V 2 - MsOHara
advertisement

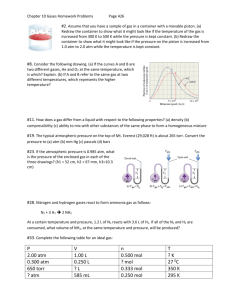
Unit 9: Gas Laws Name: Class Period: Test Date: 1 Chemistry Calendar Monday Tuesday Wednesday Thursday Friday February 6th 7th 8th 9th 10th 6 week assessment Kinetic theory Combined Gas Law Boyle’s Law Ideal Gas Law Charles’ Law Dalton’s Law of Partial Pressure Behavior of Gases 13th 14th 15th 16th 17th Review Gas Law TEST Lab Flautus Chemistry article with accompanying questions. Student Holiday Early Dismissal 18th Student Holiday 2 The Behavior of Gases DAY 1: _______________________________ REVIEW: Kinetic Theory Kinetic refers to motion The energy of an object has because of its motion is called kinetic energy . The Kinetic theory states that the tiny particles in all forms of matter are in constant motion. Watch the video segment (Kinetic Molecular Theory – Standard Deviants School Chemistry: Molecular Geometry) and fill in the missing information: Basic assumptions of the kinetic theory as it applies to gases are: 1. A gas is composed of particles, usually molecules or atoms that are far apart from one another in comparison with their own dimensions. Particles are relatively far apart from one another and between them is empty space. 2. Gas molecules are in constant random motion. They travel in straight paths (unless they collide with a wall of a container) and move independently of each other. 3. The molecules exert no force on each other or on the container until they collide with each other or with the walls of the container. 4. The average kinetic energy of the molecules of a gas is proportional to the temperature. 5. Every time a molecule collides with the wall, it exerts a force on it. 3 Applying this knowledge we know… Gases fill their containers regardless of the shape and volume of the containers. Because there is so much space between particles, gases are easily compressible. Because gases are compressible, they are used in automobile airbags and other safety devices designed to absorb the energy of an impact. All collisions are perfectly elastic. This means that during collisions kinetic energy is transferred without loss from one particle to another, and total kinetic energy remains constant. The average speed of oxygen molecules in air at 20oC is 1700 km/h. At these high speeds, the odor molecules from a hot pizza in Washington, D.C., should reach Mexico City in about 106 minutes. Whey doesn’t this actually happen? Molecules are constantly striking molecules of air and rebounding in other directions. Their path of uninterrupted travel in a straight line is short. Questions: A. What happens when a closed container is inflated? Pressure is increased by the addition of more gas particle. The pressure exerted by an enclosed gas is caused by collisions of gas particles with the walls of the container. If the number of gas particles is changed by any factor, the pressure changes by that same factor; until the container ruptures. 4 B. A gas inside a bicycle tire exerts a pressure of 35 pounds per square inch (psi). How much air must be pumped into the tire to produce a pressure of 70 psi? ** The relationship between amount of gas and pressure is proportional, assuming the volume & temperature stay the same. C. What happens to pressure when a closed container is deflated? The pressure of the gas is decreased because there are fewer gas particles and less collisions. If the number of gas particles decreases by half, the pressure decreases by half. (Note: Gas particles move from region of higher pressure to lower pressure until equilibrium is reached.) 5 Day 2:___________________________ COMBINED GAS LAW The Combined Gas Law helps us explain what happens to gases as the pressure, temperature, and volume changes. P1 V1 P2 V2 __________ = n1T1 __________ n2T2 Letter or Variable Name Unit Number P Pressure atm (atmosphere) mmHg (millimeters of mercury) torr (named after scientist E. Torricelli) kPa (kiloPascals) V Volume L (Liters) mL (milliliters) cm3 (centimeter cubed) m3 (meter cubed) n* T 1 2 Moles Temperature Initial variable Final variable Moles K (Kelvin) Conversions 1 atm = 760 mmHg = 760 torr = 101.3 kPa 1 L = 1000 mL 1 mL = 1 cm3 K = oC + 273 *NOTE: If “n” is not given in a problem, assume it to be 1 mole. STP = Standard Temperature (273 K) 6 and Pressure (1 