Asha Devi - Pharmascope.org
advertisement

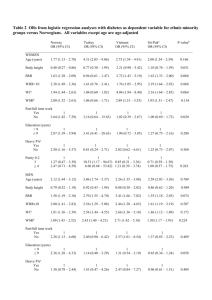
Herbaceous Phyllanthus species extracts promotes longevity in Drosophila melanogaster Manasa N and Ashadevi JS* Department of Zoology, Yuvaraja’s College (Autonomous), University of Mysore, Mysore570005, Karnataka, India Abstract Dietary antioxidants have been associated with various health benefits including ageing and age related diseases. Antioxidant rich diet can neutralize the reactive oxygen species and protect against free radical damage. Many antioxidants are involved in delaying the ageing process and prolong the lifespan. Phyllanthus species are said to be one of the potent rejuvenative herbs in Ayurvedic medicine. The present study has undertaken to investigate the influence of two herbaceous Phyllanthus species extracts namely, P. amarus (PAE) and P. debilis (PDE) in two doses (1 mg and 10 mg/ml) on Drosophila melanogaster (Oregan-K strain) lifespan along with the pre-adult parameters. Stress resistance ability in Phyllanthus extracts treated flies were carried out under starvation and stress inducing molecule (acrylamide) exposure. Our results revealed that both PAE and PDE extends the life span, prolongs the egg to adult rate of development and increases the resistance ability in D. melanogaster. Both the extracts have no effect on viability of the flies. Further, the studies revealed that dose-II of PAE fed flies significantly increased the mean lifespan by 75 % when compared to other treated groups. In addition, the resistance ability for starvation and stress induced toxicity by the supplementation of PAE showed a significant difference with PDE along with the control group. 1 Further phytochemical analysis on these Phyllanthus species might throw a light on the compounds responsible for their biological activities. Key words: Phyllanthus amarus; P. debilis; Drosophila melanogaster; Longevity; Viability; Starvation resistance; Acrylamide * Corresponding author Name: Ashadevi JS Communication address: Dr. Ashadevi JS, Assistant Professor, Department of Zoology, Yuvaraja’s College (Autonomous), University of Mysore, Mysore-570005, India Contact: 09448258374 Email: jsaycm15@gmail.com 2 INTRODUCTION Ageing is an unavoidable, universal, biological phenomenon in all multicellular organisms. Dietary antioxidants have been associated with various health benefits including ageing and age related diseases. Ageing of an organism can be detected by longevity, which is an important fitness component governed by genetic and environmental components (Partridge, 2005). In recent years, the increase in longevity has been achieved by supplementing antioxidant rich pharmaceuticals and medicinal plants. Antioxidant rich diets can neutralize the reactive oxygen species and protect against free radical damage. Many antioxidants are involved in delaying the ageing process and prolong the lifespan. It has been reported that the antioxidant activity of plants might be due to their phenolic compounds that have capacity to scavenge the free radicals (Huda-Fanjan, 2009). One such medicinal plant having high antioxidant property and high phenolic compound is Phyllanthus belongs to the family Phyllanthaceae (Harish and Shivanandappa, 2006; Kumaran and Karunakaran, 2007). Herbaceous Phyllanthus species are potent rejuvenative herbs in Ayurvedic medicine. Major phytochemicals present in these herbs are alkaloids, flavonoids, lignans, phenolics, tannins, steroids and triterpenes (Nahar, 2012). It possesses anti-inflammatory action, with free radical scavenging and protects molecular inflammation (Kassuya, 2006). It is also possess hepatoprotective, anticancer, antiviral, antihepatitis, antidiabetic and immunomodulatory activities (Thyagarajan, 1988 & 2002). Most of the studies on ageing were conducted using long-lived animals like rodents. Since many invertebrates have proved enormous utility in defining the ageing process, researchers have begun to search for pharmacological agents