File
advertisement

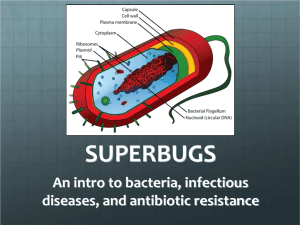
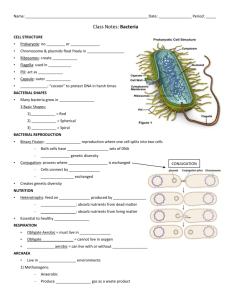
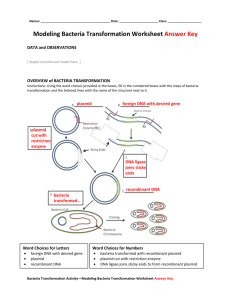
Samar Almarzooqi Biology 110 Section 907 12/3/12 Lab Report Bacterial Plasmid Antibiotic Resistance Genes in Chicken Farms Introduction: Bacterial contamination is often caused by the plasmids located within the bacteria. Plasmids are “circular DNA molecules” separate from the chromosome that are replicated and transmitted to daughter cells during mitosis (Hass and Ward, 2). Plasmids often code for factors that allow the bacterium to live under environmental pressures. One of the most important aspects of a plasmid is that they carry and spread antibiotic resistance genes, making them immune to certain toxins. Bacteria with plasmids containing antibiotic resistance genes have certain advantages because they are harder to kill with antibiotics. Antibiotics are “produced by organisms such as fungi and bacteria” and are utilized by scientists and researchers to prevent bacterial infections and contamination (Hass and Ward, 2). One such example is the use of antibiotics in chicken feed by chicken growers “in order to produce healthier chickens and prevent harmful bacteria from getting into meat and eggs” (Hass and Ward, 2). Over time, the bacteria develop antibiotic resistance, making it difficult to control their growth. As a result, the frequency of antibiotic resistance amongst bacteria has increased. Chicken farms use the antibiotic kanamycin, which helps to interfere with the growth of bacteria. For the lab, three chicken farms, A, B, and C, experiencing problems with Salmonella enteriditis, a bacteria contamination common to eggs, were examined. S. enteriditis cause food poisoning and other health factors, which is why its growth needs to be controlled and prevented. Scientists found that the bacteria contain plasmids with antibiotic resistance to kanamycin, which means that they are resistant to the antibiotic meant to keep them from growing. The three chicken farms, A, B, and C, were isolated as the source of the contaminant eggs. Scientists and the FDA question whether all three farms shared a common source for the bacterial contamination, or if the kanamycin-resistant bacteria development occurs independently in each farm. (Hass and Ward, 2). Kanamycin resistance is controlled by three different genes that code for enzymes that “chemically alter the kanamycin molecule” to make it inactive by disabling it from reaching the ribosomes and keeping the bacteria from synthesizing proteins (Hass and Ward, 3). The presence of kanamycin in chicken feed therefore has no affect on the bacteria and does not prevent contamination due to the presence of the antibiotic resistance in the plasmid. Bacteria reproduce quickly and can grow “to very high concentrations under optimal growth conditions” (Hass and Ward, 14). Samples of bacterial growth on agar plates often lead to billions of bacteria per milliliter, or a “lawn” of bacterial growth, which is uncountable. For this reason, in order to determine the concentration of bacteria, a technique called serial dilution is used. Serial dilutions involve diluting the bacterial sample so that colonies can be easily counted. The dilution is multiplicative, diluting the sample by 1:10 in each step. With each dilution, 10 uL of bacteria to 90 uL of dilutant is the ratio of bacteria to dilutant solution used. The concentration of bacteria is decreased by the same constant each time, so using the equation B=N/D, where B= the initial population size, N= number of colonies, and D= dilution factor, the original concentration, B, of the bacteria can be determined for the (+/-) kanamycin bacteria from the chicken farms (Graziano, 2012). To examine the plasmid containing the kanamycin-resistance, PCR is used to duplicate large amounts of specific DNA fragments. PCR utilizes the DNA polymerase of bacteria who live in extreme conditions, meaning that their enzymes can function in conditions that would normally denature proteins. DNA polymerase from the bacteria Thermus aquaticus is often used, abbreviated Taq polymerase. With the involvement of a DNA template and primers, new DNA can be replicated. PCR involves three different parts: Denaturation, Annealing, and Elongation. In denaturation, the DNA is separated by heating the solution to separate the double helix into two single strands. During annealing, the DNA primers bind to the separate strands, which occurs once the