1. Introduction - I
advertisement

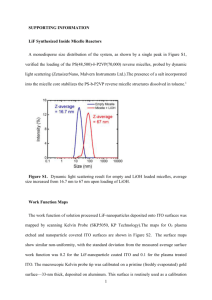
6th International Science, Social Sciences, Engineering and Energy Conference 17-19 December, 2014, Prajaktra Design Hotel, Udon Thani, Thailand I-SEEC 2014 http//iseec2014.udru.ac.th Self-catalyst synthesis of ITO tree-like nanostructures by chemical vapor deposition technique Phan Van Cuonga,e1, Tran Tien Phucb,e2, Do-Hyung Kimc,e3 b a Department of Physics, Nha Trang University, 02 Nguyen Dinh Chieu Street, Nha Trang, Vietnam Department of Industrial Electricity, Nha Trang University, 02 Nguyen Dinh Chieu Street, Nha Trang, Vietnam. c Department of Physics, Kyungpook National University, Daegu 702-701, Republic of Korea e1 cuongpv@ntu.edu.vn , e2phuctt@ntu.edu.vn, e3kimdh@knu.ac.kr Abstract Indium-tin oxide (ITO) tree-like nanostructures (Ns) were self-catalyst synthesized successfully using the chemical vapor deposition (CVD) technique. The synthesized products were characterized and analyzed using the scanning electron microscopy (SEM, Hitachi, S-4300), and the high-resolution transmission electron microscopy (HRTEM, Philips, CM 200). Characterized results show that the ITO nanostructures have branches with the four-fold symmetry, the trunk of tree-like Ns is about 100nm and the branches are smaller than 50nm. At the end of the trunk and the branches remain the sphere nanosized droplets, which play a role as a self-catalyst for nano structure growth. The chemical components on the top, middle, and bottom of the tree-like Ns were analyzed and shown that the indium (In) component dominated over other components in the ITO Ns. In this report, we also highlighted and explained the possible growth mechanisms of ITO Ns. Keywords: ITO, nanostructure, chemical vapor deposition, CVD 1. Introduction Synthesis and control of nanostructure growth based on a bottom-up approach is important for future nanodevice applications. Many oxide nanomaterials have been synthesized and studied in order to investigate the novel properties and applications [1, 2]. Indium-tin oxide is one of the most widely used transparent conducting oxides. There have been several reports on the synthesis and the structure characterization of ITO nanostructures [3, 4]. Here we report on self-catalyst synthesis of ITO tree-like nanostructures via chemical vapor deposition (CVD) technique. 2 Nomenclature ITO Indium-tin oxide Ns Nanostructures CVD Chemical vapor deposition SEM Scanning electron microscopy TEM Transmission electron microscopy HRTEM High-resolution transmission electron microscopy In Indium Sn Tin nm Nanometer sccm Standard cubic centimeters per minute SAED Selected area electron diffraction VLS Vapor liquid solid 2. Experimental Equal amounts of commercial ITO powder (10 wt% SnO 2 and 90 wt% In2O3, 99.99% purity) and graphite powder (99.99%) were mechanically pulverized together and transferred to an alumina boat. The alumina boat was placed in the center of a quartz tube furnace. Bare silicon substrates were inserted into a tube positioned about 20cm from the center of the boat under a constant flow of argon [Ar, 99.999%, 50 sccm (Standard cubic centimeters per minute)] as the gas carrier. Oxygen (O2, 99.999%, 2 sccm) was added as the reactive gas. The pressure was maintained at 1.7 Torr. The furnace was then heated to 900oC and the corresponding substrate temperature was about 600 oC. The schematic representation of experimental set up is shown in figure 1. Figure 1. The schematic representation of experimental set up 3. Rerults and Discussion 3 The SEM images of the tree-like Ns are shown in figure 2. Figure 2a was captured from the side, the ITO Ns were grown densely and look like a “forest of ITO nanostructures”. Figure 2b was taken from the top of ITO Ns and clearly shown that the ITO Ns have branches with the four-fold symmetry. (a) (b) Figure 2. The SEM images of the tree-like Ns (a and b were captured from the side and on the top, respectively) Figure 3a shows the TEM image of the ITO tree-like Ns. The diameter of the trunk is about 100nm and the branch diameter is smaller than 50nm. At the end of the trunk and the branches remain the sphere nanosized droplets. The chemical components on the top, middle, and bottom of the tree-like Ns are shown in figure 3b which shows that the indium (In) component dominated (around 77%) over other components in the ITO Ns. The tin component is 8.9% and 9.5% at the middle and the bottom, respectively. While at the top the tin ratio is only 2.4%. (a) (b) Figure 3. (a) TEM image of the tree-like Ns, (b) Chemical components on the top, middle, and