that can extend longevity in invertebrates. A fruit fly, Drosophila melanogaster is one of the ideal model organisms to study the ageing process, as it can be maintained at extremely low cost, easy to culture and have a short 3 life span (Miva, 2004). However, a very few studies on ageing and lifespan has been made using Drosophila (Bahadorani and Hilliker, 2008; Perricone, 2006; Saratikov and Krasnov, 1987). Puri (2002) has analyzed the longevity of D. melanogaster using yellow flowered mountain plant. The extension of longevity on D. melanogaster has been reported using Rhodiola rosea roots and blue berry extracts (Jafari, 2007). Further, it has been proved that the antioxidative property of the plant extracts is the possible mechanism for the life span extension in D. melanogaster (Peng, 2012). Based on the literature survey so far, there are no age related studies on D. melanogaster using herbaceous Phyllanthus species extracts though it is considered as antioxidant rich plant. The analysis of Drosophila ageing using herbaceous Phyllanthus species will be providing a baseline data for further experiments in mammalian systems. In view of this, the present study was chosen to carry out the effect of herbaceous Phyllanthus species that includes P. amarus and P. debilis on life span and fitness parameters in D. melanogaster. MATERIALS AND METHODS Preparation of plant extracts P. amarus and P. debilis were collected from paddy fields near Mysore, Karnataka, India. The aerial parts of the plants were dried and were made into fine powder in the laboratory. The powder was subjected to ethanolic extraction using a Soxhlet apparatus. The extraction was carried out for 12-16 hrs. The crude extract thus obtained was concentrated using a flash evaporator. The collected, dried extract was used for further analysis. All the studies were analyzed in two concentrations namely doses I (1 mg/ml) and dose II (10 mg/ml). 4 Fly strain and culture Oregon- K strain of D. melanogaster was obtained from Drosophila Stock Center, Department of Studies in Zoology, University of Mysore, Mysore, Karnataka, India. Isofemale lines were established from the obtained stock and flies were maintained in ‘Wheat cream agar media’ with yeast granule. The experimental cultures were maintained at 22 ± 1º C with relative humidity of 60 - 70% and further flies were multiplied by the subculture. Yeast supplemented flies were considered as a control group. For all the analyses, extract supplemented flies were considered as treated groups. Ten µl of extracts were added to 10 ml of culture media. Longevity studies Longevity was carried out in unmated male flies of D. melanogaster using standard protocol (Yoon, 1990). Newly emerged male flies were segregated and isolated from the synchronized culture bottles under steriozoom with mild anesthetic conditions. They were released into 9 × 3 cm individual culture vials containing equal quantities of extract supplemented media of P. amarus (PAE) and P. debilis (PDE). Flies were transferred to fresh culture vials once in every three days without being etherized and survival rate was recorded in each vial. Longevity was measured in two doses of each PAE and PDE supplemented groups. Hundred such replicates were maintained in each group. Rate of Development Pre adult rate of developmental experiments were carried out as per the procedure of Ushakumari and Ranganath (1986). Synchronized eggs were used to determine the rate of development and 50 eggs were collected by standard Delcour’s technique (Ramachandra and Ranganath, 1988). The eggs were placed into each culture vial, which were supplemented with 5 PAE and PDE and eggs were allowed for further development. Every day the number of flies emerged in each vial was recorded for a period of 15 days. From this, mean developmental time (in days) was calculated. Twelve such replicates were maintained for each group. Viability For viability study, the same cultures were used which were employed for the rate of