atm) Guided Practice: 1. The volume of a gas is 27.5 mL at 22.0 oC and 0.974 atm. What will the volume be at 15 oC and 0.993 atm? Givens and Unknowns: Equation: P1 = 0.974 atm V1 = 27.5 mL n1 = 1 mole T1 = 22.0 oC + 273 = 295K P2 = 0.993 atm V2 = unknown n2 = 1 mole T2 = 15 oC + 273 = 288 K P1 V1 __________ P2 V2 = n1 T 1 __________ n2T2 Substitute & Solve: (0.974 atm) (27.5 mL) (1 mole) (295 K) = (0.993 atm) (V2) (1 mole) (288 K) 0.09079661 atm*mL = 0.003447917 atm (V2) (mole) (K) (mole) (K) 0.09079661 atm*mL(mole) (K) = (V2) 0.003447917 atm(mole) (K) 26.32419104 mL = V2 Use 3 sig figs 26.3 mL = V2 2. A 700.0 mL gas sample at STP is compressed to a volume of 200.0 mL, and the temperature is increased to 30.0 oC. What is the new pressure of gas in kilopascals (kPa)? Givens and Unknowns: Equation: P1 V1 P2 V2 P1 = __________ = __________ V1 = n1 T 1 n2T2 n1 = T1 = Substitute & Solve: 7 P2 = V2 = n2 = T2 = 8 Boyle’s Law and Charles’ Law Day 3: ______________________ The combined gas law equation is: P1 V1 P2 V2 __________ = n1 T 1 __________ n2T2 The Effect of Changing Size of Container WHAT IF…temperature and moles do not change and we just look at the relationship between pressure and volume. Our equation would look like this: P1 V1 = Boyle’s Law P2 V2 Boyle’s Law states that at a constant temperature, the volume of a gas is inversely proportional to the pressure exerted by that gas. P ∆ ∆ V Think of two kids (Paul Pressure and Victor Volume) on a see-saw. If Paul goes up, Victor goes down. If Victor goes up, Paul goes down. ∆ 9 Examples: a. If a gas is compressed from 2L to 1L, the pressure will increase by a factor of 2. b. If a gas is expanded from 1L to 3L, the pressure will decrease by a factor of 3. c. Gases cool when they expand and heat when they compress. Why? If the volume of a container is decreased in size, the pressure of gas particles in the container is increased. 10 The Effect of Temperature changes on Volume As the gas inside a balloon cools, the average KE of molecules decreases. With fewer and less collisions, the gas molecules move closer together and occupy a smaller volume than they previously did. The volume decreases, assuming no change in the amount of gas and pressure. V1 ____ = T1 V2 ____ Charles’ Law T2 Charles law states: At a constant pressure, the volume of a gas is directly proportional to the temperature in Kelvin. Mr Charles Mr. Charles is the elevator operator. Thelma Temperature and Violet Volume enter the elevator. When one goes up the other does too! 11 GUIDED PRACTICE: 1. The pressure on 2.5L of anesthetic gas is changed from 760 mmHg to 304 mmHg. What is the new volume if temp is constant? (Boyle’s Law) Givens and Unknowns: P1 = V1 = Equation: P1 V1 = P2 V2 (Boyle’s Law) Substitute & Solve: P2 = V2 = 2. A gas occupies 6.45 L at 0.860 atm. What is the pressure, in atm, if the volume becomes 15.0 L. Givens and Unknowns: P1 = V1 = Equation: P1 V1 = P2 V2 Substitute & Solve: P2 = V2 = 12 (Boyle’s Law) 3. A sample of gas occupies a volume of 80 mL at a temperature of 0oC. What is the new temperature if the volume is decreased to 32 mL? Assume no change in pressure. Givens and Unknowns: Equation: V1 V2 Charles’ Law ____ = ____ V1 = T1 T2 T1 = Substitute & Solve: V2 = T2 = 4. A volume of 3.0L of air is warmed from 50oC to 100oC. What is the new volume if the pressure remains constant? Givens and Unknowns: V1 = T1 = Equation: V1 V2 Charles’ Law ____ = ____ T1 T2 Substitute & Solve: V2 = T2 = 13 Ideal Gas Law DAY 4: ___________________________ Charles’ Law V1 ____ T1 Boyle’s Law V2 = P1 V1 ____ T2 = P2 V 2 Combined Gas Law P2 V 1 __________ P2 V2 = n1T1 __________ n2T2 An ideal gas is one that follows the gas laws at all conditions of pressure and temperature. Such a gas would have to conform precisely to the assumptions of kinetic theory. As you probably suspect, there is __ no gas __ for which this is true. An ideal gas ____ does not ______ exist. Nevertheless, at many conditions of temperature and pressure, __ real gases ___ behave very much like an ideal gas. An important behavior of real gases that differs from that of a hypothetical ideal gas is that real gases can be __ liquefied ________________ and sometimes __ solidified _____________ by cooling and by applying pressure. Ideal gasses cannot be. For example, when water vapor is cooled below 100oC at standard atmospheric pressure, it condenses to a liquid. The behavior of other real gases is similar, although lower temperature and greater pressures may be required. 