solution is cooled down to a lower temperature. In the final step, elongation, the solution is again heated and the strands are elongated with the use of Taq polymerase, which binds free nucleotides to their base pairs on the original template. This can be done many times to create several copies of a DNA sequence (“Polymerase Chain Reaction”, 2012). In order to determine the kanamycin-resistance plasmids from the bacteria samples from the chicken farms, gel electrophoresis is used to create differing DNA fragments and compare them to the known kanamycin-resistant genes. Gel electrophoresis involves running a voltage across a gel with DNA inserted into different wells. Over time, the DNA will travel from the negative end to the positive end because DNA is negatively charged due to the phosphate groups present. Depending on the size of the DNA fragments, the DNA will travel differing lengths. Larger DNA fragments will travel a shorter length than the shorter DNA fragments. The gel used contains DNA-binding dye and ethidium bromide, which bind to the DNA and allows the fragments to be seen within the gel (Hass and Ward, 17). The purpose of the lab was to determine whether the three chicken farms containing the bacteria resistant to kanamycin shared a common source that caused their bacteria to develop the resistance, or if the bacteria evolved to develop kanamycin resistance independently. By observing factors like a shared food producer, supplier of building materials, or source of laying hens, researchers can determine if the kanamycin resistant bacteria came from a common source (Hass and Ward, 2). If no evidence links the kanamycin-resistant bacteria, then the occurrence is unique and arises independently within each farm. A solution to the rise of contamination must be recommended in order to prevent further outbreak. By evaluating the frequency of kanamycin-resistant bacteria, the severity of contamination can be determined and further steps can be taken to prevent the outbreak from become more extreme. The research questions guiding the lab procedures are: 1) Is the bacterial contamination at these three farms due to the same gene or different genes? 2) What is the frequency of kanamycin resistant bacteria in the chicken farm cultures? (Hass and Ward, 3) With the understanding of how plasmids function in bacteria, our lab group developed a null hypothesis that the kanamycin-resistance bacteria is due to differing genes located in the plasmids that regulate enzymes that disable the kanamycin antibiotic. The alternative hypothesis developed in the lab group was that the kanamycin-resistance in bacteria is due to the same gene within the plasmid. Methods and Materials: The Procedure for the lab was taken from the Bio 110 handout, “Using Genes for Antibiotic Resistance to Trace Sources of Bacterial Contaminations” by Hass and Ward. Prior to the lab session, a prepared sample of bacteria from the farms A, B, and C were taken and a serial dilution was prepared by placing 100 uL on +/- kanamycin plates. The bacteria with kanamycin-resistant genes grew on the plates with kanamycin introduced, and both the number of kanamycin-resistant and non-resistant bacteria were counted. The bacteria containing the kanamycin resistance gene were then isolated and grown. The plasmids from the bacteria were then inserted into the E. coli plasmid and grown on non-kanamycin plates. The original bacterial cultures from each chicken farm each underwent a serial dilution on two separate agar plate, +/- kanamycin. This was done in order to determine the frequency of kanamycin resistant bacteria in the original bacterial population. The data was provided to us in order to calculate the frequency of kanamycin resistance at the chicken farms. The kanamycin resistance gene was then transferred from the bacterial plasmid into E. coli plasmids in order to be able to study the plasmid with a non-pathogenic bacterium. During the lab session, bacterial colonies from the dilution plate with kanamycin introduced were taken and put through PCR in order to replicate fragments of the DNA. Then, the serial dilution plates were observed and lab groups counted the colonies on the plate that yielded countable bacterial colonies for both the +/- kanamycin plates. By comparing the number of bacterial colonies growing on the plate with kanamycin compared to the plate without kanamycin for a serial dilution, a percentage can be determined for the number of cells resistant to