development. The total number of flies emerged in each vial was recorded and the percentage of egg to adult viability was calculated. Starvation Resistance Starvation resistance test was carried out as per the method of Huey (2004). Tests were conducted in PAE and PDE supplemented flies of D. melanogaster. Newly emerged male flies were isolated and distributed in groups of 25 males per vial of size 9 × 3 cm containing equal amounts of standard culture media with the extracts and flies were aged for 10 days. Then flies were transferred to empty vials, which were plugged with water soaked sponge. The vials were observed for every six hours once until all flies reached mortality and calculated the resistance ability (hours) in all the analyzed groups. Oxidative stress (OS) resistance test The OS resistance test was carried out according to method of Prasad and Muralidhara (2012) using Acrylamide (ACR)-an oxidative stress-inducing molecule (procured from Sisco Research Laboratories, Mumbai, India). To know the potentiality of the PAE and PDE for resistance under oxidative stress condition, flies were exposed to ACR at different concentrations 6 ranging from 10, 20, 30, 40 and 50 mM to fix the lethal dosage (LC50). Based on LC50 values, 20 mM of acrylamide was chosen for stress resistance test. For conducting the OS resistance test, male flies were segregated from synchronized culture bottles, transformed into culture vials containing the PAE and PDE and aged them for 10 days. Then flies were exposed to 20mM ACR in 5% sucrose solution till all the flies attains mortality and the rate of survival was recorded with an interval of six hours in control and extract supplemented groups. Hundred flies were maintained in each group. Statistical Analysis Data of all the experiments were expressed as mean ± standard error except viability. Viability experiment data was expressed as percent viability. To know the significant difference among the analyzed groups in all experiments, the obtained data was subjected to statistical analyses using the SPSS software (version 20.0). The survival curve for longevity and starvation resistance was obtained from Kaplan-Meier analysis. In each parameter, groups were compared by one-way analysis of variance (ANOVA) followed by Tukey’s test. Values of p<0.05 were considered as statistically significant. To know the relation between longevity and starvation resistance test, Pearson’s correlation coefficient was applied. 7 RESULTS Longevity studies The mean lifespan of extract supplemented flies were shown in table 1. The mortality in case of the control group was initiated at 42 days. The maximum lifespan was 76 days with the mean lifespan of 60.84 ± 0.98 days. The mortality in PAE supplemented groups began at 64 days in dose-I treatment and at 69 days in dose-II treatment. The maximum lifespan in dose-I of PAE group was found to be 117 days with the mean lifespan of 95.82 ± 1.33, whereas in a dose-II group, it was 116 days and mean lifespan was 105.75 ± 0.80 (Figure 1A). However, the mortality in both the dose treatments of PDE supplemented group was initiated at 63 days. The fly survived in maximum was 114 days in dose-I treatment, 127 days in dose-II treatment with the mean lifespan of 90.97 ± 1.46 and 97.54 ± 1.57 days respectively (Figure 1B). The data on statistical analyses reveal that all the five analyzed groups showed significant difference. Both the doses of PAE and PDE treated flies showed highly significant difference with the control group with the p<0.05. Among treatment groups dose-II of PAE showed significant differences with other extract supplemented groups. Table 1: Effect of PAE and PDE on lifespan extension in D. melanogaster Groups Control PAE-I PAE-II PDE-I PDE-II Mean lifespan of D. melanogaster Minimum Maximum Mean ± SE (days) (days) 42 76 60.84a ± 0.98 64 69 63 63 117 116 114 127 95.82b ± 1.33 105.75c ± 0.80 90.97d ± 1.46 97.54e ± 1.57 F value 185.097 df (4, 495) The strains with the different letter in