14 ****Gases behave ideally at ______ high temperatures and low pressure ____________________________________________. If we look at one side of the Combined Gas Law: P = Pressure V = Volume PV nT n = moles T = Temperature and solve it for one mole at STP, you would get a “constant” (symbolized as R). (101.3 kPa)(22.4 L) = 8.31 (L . kPa)/(K . mol) (1 mole)(273 K) We call this the ideal gas constant (R): If pressure is measured in: The ideal gas constant (R) is: kPa atm 8.31 (L . kPa)/(K . mol) 0.0821 (L . atm)/(K . mol) mmHg 62.4 (L . mmHg)/(K . mol) 62.4 (L . torr)/(K . mol) torr P = Pressure SO… PV nT =R V = Volume (must be in liters) n = moles T = Temperature (must be in Kelvin) OR R = Ideal gas constant 15 PV = nRT THIS IS THE IDEAL GAS LAW GUIDED PRACTICE: 1. Calculate the number of moles of oxygen in a 12.5 L tank containing 250 atm, measured at 22oC. Givens and Unknowns: P= Equation: PV = nRT Substitute & Solve: V= n= R = T= 2. If 4.5 g methane gas (CH4) is introduced into an evacuated 2.00 L container at 35oC, what is the pressure in the container, in atm? Givens and Unknowns: P= Equation: PV = nRT Substitute & Solve: V= n= R = T= 3. At STP 150.0 mL of an unknown gas has a mass of 0.250 grams. Calculate its molar mass. Givens and Unknowns: P= Equation: PV = nRT Substitute & Solve: V= n= R = 16 T= Molar mass = ??? Dalton’s Law of Partial Pressures Partial pressure of a gas in a mixture of gases is the pressure which that gas would exert if it were the only gas present in the container. Dalton's Law of Partial Pressures states that the total pressure in a gas mixture is the sum of the partial pressures of each individual gas. Ptotal = Pgas a + Pgas b + Pgas c + etc Dalton's Law of Partial Pressures assumes each gas in the mixture is behaving like an ideal gas. Ptotal = Pgas a = + + 17 Pgas b Dalton’s Gas Law of Partial Pressures Problems For each problem, calculate the pressure using correct units. Box final answer. SHOW ALL WORK!! 1) Three of the primary components of air are nitrogen, oxygen, and carbon dioxide. In a sample containing a mixture of only these gases at exactly one atm pressure (total), the partial pressures are P CO2 = 0.285 torr and PN2 = 593.525 torr. What is the partial pressure of oxygen? P1 = Ptotal = P2 = P3 = 2) Oxygen gas from the decomposition of potassium chlorate, KClO3, was collected by water displacement. 2 KClO3 2 KCl + 3 O2 What is the partial pressure of oxygen collected if the barometric pressure (total) was 731.0 torr and the vapor pressure of water at 20.0C was 17.5 torr? P1 = Ptotal = P2 = 3) A sample of nitrogen gas was collected over water at a temperature of 23.0C. What is the partial pressure of nitrogen if the atmospheric pressure (total) was 785 mmHg and the vapor pressure of water was 21.1 mmHg? P1 = P2 = Ptotal = 18 Day 5 REVIEW 1. Define Boyle’s Law and Charles’ Law. 2. A sample of gas occupies a volume of 95.2 mL at a pressure of 710 torr and a temperature of 30°C. What will be its volume at a standard pressure and 30°C? 3. The volume of a gas is 17.4 liters, measured at standard pressure. Calculate the pressure of the gas if the volume is changed to 19.4 liters and the temperature remains constant. 4. A gas measures 150 mL at 1.00 atmosphere and 27°C. Calculate its volume at 0°C and 1.00 atmosphere. 5. A gas occupies a volume of 4.50 liters at 27°C. At what temperature in °C would the volume be 6.00 liters (the pressure is constant). 19 6. The temperature of 1 liter of gas originally at STP is changed to 200°C at constant volume. Calculate the final pressure of the gas in torr. 7. What are the formulas for the Combined Gas Law and the Ideal Gas Law? 8. A gas occupies 500 ml at 760 torr and 0°C. What volume will it occupy at 10.0 atm and 100°C? 8. A gas occupied 20.0 L at 50°C and 780 torr. Under what pressure in torr would this gas occupy 75.0 liters at 0°C? 9. Calculate the number of moles of oxygen in a 2.5 liter tank containing 25 atm, measured at 23°C. 11. If 2 grams of CH4 (methane gas) is introduced into a 2.0 liter container at 27°C, what is the pressure in the container in mmHg? 20