File
advertisement

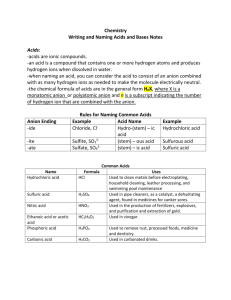
PAWAR PUBLIC SCHOOL,BHANDUP TOPIC: ACIDS, BASES AND SALTS. Question 1 a) Define the following terms. i) Acid ii) Base iii) Salt iv) Alkali v) Indicator b) What is meant by ‘ neutralization reaction’? Give an example. c) Write the chemical equations for the reactions taking place between the following pairs of substances: i) Zinc and dilute hydrochloric acid. ii) Iron and dilute sulphuric acid. iii) Magnesium carbonate and dilute hydrochloric acid. iv) Copper carbonate and dilute hydrochloric acid. d) State whether the following statements are true or false. Rewrite the false statements correctly by changing the underlined word/words. i) Sulphuric acid is a weak acid. ii)Carbonic acid is formed when carbon dioxide dissolves in water. iii) Non –metallic oxides which react with water to form acids are called acidic oxides. iv) All bases are alkalis but all alkalis are not bases. v) The acids formed from the minerals are called organic acids. Question 2 Fill in the blanks: i) Acid + Base → Salt + water ii) Acetic acid is present in __________ while _________acid is present in lemon. iii) The chemical formula of lime water is __________. iv) Water soluble bases are called ______________. v) Neutralization reaction is the reaction between an acid and a _________ to form _______ and _________. vi) Acids turn _______litmus ______. vii) P2O5 is an example of ________ oxide. viii) Acidic oxides react with water to form _________. ix) Na2O is an example of ___________ oxide. x) The chemical name of washing soda is _________. Question 3 a) Choose the odd one and give reasons: i) sulphuric acid, hydrochloric acid, nitric acid, carbonic acid. ii) potassium hydroxide, calcium hydroxide, aluminium hydroxide, iron hydroxide. iii) sodium oxide, carbon dioxide, sulphur dioxide, phosphorus pentoxide. iv) sodium hydroxide, potassium hydroxide, ammonium hydroxide, magnesium hydroxide. v) zinc nitrate, potassium chloride, sodium chloride, calcium hydroxide. b) Rewrite the false statements correctly: i) Acids have bitter taste and they are soapy to touch. ii) Nitric acid is a weak acid. iii) Potassium hydroxide is a weak alkali. iv) The acids obtained from minerals of earth are called organic acids. v) The oxides or hydroxides of sodium or potassium are called weak bases. c) Name the following. i) The acid present in oranges and lemons. ii) An alkali used in the manufacture of soaps. iii) A caustic alkali corrosive in nature. iv) The gas liberated when an alkali is heated with an ammonium salt. v) An example of an insoluble base.