the parenthesis are significantly different at the 5 % level according to DMRT 8 A B Figure 1: Survival curve of D. melanogaster flies supplemented with PAE (A) and PDE (B) along with the control group. Rate of development The patterns of rate of development from egg to adult of all the analyzed groups were graphically represented in figure 2. It was observed that the adults of the control group were started to emerge first where it started to eclosing on the 11th day and continued up to the 17th day, and the peak of emergence was on the 13th day with the mean emergence of 13.51 ± 0.04 day. The adult from the groups of both PAE-I and II dose treatments were started eclosing on 13th day and continued up to 23rd and 24th day respectively with the peak emergence on 16th day (Figure 2A). The mean emergence of PAE-I and II doses were found to be 16.87 ± 0.07 day and 16.40 ± 0.07 day respectively. However, the dose-I and II treatment of PDE groups started emerging on 12th and 13th day respectively, the emergence was extended up to 23rd day. The peak of emergence in dose-I and II were found to be on 17th and 16th day, with the mean emergence of 17.35 ± 0.08 day and 17.01 ± 0.06 day respectively (Figure 2B). The statistical analysis revealed 9 that irrespective of dose, the extract supplemented groups of both the species showed significant 300 300 250 250 % Emergance % Emergance difference with the control group. 200 150 100 50 200 150 100 0 0 11 12 13 14 15 16 17 18 19 20 21 22 23 24 11 12 13 14 15 16 17 18 19 20 21 22 23 24 Days Days Con A 50 PAE I PAE II B Con PDE I PDE II Figure 2: Pattern of development in D. melanogaster flies supplemented with PAE (A) and PDE (B) along with the control group at 22 ± 1 ºC. Viability The data on viability of the extract supplemented groups were graphically complied in the figure 3. The percentage of total flies emerged in control group was found to be 81.5 %, whereas in dose-I and II of PAE showed 81.32 % and 77.33 % respectively. Similarly, the viability in dose-I and II of PDE were found to be 79.5 % and 82.16 % respectively. Among the analyzed groups, the adult viability was found to be least in PAE dose-II, while the highest in PDE dose-II. Statistical analysis revealed that there is no significant difference among control and extract supplemented groups. 10 % Viability 83 82 81 80 79 78 77 76 75 74 CONTROL DOSE-I DOSE-II PAE PDE Groups Figure 3: Viability in D. melanogaster flies supplemented with PAE and PDE along with the control group at 22 ± 1 ºC. Starvation resistance Survival curves for starvation resistance of extracts supplemented flies were plotted in the figure 4. Under starvation, the mortality in control groups was started at 12th hour, the maximum resistance ability was prolonged till 42nd hour. The mean starvation resistance ability was found to be 34.32 ± 0.73 hour. The mortality in PAE dose-I initiated at 12th hour and extended till 54th hour, with the mean starvation resistance ability was found to be 36.3 ± 0.90 hour. In dose-II of PAE supplemented flies mortality was started at 18th hour and the ended at 60th hour with the mean value of 37.14 ± 0.77 hour (Figure 4A). However, in PDE-I and II doses mortality was initiated at 18th hour and extended till 54th hour, with the mean resistance ability was found to be 39.96 ± 0.79 hour and 40.14 ± 0.71 hour respectively (Figure 4B). The statistical result revealed that all the extract-supplemented groups are significantly difference with the control group. 11 A B Figure 4: Survival curve of D. melanogaster on Starvation resistance by the supplementation of PAE (A) and PDE (B). Correlation analysis between longevity and starvation resistance Correlation studies were performed between longevity and starvation resistance in both the PAE and PDE extracts supplemented groups along with control group. The obtained results were plotted in the figure 5 as scatter plots. The results revealed that there is a strong positive correlation exists between the two parameters in all the analyzed groups with the maximum ‘r’ value of 0.961 in PAE dose-I and with the minimum ‘r’ value of 0.813 in dose-II of PAE. In case of PDE-I and II doses the ‘r’ value was found to be 0.935 and 0.954 (P<0.01) respectively. 