012. (2012). Källén, B., Nilsson, E. & Otterblad Olausson, P. Delivery outcome after maternal use of drugs for migraine: a register study in Sweden. Drug Saf 34, 691-703 (2011). Nezvalová-Henriksen, K., Spigset, O. & Nordeng, H. Triptan exposure during pregnancy and the risk of major congenital malformations and adverse pregnancy outcomes: results from the Norwegian Mother and Child Cohort Study. Headache 50, 563-575 (2010). Nezvalová-Henriksen, K., Spigset, O. & Nordeng, H. Errata in "Triptan exposure during pregnancy and the risk of major congenital malformations and adverse pregnancy outcomes: results from the Norwegian Mother and Child Cohort Study". Headache 52, 1319-1320 (2012). Källén, B. & Lygner, P.E. Delivery outcome in women who used drugs for migraine during pregnancy with special reference to sumatriptan. Headache 41, 351-356 (2001). Olesen, C., Steffensen, F.H., Sørensen, H.T., Nielsen, G.L. & Olsen, J. Pregnancy outcome following prescription for sumatriptan. Headache 40, 20-24 (2000). O'Quinn, S. et al. Pregnancy and perinatal outcomes in migraineurs using sumatriptan: a prospective study. Arch Gynecol Obstet 263, 7-12 (1999). Shuhaiber, S.B. et al. Pregnancy outcome following first trimester exposure to sumatriptan. Neurology 51, 581-583 (1998).