kanamycin. The products from PCR were then loaded into a gel and underwent gel electrophoresis. The gel was created by weighing 300 mg of agarose and mixed with 30 mL of 1x TAE buffer measured in a graduated cylinder, was placed in a 125 mL Erlenmeyer flask. The flask was microwaved for about 35 seconds and allowed to cool. Wearing disposable gloves, 1 uL of ethidium bromide was added to the solution. The gel was then placed into the gel electrophoresis machine within the slots. A current was then run through it, and once the DNA fragments had traveled about halfway across the gel, a photograph was taken of the gel. From the photograph of the completed gel, the size of the kanamycin resistance genes from Farms A, B, and C could be determined and compared to the known sizes of kanamycin resistance genes A, B, and C. The frequency of kanamycin resistance at the chicken farms was also calculated, and the severity of the contamination was then determined. Finally, a recommendation to the CDC was made on how to treat the contamination problem. Results: Figure 1: Gel Electrophoresis of Plasmid A Lab groups took pictures of individual plasmids that underwent gel electrophoresis. The figure above shows the DNA fragments from the plasmid from the bacteria from Chicken Farm A. The DNA fragments each traveled a different distance depending on size. The gel includes a DNA ladder and controls A, B, and C for the three known kanamycin genes. The DNA fragment from plasmid A is similar to the known kanamycin resistance gene A; each DNA fragment traveled the same distance. The DNA ladder provides sample to compare each other DNA fragment to and calculate the distance traveled. Figure 2: Gel Electrophoresis of Plasmid B The plasmid from the bacteria taken from Chicken Farm B is shown in the above figure. The differing DNA fragments traveled different distances. The DNA ladder provides a baseline to compare the other DNA fragments to. The known kanamycin resistant genes A, B, and C are controls to compare the plasmid DNA from Farm B to. The plasmid from Chicken Farm B is the same as the kanamycin resistant gene B according to the gel above. Figure 3: Gel Electrophoresis of Plasmid C The plasmid from the bacteria taken from Chicken Farm C is figured above. The plasmid, when compared to the three known kanamycin resistance genes, matches with kanamycin resistant gene C. Similar to Figures 1 and 2, a DNA ladder and the three controls A, B, and C for known kanamycin resistance genes are used. Table 1: Number of Bacteria through Serial Dilutions for Chicken Farms A, B, and C K (N= Number of Colonies) Sample Code (Letter) 10^-2 10^-4 Farm A Farm B Farm C lawn 432 120 20 11 21 10^-6 2 6 11 Non-K (N= Number of Colonies) 10^-2 lawn lawn lawn 10^-4 lawn lawn lawn 10^-6 496 1072 600 Data from different groups was compiled together in class data. The plates that were only diluted by a small degree (10-2 and 10-4 ) were mostly not diluted enough to produce a countable number of bacteria colonies, producing “lawns”. Bacteria from Farm B have the highest number of colonies that grew on plates without the kanamycin resistance. Farm C has the highest number of bacteria with the kanamycin resistance, with 11 colonies growing on 10-6 dilutant plate and 21 colonies on the 10-4 dilutant plate. Table 2: Countable Bacteria from Chicken Farms A, B, and C through Serial Titration Sample Code (Letter) Farm A Farm B Farm C Number of viable bacteria in Original sample (B = N / D ) 10^-2 10^-4 10^-6 4.98x10^9 1.08x10^10 6.11x10^9 The graph shows the number of viable bacteria calculated using the equation B =N/D from the value from Table 1 to calculate the original number of bacteria from each farm before the titration. The original concentration can only be calculated from 10-6 dilutant because the other two contained lawns, which are uncountable. Farm B has the higher number of bacteria from the original sample, Farm C has the second highest, and Farm A has the least amount of original bacteria concentration. Table 3: % Contamination of Bacteria from Chicken Farms A, B, and C Farm A Percent Contamination = level of bacterial contamination x 100 0.133 % Farm B Farm C 0.187 % 0.61 % Sample Code (Letter) The % contamination represents the contamination efficiency of the bacteria from all three farms, and contamination efficiency above 0% is attributed as successful. Farm C had the most successful contamination rate (.61%) which means that .61% of the eggs from the farms are contaminated. Farm B has the second highest percent contamination, .187%, and Farm A has the least amount of percent contamination, .133%. Table 4: Frequency of Antibiotic Resistant Bacteria in Chicken Farms A, B, and C Sample Code (Letter) Farm A Farm B Farm C Frequency of antibiotic resistant bacteria = K / no K 10^-2 10^-4 10^-6 #VALUE! #VALUE! 