12 Starvation Resistance 60 CON 50 40 30 20 10 0 0 20 40 60 80 Longevity 70 PAE I Starvation Resistance Starvation Resistance 70 60 50 40 30 20 10 50 40 30 20 60 80 100 Longevity 120 60 70 70 80 90 100 110 120 Longevity 70 Starvation Resistance Starvation Resistance PAE II 60 PDE I 60 50 40 30 20 10 0 PDE II 60 50 40 30 20 10 0 0 50 100 Longevity 150 0 50 100 Longevity 150 Figure 5: Scatter plots showing correlation between lifespan and starvation resistance in flies supplemented with PAE and PDE. 13 Oxidative Stress resistance test The result obtained from oxidative stress resistance test was shown in figure 6. It revealed that mortality in the control group was initiated from 12th hour and prolonged until 48th hour, with mean resistance of 30.96 ± 0.82 hour. In case of PAE-I and II doses, mortality was initiated from 18th hour and ended at 56th hour, with the mean resistance ability of 33.4 ± 0.71 hour and 34.48 ± 0.67 hour respectively. Similarly, in PDE dose–I and II showed the mean value of 30.80 ± 0.78 hour and 32.1 ± 0.89 hour, as the mortality in both the doses of PDE started at 12th hour and completed at 56th hour. Both the doses of PAE and dose–II of PDE treated groups showed better ACR resistance when compared to control group. 120 % Survival 100 80 Con PAE I 60 PAE II 40 PDE I PDE II 20 0 0 6 12 18 24 30 36 Mortality (Hours) 42 48 56 Figure 6: Effect of PAE and PDE on ACR-induced mortality in D. melanogaster flies. 14 DISCUSSION Longevity is the total duration of life span of an individual. It is a good indicator of viability of organisms (Medina, 1996; Strickberger, 1962). Adult diet manipulation has been the principle means of longevity extension in fruit flies (Min, 2007). In the present study, a short life spanned D. melanogaster has been selected as a model organism to know the effect of herbaceous Phyllanthus species extracts on life span and fitness parameters. Experiments were carried out in unmated male flies by supplementing extracts of P. amarus (PAE) and P. debilis (PDE) through adult feeding. Unmated male flies were used for the experiments because of less hormonal effect in these male flies (Demerce, 1994). Jafari (2007) have shown that the root extract of Rhodiola rosea can extend the longevity in D. melanogaster. Antioxidants are considered as longevity promoters (Suckow and Suckow, 2006; Krishna and Watson, 2013). The antioxidant activity of blueberry extracts prolongs the mean lifespan in D. melanogaster and decreased the paraquat-induced mortality rate (Peng, 2012). The high antioxidant rich plants Curcuma longa and Emblica officinalis increases the life span significantly in D. melanogaster (Rawal, 2014). Further it has been reported that a bioactive polyphenolinc compound curcumin extracted from Curcuma longa increases the life span of D. melanogaster (Shen, 2013). The flavonoids and polyphenols of Cocoa increased the mean life span in D. melanogaster (Bahadorani and Hilliker, 2008). Based on the obtained result, the present study revealed that the longevity was increased in both PAE and PDE fed flies when compared to control group. PAE fed groups showed an increase of longevity by 58.3% in dose I treatment and 75% in dose II treatment. The longevity in PDE supplemented groups was increased by 50% and 61.66% in dose I and dose II treatment respectively. Present investigation 15 supports the observation made by earlier researchers on the potentials of plant extracts as longevity promoters (Peng, 2012; Rawal, 2014). The perusal of literatures confirmed the free radical scavenging, antioxidant and other bioactive properties of Phyllanthus species. Further, antioxidant compounds such as polyphenols and flavonoids are reported from these species (Asha, 2004 & 2007; Hashim, 2013; Kumara, 