Supplimentary Material
advertisement

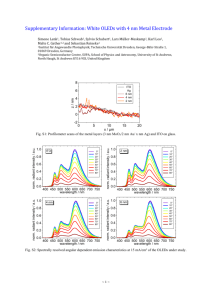
Supplementary Information Tuning nano electrical field to affect restrictive membrane area on localized single cell nano-electroporation 1.Simulation For electric field simulation, we have used comsol multiphysics software(version 3.5, Sweden) with consideration of AGS cell (human caucasian gastric adenocarcinoma cell ) membrane diameter as 10µm and other parameter such as membrane conductivity and permittivity as standard parameters [1]. 2. Fabrication To fabricate the LSCNEP chip, initially cover glass was cleaned using piranha solution and then 90nm ITO (Indium Tin Oxide) film was deposited using RF sputter and patterned it by wet chemical etching. Finally the ITO lines were formed. The X-Ray diffraction (XRD) results shows, the ITO films have proper orientation (see Fig S1), which is similar with other results [2]. The resistivity of deposited ITO film was 5 × 10-4 ohm/cm. The transparency of this ITO film was 85%-98% with visible light range (see Fig S2). After patterned ITO lines Focused Ion Beam (FIB NOVA 200, USA) technique has been used to form 500nm gap between two ITO nanoelectrodes and finally a 250nm SiO2 layer was deposited on top of the nano-electrode using Plasma Enhanced Chemical Vapor Deposition (PECVD). The final device was package with printed circuit board (PCB). Fig:S1. X-Ray diffraction of ITO films after annealing at 330 0C for 3 hours. The plane shows good uniformity of the ITO films which obey with other results (Hoshiy at al) Fig S2. Light transmission of Indium Tin Oxide (ITO) films by UV-VIS spectroscopy measurement. The light is almost 85%-98% transparent through ITO films. 3. Calculation of diffusion coefficient and deviation factor from experiment To calculate diffusion coefficient and deviation factor from experimental values, we consider differential equation for Fick’s second law of diffusion in a sphere is given by 2c 2 c c D 2 ----------------------------------------------(1) t r r r Where c is concentration at time t, D is diffusion coefficient of the material in cytoplasm and r is the distance. For a homogeneous permeable sphere of radius R, initially free of solute and placed in a well stirred solution such that C=C0 at r=R, the concentration profile in the sphere is given by 2 R 1n 1 n 2 2 Dt nr c c0 1 sin exp --------------------(2) n R R 2 n 1 r The total amount of solute that has entered into the sphere at time t is given by 8R 3c0 Q 2 1 n 2 2 Dt 2 exp ---------------------------------(3) R 2 6 n 1 n We assume the cell as perfect sphere of diameter 10 µm. The florescence intensity is simply taken to be proportional to the amount of solute that has entered into the cell. So the florescence intensity at any time t can be written as 6 F F0 1 2 n 2 2 Dt 1 exp ---------------------------------- (4) 2 R 2 n 1 n Where F0 is florescence intensity at t For localized electroporation, a very small fraction of the membrane was made permeable for diffusion by applying a voltage to the electrodes, so that a small fraction of membrane is affected. Hence the florescence intensity expression is modified as [3] 6 F F0 1 A 2 n 2 2 Dt 1 exp -------------------------------------- (5) 2 R 2 n 1 n Where A term is introduced to take into account the deviation from perfect diffusion process. It may include the deviation of the cell from proper sphere, non-radial diffusion mechanism through the localized area of the single cell etc. which are not taken into account into the theoretical calculations. The equation 5 is fitted with the normalized intensity for n from 1 to 4. Fig 5 shows the normalized intensity as a function of time elapsed after the application of external voltages and the theoretical fitted curves for different applied voltages. The diffusion coefficient estimated from the fitting parameters data are listed in the table I in manuscript. 4. Cell culture onto the chip To culture AGS cell line, 10 ml phosphate buffer saline (PBS) was added to clean the cell surface properly. Then 1 ml trypsin (0.05% trypsin-EDTA, GIBCO) was added into the disc to detach the cells from the disc surface. Finally 9 ml DMEM (Dulbecco's Modified Eagle's Medium) medium was added on to the disc for suspension of AGS cells. Then cells were added into the UV treated chip surface with concentration 2.2 × 105 cells/ml. After that the chip was incubated (5% CO2, 37 0C) for 6-8 hours to increase adhesion between cells and chip surface. Fig:S3. AGS cancer cell distribution throughout the chip surface after 6-8 hours incubation References: [1] H.Ye, M.Cotic, E.E.Kang, M.G.Fehlings, P.L.Carlen, Journal of Neuro Engineering and Rehabilitation. 7,12( 2010). [2] Y.Hoshiy, R.Ohki, Electrochimica Acta. 44, 3927 3932 (1999). [3] Y.Qiu, Y.Chen, Geoff.G.Z.Zhang, L.Liu, W.R.Porler, Developing solid oral dosage forms: Pharmaceutical theory and practice (Elsevier, MA01803, USA, 2009), P.152.