0.004032258 #VALUE! #VALUE! 0.005597015 #VALUE! #VALUE! 0.018333333 The graph represents the frequency of antibiotic resistant bacteria compared to the bacteria without the antibiotic resistance to kanamycin using the equation: 𝑛𝑢𝑚𝑏𝑒𝑟 𝑜𝑓 𝑘𝑎𝑛𝑎𝑚𝑦𝑐𝑖𝑛 𝑟𝑒𝑠𝑖𝑠𝑡𝑎𝑛𝑡 𝑏𝑎𝑐𝑡𝑒𝑟𝑖𝑎 𝐾 = 𝑛𝑜 𝐾 for the bacteria diluted to 10-6. Farm C 𝑛𝑢𝑚𝑏𝑒𝑟 𝑜𝑓 𝑏𝑎𝑐𝑡𝑒𝑟𝑖𝑎 𝑤𝑖𝑡ℎ𝑜𝑢𝑡ℎ 𝑘𝑎𝑛𝑎𝑚𝑦𝑐𝑖𝑛 𝑟𝑒𝑠𝑖𝑠𝑡𝑎𝑛𝑐𝑒 has the highest frequency of antibiotic resistance amongst the bacteria, 0.018, while Farm B and A have similar frequencies, both smaller than that of Farm C, 0.006 and 0.004 respectively. Sample Calculations: Sample Equation 1: Table 2 Calculations B= N/D Farm A: B= 496/ (1 x 10-6) = 4.98 x 109 bacteria Sample Equation 2: Table 4 Calculations Frequency of antibiotic resistant bacteria: K/no K Farm A (10-6): 2/496= 0.004 Table 5: Identification of Plasmid Antibiotic Resistance for Chicken Farms A, B, and C FARM PLASMID Plasmid B Plasmid A A Plasmid C X X B C X Using the gel electrophoresis images in Figures 1, 2, and 3 of the plasmids from the different Chicken Farms, the identity of the DNA fragments can be determined. Farm A bacteria has resistance gene for kanamycin A. Farm B bacteria has the resistance gene for kanamycin B. Farm C bacteria has the resistance gene for kanamycin C. Table 6: Recommendation Guidelines for Bacterial Contamination at Egg Producing Facilities Level of Contamination Bacterial contamination < 5% Bacterial contamination between 6-30% Bacterial contamination >31% Standard Recommendations Divert eggs to pasteurization facility until contamination levels have been <1% for 8 weeks Monitor farm weekly Identify source(s) of contamination Change antibiotic regime Destroy all eggs until levels have been <1% for 8 weeks Identify and destroy infected individuals Monitor farm weekly Identify source(s) of contamination Change antibiotic regime Cull entire population Disinfect entire facility Identify source(s) of contamination Submit contamination prevention plan From “Using Genes for Antibiotic Resistance to Trace Sources of Bacterial Contaminations” by Hass and Ward. Using the table above, one the level of contamination has been determined for each farm, the steps above are the precautions that need to be recommended to each farm. Discussion: From the data presented in the Results section, the research questions the lab sought to answer can be explained and determined. 1) Is the bacterial contamination at these three farms due to the same gene or different genes? 2) What is the frequency of kanamycin resistant bacteria in the chicken farm cultures? The first question can be answered by interpreting the images for the gel electrophoresis of the three plasmids from the three farms. For each gel, a sample of the DNA fragment after PCR was conducted was placed in a well. A DNA ladder and three controls of the three known kanamycin resistant gene in bacteria (A, B, and C) were also placed in differing wells. By comparing the distance the plasmid from Farm A traveled, it matches the distance traveled by the bacteria kanamycin resistance gene A. Both DNA fragments, meaning that they are the same size, therefore the same gene. By observing the images of gel electrophoresis for plasmids from Farms B and C, it can be determined that the plasmid from Farm B contains gene B for the kanamycin resistance gene and Farm C bacteria with kanamycin resistance contains gene C. Due to the differing genes for kanamycin resistance genes amongst the three farms, the bacterial contamination is due to separate genes. If they were due to the same genes, then all of the plasmids from all three farms would have the same band distance after gel electrophoresis because they would all be the same size. Because the source of contamination is due to separate genes, then the contamination is unique to each farm and did not stem from a common source like common feed producers or building material manufacturers each farm uses. Therefore, each farm experienced a different source of contamination for bacteria S. enteriditis, leading to separate antibiotic resistance genes to kanamycin in the chicken feed. This conclusion supports the null hypothesis adapted by