Separation of Mixture Lab: NaCl, Benzoic Acid, SiO2
advertisement

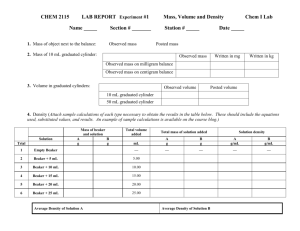
Separation of a Mixture Objective You will be given a mixture containing sodium chloride (NaCl, table salt), benzoic acid (C7 H6O2, a common food preservative), and silicon dioxide (SiO2, sand). Your goal is to separate the substances and determine the percent of each in the original mixture. Introduction Mixtures are not unique to chemistry; we use and consume them on a daily basis. The beverages we drink each morning, the fuel we use in our automobiles, and the ground we walk on are mixtures. Very few materials we encounter are pure. Any material made up of two or more substances that are not chemically combined is a mixture. The isolation of pure components of a mixture requires the separation of one component from another. Chemists have developed techniques for doing this. These methods take advantage of the differences in physical properties of the components. The techniques to be demonstrated in this laboratory are the following: Extraction. This uses a solvent to selectively dissolve one component of the solid mixture. With this technique, a soluble solid can be separated from an insoluble solid. Decantation. This separates a liquid from insoluble solid sediment by carefully pouring the liquid from the solid without disturbing the solid. Recrystallization. This involves dissolving a mixture of solids in a hot solvent, followed by cooling, and crystallization of the more insoluble material. Filtration. This separates a solid from a liquid through the use of a porous material as a filter. These materials allow the liquid to pass through but not the solid. Remember the liquid that passes through the filter is called the filtrate. Evaporation. You know what this is. Procedure 1. Mass the clean, dry 250 mL beaker, “Beaker 1” Mass out about 5 g of your benzoic acid, sand, salt mixture and transfer the mixture to the 250 mL beaker (Beaker 1) 2. Add about 50-mL of distilled water to this beaker and place on the hot plate. While stirring occasionally with your stirring rod, bring the mixture to boiling. Avoid breathing any fumes from the hot liquid. 3. Meanwhile, place about 50 mL of distilled water in a clean 150 mL beaker on the hot plate and heat it until it is boiling. Also, set up an ice water bath using the 1000 mL beaker (fill it about ½ full of ice and add some tap water to it to create an ice/water mixture.) 4. After the liquid begins to boil, use the hot hands to remove the beaker from the hot plate; all benzoic acid and sodium chloride should be in solution (there should be no white solids visible). If necessary, add ~10 mL increments of water/continue heating until dissolution is complete. Too much water will lower your final percent recovery. 5. Immediately decant the supernatant liquid from your hot mixture into a second 250-mL beaker (Beaker 2) guiding the solution by pouring down a glass rod. Use hot hands to hold the beaker of hot solution. Do not let the sand get into the second beaker. 6. To make sure that all the soluble materials are separated from the sand, wash the sand with about 20-mL of boiling water and decant the washing into the second beaker. 7. Place Beaker 2, containing the aqueous solution of benzoic acid and salt, into the ice bath and let it cool for about 15 minutes. Make sure the beaker is surrounded by the ice/water mixture – be careful that the beaker doesn’t tip over and no ice water enters the beaker) 8. While Beaker 2 is cooling, place Beaker 1 (containing the sand) into the gray drying oven. To ensure the sand is completely dry, leave it in the oven for at least 20 minutes. 9. Mass a piece of filter paper and then place it into the Buchner funnel and wet it with a little bit of distilled water. Make sure tubing is connected to water aspirator. 10. After 15 minutes, remove Beaker 2 from ice water bath. Turn on water. Swirl the beaker to suspend the benzoic acid crystals in the liquid and then pour the crystals into the Buchner funnel. Some crystals may remain stuck to the beaker. Use a very small amount of ice water from the water bath to rinse crystals out of the beaker into the Buchner funnel. After all the liquid has passed out of the Buchner funnel into the filter flask, allow the water aspirator to run for a few minutes more – this will draw any remaining traces of liquid through the funnel and away from the benzoic acid crystals. Turn off water. 11. Carefully remove the benzoic acid crystals and filter paper from the Buchner funnel by using a micro spatula to lift the filter paper. Place the crystals/filter paper on a watch glass and place in brown drying oven. Let it dry for a few minutes (If not dry after a few minutes, leave in the oven longer.) Mass the watch glass with filter paper and benzoic acid crystals. 12. Remove the beaker of sand from the oven, and allow it to cool for a while (make sure sand is dry – if not leave it in the oven longer.) Mass the beaker containing the sand. Clean up: Dispose of sand in trash can (do not let any sand go down the sink!) Dispose of the filtrate (containing the dissolved NaCl ) down the drain. Dispose of the filter paper with crystals in the trash. Data: Construct your own data table. As usual, be very descriptive and do not forget units. Calculations: Due to time constraints, we will not recover NaCl (s) from the filtrate in our filter flask. You will calculate % sand and % benzoic acid in the original mix based on 100% yield; calculate % NaCl by subtraction. Post Lab: You will have a post-lab quiz on Friday. You will turn in the lab report before you take the quiz on Friday. The lab report will be worth 35 pts. And the post lab quiz will be worth 50 pts.