Basic Characteristics of Light
advertisement

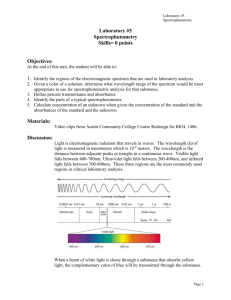
Lab 3 Spectrophotometry Photometry: - Photometry means “the measurement of light” - Photometer & Spectrophotometer are instruments used for this type of measutment, in which a photocell or photomultiplier tube is used to detect the amount of light that passes through a colored solution from a light source. Basic Characteristics of Light: - Light is a form of electromagnetic energy that travels in waves. - The wavelength of light is the distance between two beaks of the light wave, is inversely proportional with its energy. - In the visible region the color of light is a function of its wave length increasing from violet towards the red color. - Objects that appear colored absorb light at particular wavelength and reflect the other parts of the visible spectrum resulting in many shades of color. 1 Spectrophotometry : Is a method to measure how much a chemical substance absorbs light by measuring the intensity of light as a beam of light passes through sample solution. Each compound absorbs or transmits light over a certain range of wavelength. This measurement can also be used to measure the amount of a known chemical substance. Every chemical compound absorbs, transmits, or reflects light (electromagnetic radiation) over a certain range of wavelength. Spectrophotometry is a measurement of how much a chemical substance absorbs or transmits. Is an instrument that measures the amount of photons (the intensity of light) absorbed after it passes through sample solution. With the spectrophotometer, the amount of a known chemical substance (concentrations) can also be determined by measuring the intensity of light detected. Types of spectrophotometers : Depending on the range of wavelength of light source, it can be classified into two different types: 1. UV-visible spectrophotometer: uses light over the ultraviolet range (185 - 400 nm) and visible range (400 - 700 nm) of electromagnetic radiation spectrum. 2. IR spectrophotometer: uses light over the infrared range (700 15000 nm) of electromagnetic radiation spectrum. Basic structure of spectrophotometers 2 Spectrophotometer in general consists of two devices: • Spectrometer: It produces a desired range of wavelength of light. First a collimator (lens) transmits a straight beam of light (photons) that passes through a monochromator (prism) to split it into several component wavelengths (spectrum). Then a wavelength selector (slit) transmits only the desired wavelengths . • Photometer: After the desired range of wavelength of light passes through the solution of a sample in cuvette, the photometer detects the amount of photons that is absorbed and then sends a signal to a galvanometer or a digital display . Beer-Lambert Law: Beer-Lambert Law (also known as Beer's Law) states that there is a linear relationship between the absorbance and the concentration of a sample. For this reason, Beer's Law can only be applied when there is a linear relationship. Beer's Law is written as: where A is the measure of absorbance (no units), ϵ is the molar extinction coefficient or molar absorptivity (or absorption coefficient), l is the path length, and c is the concentration. The molar extinction coefficient is given as a constant and varies for each molecule. Since absorbance does not carry any units, the units for ϵ must cancel out the units of length and concentration. As a result, ϵ has the units: L·mol-1·cm-1. The path length is measured in centimeters. Because a standard spectrometer uses a cuvette that is 1 cm in width, l is always assumed to equal 1 cm. Since absorption, ϵ, and path length are known, we can calculate the concentration c of the sample. 3 If the concentration of a solution is increased, the It will decrease and then % T is decreased. - The relationship between the concentration and %T is not linear, but if the logarithm of the %T is plotted against the concentration, a straight line is obtained . -The term absorbance is used to represent – log % T A = - log % T = 1/ log % T % T = It \ Io x 100 % For x substance For standard substance Ax = a b c x Ast = a b cst From 1,2 Ax Cx Ast Cst 4 (1) (2) Cx = Ax . Cst Ast Glucose test (GOD) Equipment: Automatic pipettes Tips Test tubes Glucose reagent Reagents TRIS buffer pH 7.5, phenol, Glucose oxidase, Peroxidases, 4 – Aminophenazone Principle Glucose is oxidized by glucose-oxidase to gluconate and hydrogen peroxide and gives pink color read at 500nm. Sample Serum free of hemolysis plasma. Serum or plasma glucose is stable for at least 3 days at 2-8ºc. Procedure Blank Standard Stander Sample 10µl Sample 10 µl Control Reagent Contro l 1.0µl 1.0ml 1.0 ml 1.0 ml Mix. Incubate 10 min. at 37ºc. or 30 min at room temperature. 5 Measure the Absorbance at 505nm (490-550) against the blank. The color is stable 60 min. Calculation Glucose (mg/dl) = (Abs. sample/Abs. standard)* standard conc. Linearity This method is linear up to 500mg/dl. If the glucose concentration is greater than 500mg/dl. , dilute the sample 1:2 with saline solution and repeat the determination and multiply the result by 2 Reference value Serum: 70 -110mg/dl 6