the group that the kanamycin-resistance bacteria from the three farms stems from differing genes located in the plasmids that regulate enzymes that disable the kanamycin antibiotic. The alternative hypothesis that that the kanamycin-resistant bacteria is due to the same gene within the plasmid does not hold true because as evidence by gel electrophoresis of the bacterial DNA from the three farms, the genes for the kanamycin resistance are different. The number of colonies of bacteria from the three farms growing on the plate with kanamycin introduced represents the bacteria with the kanamycin resistance gene. A serial dilution needed to be conducted because for both the 10-2 and 10-4 had lawns of bacteria, making it uncountable. For the bacteria growing on the dilutant 10-6 plate, the number of colonies could be counted for both the non-kanamycin plate and kanamycin plates. Table 1 shows that Farm C had the greatest number of bacterial colonies that grew on the plates with kanamycin, meaning that it had the greatest number of bacteria with kanamycin resistance genes. From Table 1, the original number of colonies from each farm were determined in Table 2 using the equation B=N/D. The values of B, the original number of bacteria, are represented in Table 2. The bacterial concentration from the three farms range from the highest concentration in Farm B, 1.08x1010 bacteria, to the lowest concentration from Farm A, 4.98 x 109 bacteria. Farm C had the middle concentration of bacteria in the original sample, 6.11 x 109 bacteria. The original number of colonies of bacteria from each farm is used to determine the percent contamination in Table 3. The percent contamination represents the percent efficiency of the plasmid transfer from the S. enteriditis bacteria to the E. coli plasmid used in the lab due to the danger of using the harmful bacteria contaminating the eggs and leading to diseases. A percent contamination about 0% is classified as successful, but the higher percentages represents a more successful and efficient transformation. Farm C had the greatest percent contamination, 0.61%. Farm B had the second higher, 0.187% and Farm A had the lowest percent contamination, 0.133%. All three bacteria samples from the three farms were efficiently transformed into the E. coli, but the percentages are still very low, all less than 1.0%. The sources of error in the transformation of the plasmid to the E. coli plasmid can be due to error in the laboratory procedures. Incorrectly counting the number of bacteria growing on the plates leads to an inaccurate original concentration of bacteria, and because there were many colonies even in the serial dilution plates, lab groups still had to count hundreds of colonies. Overestimating the number of bacterial colonies growing on the non-kanamycin plate leads to a smaller percent contamination value since the number of bacterial colonies growing on plates with kanamycin resistance were easy distinguishable and countable. Addressing the second research question of the lab, the frequencies of kanamycin resistance in chicken farm cultures are shown in Table 4. As a class, the frequencies were calculated since each lab group examined bacteria from one of the three farms. The frequencies of antibiotic resistance were calculated by using the question: K/no K from Table 1. Farm C had the highest frequency of antibiotic resistance to kanamycin amongst the bacteria, 0.018. Farm B had the second highest, 0.006 frequency of antibiotic resistance, and Farm A had the lowest frequency of antibiotic resistant bacteria, 0.004.The order Farm C, Farm B, Farm A represents both the ranking from highest to lowest frequency and the ranking of percent contamination for the recombination of efficient transformation. Using the Table 6, recommendations need to be given to each farm depending on the bacterial contamination percentage. All three farms had a level of contamination <5%, which means that all three farms are recommended to follow the same steps to make sure a contamination outbreak does not occur again. All three farms should move all eggs produced to a pasteurization facility until percent contamination levels are below 1% for about 8 weeks. The farm needs to identify the sources of contamination, and since the kanamycin resistant bacteria all had separate genes, A, B, or C, the source of contamination is most likely not similar amongst the farms. The antibiotic regime of kanamycin in the chicken feed needs to be changed