Essential Knowledge: Ch 9 Cellular Respiration - Course
advertisement

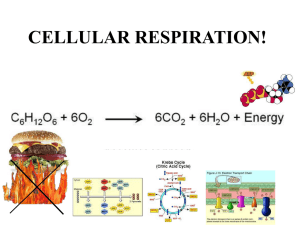
AP Biology R-11 Essential Knowledge: Ch 9 Cellular Respiration 9.1 Catabolic pathways yield energy by oxidizing organic fuels To keep working, a cell must regenerate its supply of ATP Organic Compounds + Oxygen → Carbon Dioxide + Water + Energy C6H12O6 + 6O2 → 6CO2 + 6H2O + ATP Redox reactions involve the transfer of electrons The substance losing electrons is oxidized, the substance receiving electrons is reduced (LEO-GER or OIL RIG) The oxidizing agent is reduced; the reducing agent is oxidized During cellular respiration, glucose is oxidized to carbon dioxide, and oxygen is reduced to water Energy is released as electrons are transferred from organic compounds to oxygen (reduction releases energy); the energy is used to make ATP Oxygen is one of the strongest oxidizing agents because it is so electronegative Although the respiration equation includes glucose, all carbohydrates and fats are excellent fuel sources because of their many electrons associated with hydrogen Electrons from organic compounds are first transferred to an electron carrier, NAD+ When NAD+ is reduced, it becomes NADH Hydrogen ions (protons) are transferred along with electrons During cellular respiration, most electrons travel a “downhill” route: Glucose → NADH → ETC →Oxygen Cellular respiration consists of three stages: 1. Glycolysis (cytosol) 2. Citric Acid Cycle (mitochondria) 3. Oxidative Phosphorylation (mitochondria) Substrate level phosphorylation uses something other than Pi to phosphorylate ADP to form ATP Glycolysis and fermentation are examples of substrate level phosphorylation 9.2 Glycolysis harvests chemical energy by oxidizing glucose to pyruvate Glycolysis splits one molecule of glucose into two molecules of pyruvate; most of the energy of the glucose molecule remains stored in pyruvate Two ATP are invested and four ATP are produced which yields a net gain of two ATP The products of glycolysis are 2 pyruvate, 2 H2O, 2 ATP, 2 NADH, and 2 H+ Glycolysis occurs whether oxygen is present or not 9.3 The citric acid cycle completes the energy-yielding oxidation of organic molecules Pyruvate from glycolysis enters the mitochondria if molecular oxygen is present Pyruvate cannot enter the citric acid cycle; it must first be converted to acetyl-CoA Three reactions complete the conversion of pyruvate to acetyl-CoA 1. Pyruvate’s carboxyl group is removed and released as a molecule of carbon dioxide 2. The remaining two carbon fragment is oxidized to form acetate and electrons (and protons) are added to NAD+ to form NADH 3. Coenzyme A is attached to acetate FAD is a lower energy electron carrier than NAD+, it is reduced to form FADH2 The citric acid cycle (Kreb’s cycle) spins one time for each pyruvate, so twice for each glucose The products of the citric acid cycle (per pyruvate) are 1 ATP, 2 CO2, 2 FADH2, 3 NADH, and 3 H+ The products of the citric acid cycle (per glucose) are 2 ATP, 4 CO2, 4 FADH2, 6 NADH, and 6 H+ All of the carbon molecules originally found in glucose are oxidized and released as carbon dioxide 9.4 During oxidative phosphorylation, chemiosmosis couples electron transport to ATP synthesis Oxidative phosphorylation has two components: the electron transport chain and chemiosmosis (chemiosmotic phosphorylation) At this point in the cycle, glycolysis has produced 2 ATP and the citric acid cycle has produced 2 ATP; most of the energy extracted from glucose is found in NADH and FADH2 The electron transport chain (ETC) is a collection of molecules (mostly proteins) embedded in the inner membrane of the mitochondria NADH carries its electrons and associated protons to the top of the ETC. FADH2 enters at a lower energy level. As electrons are handed down the chain, the carriers alternate between being reduced and oxidized as they accept and donate electrons Each time electrons are handed off, a small amount of energy is released; this energy is used to pump H+ across the inner mitochondrial membrane into the intermembrane space The purpose of the ETC is to establish a proton gradient in the intermembrane space of the mitochondria Molecular oxygen is the terminal electron acceptor at the bottom of the ETC; when oxygen accepts the low energy electrons, it also combines with hydrogen ions to form water As H+ build up in the intermembrane space, a proton gradient is established; this proton-motive force has the ability to do work Chemiosmosis uses the energy stored in the H+ gradient to do cellular work ATP synthase is an enzyme embedded in the intermitochondrial membrane; they provide the only sites for H+ to reenter the mitochondrial matrix through the membrane ATP synthase is built like a rotary motor; the diffusion of H+ down its concentration gradient through the rotor generates the energy to phosphorylate ADP to form ATP Oxidative phosphorylation generates 32-34 ATP per glucose One glucose generates 36-38 ATP ( 2 glycolysis + 2 citric acid cycle + 32-34 oxidative phosphorylation) 9.5 Fermentation and anaerobic respiration enable cells to produce ATP without the use of oxygen At the end of glycolysis, if molecular oxygen is available then pyruvate enters the mitochondria and ATP is produced through the aerobic processes of the citric acid cycle and oxidative phosphorylation If oxygen is not available, pyruvate remains in the cytosol and fermentation generates ATP by substrate level phosphorylation NAD+ is the terminal electron acceptor in fermentation There are two kinds of fermentation: alcoholic and lactic acid In alcoholic fermentation, pyruvate is oxidized to ethanol and carbon dioxide; beer, wine, and bread are produced by alcoholic fermentation In lactic acid fermentation, pyruvate is oxidized to form lactate and no CO2 is produced; cheese and yogurt are produced by lactic acid fermentation Human muscle cells can make ATP by lactic acid fermentation when oxygen is scarce 9.6 Gylcolysis and the citric acid cycle connect to many other metabolic pathways Catabolic pathways funnel electrons from many kinds of organic molecules into cellular respiration to produce ATP for cell processes Anabolic pathways use ATP to build the organic molecules needed by the organism (biosynthesis) Feedback mechanisms control the rate of cellular respirations; enzymes at key points in glycolysis and the citric acid cycle increase or decrease the amount of ATP produced as needed