2012; Kumaran and Karunakaran, 2007; Londhe, 2008; Naaz, 2007; Promyothin, 2007; Raphel and Kuttan, 2003). Thus, antioxidant compounds present in the PAE and PDE could be playing a major role in extending the mean lifespan of D. melanogaster. The rate of development and viability are the key fitness parameters in Drosophila. The duration of hatching from egg to adult is rate of development and the total number of flies emerged from known densities of eggs is known as viability. Lint and Lints (1969) have reported the relationship between rate of development and longevity. In the current study, rate of development and adult viability were measured in PAE and PDE fed D. melanogaster flies to know their association with longevity. The result indicated the prolonged rate of development and unchanged viability in both the extract fed flies compared to control flies. This result suggests that PAE and PDE has significant role only in the rate of development, not with the viability of flies. There will be an association between longevity and pre-adult developmental time in extract fed groups. The phytochemicals present in the Phyllanthus extracts could be responsible for increase in longevity and rate of development. One of the ubiquitous causes of stress is the shortage or suboptimal quality of food which can be studied through various resistance test like heat, cold, starvation and oxidative stress. To understand the resistance ability of Phyllanthus species under stress conditions, PAE and PDE 16 fed flies were exposed to starvation as well as oxidative stress with stress-inducing molecule and observed the rate of survival. Starvation resistance can be defined as the resistance to food deprivation (Wayne, 2006). In the present study, the survival rate of PAE and PDE fed flies were significantly more and exhibited better starvation resistance compared to control flies. Among the analyzed test groups, dose II of PDE showed high starvation resistance ability when compared to other groups. Starvation resistance can influence the longevity. It has been reported that the increased mean longevity of Drosophila are due to starvation resistance (Baldal, 2005; Chippindale, 1996 and 1998; Harshman, 1999). Rose (1984) has shown that the increased resistance to starvation was correlated with lifespan extension. The present result of correlation analysis confirms the existence of positive correlation between lifespan extension and starvation resistance in PAE and PDE supplemented flies (Figure 5). This result is in line with the observation of Rose (1984). Numerous studies have demonstrated the effects of plant extracts in reducing oxidative damage and promoting healthy ageing in invertebrate models (Dong, 2012; Mani and Thulasimani, 2012). Similarly, in the present study OS with Phyllanthus extract treated flies had better survival rate compared to OS induced control flies. The rate of survival was more in PAE fed flies under stress induction. This result confirms that the analyzed Phyllanthus species extracts have ability to reduce the oxidative damage. In conclusion, present study summarizes that P. amarus and P. debilis ethanolic extracts increases the life span, rate of development and starvation resistance ability in D. melanogaster. They also show resistance against oxidative stress. The antioxidant compounds present in the PAE and PDE might be causative factors for enhancing these parameters. There is a positive correlation between longevity and starvation resistance. Therefore, we suggest that there is a 17 need for further comparative studies in this area to know which of the antioxidant compounds present in the herbaceous Phyllanthus species has the ability to enhance life span and prevents early ageing in humans. ACKNOWLEDGEMENTS The authors are thankful to the Principal, Yuvaraja’s College, University of Mysore, Mysore for providing the facilities. The first author is thankful to Indian Council of Medical Research, New Delhi for the financial assistance received in the form of Senior Research Fellowship. REFERENCES 1. Asha VV, Akhila S, Wills PJ, Subramoniam A. Further studies on the anti-hepatotoxic activity of Phyllantus maderaspatensis Linn. Journal of Ethnopharmacology 2004; 92: 67-70. 