bottom of the treelike Ns. 4 Figure 4a shows The HRTEM images taken from the trunk of ITO Ns are shown in figure 4a and 4b. The images reveal that the surface of ITO Ns is quite clean. The inset of figure 4b shows the corresponding SAED pattern and indicates that ITO Ns are single crystalline. The HRTEM image (4b) exhibits a lattice spacing of the ITO Ns about 0.25 nm. (a) (b) Figure 4. The HRTEM images taken from the trunk of ITO Ns. The inset of 4b shows the corresponding SAED pattern. The key synthesis technique of the ITO Ns follows the chemical vapor deposition technique (CVD, also called VLS mechanism [5] suggested for the first time by Wagner and Ellis in 1964). Using this method, Wagner and Ellis synthesized the micro size silicon wires with gold catalyst. The CVD processes can be summarized as follow: The thin gold film was prepared on the silicon substrate, when the thin gold film was heated at a suitable temperature; it was melted and formed the nanosize gold droplets on the silicon substrate for the first state. The silicon vapour source was carried and adsorbed onto the gold droplets, then deposited onto silicon substrate, these processes were repeated - forming the micro size silicon wires (see figure 5). Figure 5. The scheme of the CVD mechanism for growing the micro size silicon wires 5 In our study, bare silicon substrate without thin gold film deposited was used to do experiments. Base on the images of SEM and TEM, the chemical components which were investigated above, our suggestion and explanation for the self-catalyst growth mechanism of tree-like Ns following the CVD mechanism, are shown in figure 6. It is well known that indium metal had a lower melting point (about 156oC) than tin metal (Sn, about 232oC). Moreover, the weight ratio of In2O3 was much larger than that of SnO2 in source materials and In components dominated (see figure 3b) of the ITO Ns. We conclude that In metal may play a role as a self-catalyst. At the first stage (I), In formed clusters, then nanozise droplets of In were formed on the Si substrate, these nanozise droplets play a role as the gold catalyst droplets. Then the growth processes of the ITO Ns trunk will be followed the CVD method using the ITO vapour material source. We also know that inside the quartz tube, the condense level of In vapor was much higher than that of Sn vapor; thus, In nanosize droplets may form on the side walls of the trunk, these In nanosize droplets will be served as a catalyst for the growing of the ITO Ns branches). Similarly, ITO vapor from the source material, carried by Ar, adsorbed on the nanosize droplets (formed on the trunk) to grow the ITO Ns branches (stage II). Figure 6. The suggestion of growth mechanism of ITO tree-like Ns follows CVD mechanism with a self-catalyst. 4. Conclusion Tree-like ITO nanostructures were successfully synthesized by the chemical vapor deposition technique. The products were characterized and analyzed using SEM, TEM. The results show that the ITO nanostructures have branches with the four-fold symmetry. The ITO Ns diameter of the trunk and the branch is less than 100nm and 50nm, respectively. At the end of the trunk and the branches remain the self-catalyst sphere nanosized droplets. The chemical components on the top, middle, and bottom of the tree-like Ns were analyzed and shown that the indium (In) component dominated over other components of the ITO Ns. The HRTEM images and SAED pattern indicate that ITO Ns are single crystalline with the lattice spacing about 0.25 nm. The possible growth mechanisms of ITO Ns are also suggested and explained in this paper. 6 Acknowledgements This work has been supported by KRF grant (No. KRF-2006-C00536). References [1] [2] [3] [4] [5] Rao CNR, Kulkarni GU, Govindaraj A, Satishkumar BC, and Thomas JP. Metal nanoparticles, nanowires, and carbon nanotubes, Pure Appl. Chem. 2000; 72: 21-3. Rao CNR, Agrawal VV, Biswas K, Gautam UK, Ghosh M, Govindaraj A, Kulkarni GU, Kalyanikutty KP, Sardar K, and Vivekchand SRC, Soft chemical approaches to inorganic nanostructures, Pure Appl. Chem. 2006; 78: 1619–50 Nguyen P , Hou TN, Kong JK, Cassell AM, Quinn R, Li J ,Han J, McNeil M, and Meyyappan. M, Epitaxial Directional Growth of Indium-Doped Tin Oxide Nanowire Arrays, Nano Lett. 2003; 3: 925–8 Wan Q, Wei M, Zhi D, MacManus-Driscoll JL, and Blamire M, Epitaxial Growth of Vertically Aligned and Branched SingleCrystalline Tin-Doped Indium Oxide Nanowire Arrays, Adv. Mat. 2006; 18: 234–8 Wagner RS and Ellis WC, VAPOR-LIQUID-SOLID MECHANISM OF SINGLE CRYSTAL GROWTH, Appl. Phys. Lett. 1964; 4: 89-90