since the bacteria have evolved to be able to disable the kanamycin antibiotic from working, and a monitoring of the farm would ensure that the eggs are not being subject to contamination again (Hass and Ward, 12). Sources of error in the lab could have occurred in many different steps since the lab required a lot of precise measurements to be conducted, especially in the PCR and gel electrophoresis portion. For gel electrophoresis, similar quantities and concentrations of DNA needed to be placed into the wells without contamination. Any error could influence the behavior of the DNA strands once the current was run across the gel. If too much DNA samples were placed into wells, then the DNA would not travel as far across the gel electrophoresis not because of the size of the DNA fragments, but because of the concentration and amount of DNA added to the wells. Other sources of error include human error in counting the number of bacterial colonies on the plates. Any miscalculation impacts the calculations for frequency and percent contamination of each farm due to the bacterial development of the antibiotic resistance to kanamycin gene. For future experiments to perform, it would be useful to further study the development of antibiotic resistance in bacteria because if the development of antibiotic resistance could be controlled and depleted, antibiotics would be more effective and new treatment options would not have to be examined every time a strain of bacteria developed resistance to antibodies. One such experiment could be comparing the frequency of antibody resistance for two separate antibodies to determine the most useful and efficient antibody to use for chicken feeders. A study conducted by Dr. Levy in “The Spread of Antibiotic Resistant Bacteria from Chicken to Farmers” involved the usage of a separate antibiotic, tetracycline, to determine the frequency of the development of antibody resistance amongst the bacteria (Solomon, 2011). According to the study, within a week of the introduction of the antibiotic to the chicken feed, almost all of the bacteria contained tetracycline resistant genes. Depending on the antibiotic, there are different rates of the development of the resistance, some much more easily than others. By examining the differing frequencies of antibiotic resistance development for differing antibiotics, a more effective antibiotic can be developed and used (Solomon, 2011). The same procedures conducted in this lab would be conducted, but multiple times using differing antibiotics. The frequencies of antibiotic resistance would be examined and the bacteria with the least frequency would be the most successful antibiotic because the bacteria is then less likely to develop resistance at a faster rate. Conclusion: From the results of frequencies of the antibiotic resistance kanamycin in the lab, Farm C was determined to have highest frequency, 0.018. All three farms had percent contaminations less than 5%, which means all three farms need to take similar steps to prevent future or further breakout, which includes monitoring the eggs. From gel electrophoresis, the kanamycin resistance genes for the bacteria from the three farms was determined to be different, Farm A had gene A, Farm B had gene B, Farm C had gene C. The null hypothesis was accepted because it was determined that bacterial contamination at these three farms was due to different genes, and therefore did not stem from a similar source. Further studies should be researched to determine the most efficient use of antibiotics that limit the ability of the bacteria to develop the resistance because antibiotics provide a way to prevent bacterial contamination. Resources: Cyr, R., 2002. Title of the tutorial being cited. In, Biology 110: Basic concepts and biodiverity course website. Department of Biology, The Pennsylvania State University. http://www.bio.psu.edu/ Cyr, R., Hass C., Woodward D., and Ward A., 2010. Using Genes for Antibiotic Resistance to Trace Source(s) of Bacterial Contamination. In, Biology 110: Basic concepts and biodiverity course website. Department of Biology, The Pennsylvania State University. http://www.bio.psu.edu/ Graziano, Maria. “Serial Dilution.” Class lecture, Pennsylvania State University, State College, PA, October 22, 2012. “Polymerase Chain Reaction”. Cold Spring Harbor Laboratory, 2012. Web. Solomon, Gina. The Spread of Antibiotic Resistant Bacteria from Chickens to Farmer. Natural Resources Defense Council, 2011. Web.