2. Asha VV, Sheeba MS, Suresh V, Wills PJ. Hepato-protection of Phyllanthus maderaspatensis against experimentally induced liver injury in rats. Fitoterapia 2007; 78: 134-141. 3. Bahadorani S, Hilliker AJ. Cocoa confers life span extension in Drosophila melanogaster. Nutrition Research 2008; 28: 377-382. 4. Baldal EA, van der Linde K, van Alphen JJM, Brakefield PM, Zwann BJ. The effects of larval density on adult life-history traits in three species of Drosophila. Mechanisms of ageing and development 2005; 126: 407-416. 18 5. Chippindale AK, Chu TJF, Rose MR. Complex trade-offs and the evolution of starvation resistance in Drosophila melanogaster. Evolution 1996; 50 (2): 753-766. 6. Chippindale AK, Gibbs AG, Sheik M, Yee KJ, Djawdan M, Bradley TJ, Rose MR. Resource acquisition and the evolution of stress resistance in Drosophila melanogaster. Evolution 1998; 52: 1342-1352. 7. Demerec M. Biology of Drosophila. Cold Spring Harbor Laboratory, New York 1994. 8. Dong Y, Guha S, Sun X, Cao M, Wang X, Zou S. Nutraceutical interventions for promoting healthy aging in invertebrate models. Oxidative Medicine and Cellular Longevity 2012; 718491. 9. Harish R, Shivanandappa T. Antioxidant activity and hepatoprotective potential of Phyllanthus niruri. Food Chemistry 2006; 95: 180-185. 10. Harshman LG, Hoffmann AA, Clark AG. Selection for starvation resistance in Drosophila melanogaster: physiological correlates, enzyme activities and multiple stress responses. Journal of Evolutionary Biology 1999; 12: 370-379. 11. Hashim AM, Khan S, Khan MS, Baig MH, Ahmad S. Antioxidant and 𝛼-amylase inhibitory property of Phyllanthus virgatus L.: An in vitro and molecular interaction study. BioMed Research International 2013; 729393. 12. Huda-Fanjan N, Noriham A, Norrakiah AS, Babji AS. Antioxidant activity of plants methanolic extracts containing phenolic compounds. African Journal of Biotechnology 2009; 8 (3): 484-489. 13. Huey RB, Suess J, Hamilton H, Gilchrist GW. Starvation resistance in Drosophila melanogaster: testing for a possible ‘cannibalism’ bias. Fun. Eco., 2004; 18: 952–954. 19 14. Jafari M, Felgner JS, Bussel II, Hutchili T, Khodayari B, Rose MR, Cruz CV, Muller LD. Rhodiola: A promising Anti-aging Chinese herb. Rejuvenation Res 2007; 10: 587-602. 15. Kassuya CA, Silvestre A, Menezes-de-Lima OJr, Marotta DM, Rehder VL, Calixto JB. Anti-inflammatory and antiallodynic actions of the lignan niranthin isolated from Phyllanthus amarus. Evidence for interaction with platelet activating factor receptor. European Journal of Pharmacology 2006; 546 (1-3): 182-188. 16. Krishna MS, Watson U. The effect of pyrogallol on the pre-adult fitness of Drosophila bipectinata. Drosophila Information Service, 2013; 96: 157-164. 17. Kumara SKK, Chethan J, Manasa N, Ashadevi JS, Prakash HS, Bioactive potential of herbaceous Phyllanthus species. International Journal of Pharmacy and Pharmaceutical Sciences 2012; 4(4): 457-461. 18. Kumaran A, Karunakaran RJ. In vitro antioxidant activities of methanol extracts of five Phyllanthus species from India. LWT-Swiss Society of Food Science and Technology 2007; 40: 344-352. 19. Lints FA, Lints CV. Influence of preimaginal environment on fecundity and ageing in Drosophila melanogaster hybrids. I. Preimaginal population density. Experimental Gerontology 1969; 4: 231-238. 20. Londhe JS, Devasagayam TPA, Foo LY, Ghaskadbi SS. Antioxidant activity of some polyphenol constituents of the medicinal plant Phyllanthus amarus Linn. Redox Report 2008; 13: 199-207. 21. Mani V, Thulasimani S. Antiaging effects of Phyllanthus species. In: Kuttan R, Harikumar KB, editors. Phyllanthus Species: Scientific Evaluation and Medicinal Applications. CRC Press, Taylor and Francis Group, USA. 2012; 267-274. 