Regents Chemistry Lab # 6
advertisement

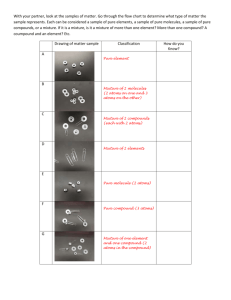
RUBRIC Regents Chemistry Lab # 5 2014-15 Name: _____________________________ Date Due: Friday 10/17/14 Nuts & Bolts Classifying Activity Section 1: Define the following: 1. Element: Section 1:________/7 Section 2:________/3 Section 3:________/5 Section 4:________/5 2. Compound: 3. Molecule: 4. Atom: 5. Mixture: 6. Pure Substance 7. Homogenous Mixture: 8. Heterogeneous Mixture: 9. Can a pure substance contain more than 1 kind of atom? Explain. 10. Explain how can you tell atoms and molecules apart? Section 2: Practice a) mixture or pure substance b) element only, compound only, or both c) atoms only, molecules only, or both d) symbolic notation can hand write these answers Section 3: Data Table Petri dish # #1 #2. #3. #4. #5. #6. #7. #8. #9. (A) Mixture or pure substance (B) Compounds only, Elements only, or both (C) Atoms only, Molecules only, or both Symbolic Notation hand write answers in this column before turn in final lab report Section 4: Draw particle diagrams depicting each of the following: use if need to redraw your illustrations monatomic element only diatomic element only triatomic element only diatomic compound only triatomic compound only mixture of elements only mixture of compounds only element in the liquid phase mixture of compounds & elements element in the gas phase Looking at the particle diagrams on the right… 1) which contains a mixture of compounds? 2) which contains elements in the solid phase? 3) which contains elements in the gaseous phase? 1. 2. 3. element in the solid phase mixture:compounds(g) &elements(s)