20 22. Medina J. The Clock of Ages, The Cambridge University Press, Cambridge. 1996. 23. Min KJ, Flatt T, Kulaots I, Tatar M. Counting calories in Drosophila diet restriction. Experimental Gerontology 2007; 42: 247-51. 24. Miva S, Riyahi K, Partridge L, Brand MD. Lack of correlation between mitochondrial ROS production and lifespan in Drosophila. Annals of the New York Academy of Sciences 2004; 1019: 388-391. 25. Naaz F, Javed S, Abdin MZ. Hepatoprotective effect of ethanolic extract of Phyllanthus amarus Schum. et. Thonn. on aflatoxin B1-induced liver damage in mice. Journal of Ethnopharmacology 2007; 113: 503-509. 26. Nahar L, Sarker SD, Delazar A. Phytochemistry of the Genus Phyllanthus. In: Kuttan R, Harikumar KB, editors. Phyllanthus Species: Scientific Evaluation and Medicinal Applications. CRC Press, Taylor and Francis Group, USA. 2012; 120-137. 27. Partridge L, Pletcher SD, Mair W. Dietary restriction, mortality trajectories, risk and damage. Mechanisms of ageing and development 2005; 126, 35–41. 28. Peng C, Zuo Y, Kwan KM, Liang Y, Ma KY, Chan HYE, Huang Y, Yu H, Chen ZY. Blueberry extract prolongs lifespan of Drosophila melanogaster. Experimental Gerontology 2012; 47:170–178 29. Perricone N. Stress Reduction Equals Life Extension. 7 Secrets to Beauty, Health, and Longevity: The Miracle of Cellular Rejuvenation. Ballantine Books, 2006. 30. Prasad SN, Muralidhara. Evidence of acrylamide induced oxidative stress and neurotoxicity in Drosophila melanogaster – Its amelioration with spice active enrichment: Relevance to neuropathy. NeuroToxicology 2012; 33(5): 1254-1264. 21 31. Promyothin P, Ngamtin N, Poungshompoo S, Chaichantipyuth C. Hepatoprotective activity of Phyllanthus amarus Schum. et. Thonn. extract in ethanol treated rats: In vitro and in vivo studies. Journal of Ethnopharmacology 2007; 114: 169-173. 32. Puri HS. Traditional Herbal Medicinal for Modern times ‘Rasayana’-Ayurvedic Herbs for Longevity and Rejuvenation. (Dr. Ronald Hardman), Vol 2. 2002. 33. Ramachandra NB, Ranganath HA. Multifaceted approach to evaluate the relationship among closely related forms of Drosophila. Genome 1988; 30: 58. 34. Raphael KR, Kuttan R. Inhibition of experimental gastric lesion and inflammation by Phyllanthus amarus extract. Journal of Ethnopharmacology 2003; 87: 193-197. 35. Rawal S, Singh P, Gupta A, Mohanty S. Dietary intake of Curcuma longa and Emblica officinalis increases Life Span in Drosophila melanogaster. BioMed Research International 2014. 910290, 7 pages. 36. Rose MR. Laboratory evolution of postponed senescence in Drosophila melanogaster. Evolution 1984; 38: 1004-1010. 37. Saratikov AS, Krasnov EA. Tomsk, Russia: Tomsk State University. 1987; 69-90. 38. Shen LR, Xiao F, Yuan P, Chen Y, Gao QK, Parnell LD, Meydani M, Ordovas JM, Li D, Lai CQ. Curcumin-supplemented diets increase superoxide dismutase activity and mean lifespan in Drosophila. Age 2013; 35:1133-1142. 39. Strickberger MW. Experiments in Genetics with Drosophila. John Wiley and Sons, Inc. New York, 1962. 40. Suckow BK, Suckow MA. Lifespan Extension by the Antioxidant Curcumin in Drosophila melanogaster. International journal of Biomedical Science 2006; 2(4): 402405. 22 41. Thyagarajan SP, Subramoniam S, Thirunalasundari T. Effect of Phyllanthus amarus on chronic carriers of hepatitic virus. Lancet 1988; 2: 764-766. 42. Thyagarajan SP, Jayaram S, Gopalakrishnan V, Hari R, Jeyakumar P, Sripathi MS. Herbal medicines for liver diseases in India. Journal of Gastroenterology and Hepatology 2002; 17: S370-S376. 43. Wayne ML, Soundararajan U, Harshaman LG. Environmental stress and reproduction in Drosophila melanogaster: Starvation resistance, ovariole numbers and early age egg production. BMC Evolutionary Biology 2006; 6: 57. 44. Ushakumari A, Ranganath HA. Egg to adult viability and rate of development of five strains of Drosophila in four different media. Drosophila information service 1986; 63: 133. 45. Yoon JS, Gagen KP, Zhu DL. Longevity of 68 Species of Drosophila. The Ohio Journal of Science 1990; 90: 16-32. 23