WEEK 5 Feb. 4

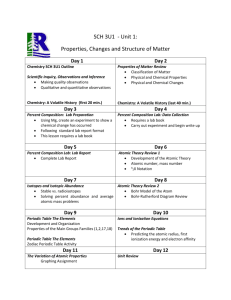
Honors Chemistry Week-At-A-Glance
Goal: SC3 Atomic Structure https://sites.google.com/site/harrisonhighchemistrytarvin/
Monday
Essential Question: What’s wrong with Jimmy Neutron’s model of the atom?
Standard: SC3b: Use the orbital configuration of neutral atoms to explain its effect on the atom’s chemical properties. SC3f:
Relate light emissions and movement of electrons to the identification of the element.
Objectives:
Understand the arrangement of electrons in the atom (aufbau, Pauli, and Hund’s rules)
Describe the quantum mechanical model of the atom
Tuesday
Essential Question: What’s wrong with Jimmy Neutron’s model of the atom?
Standard: SC3b: Use the orbital configuration of neutral atoms to explain its effect on the atom’s chemical properties. SC3f:
Relate light emissions and movement of electrons to the identification of the element.
Objectives:
Illustrate the orbital configuration of electrons in atoms
Stability & exceptions to aufbau principle
Wednesday
Essential Question: How is the periodic table organized?
Standard: SC4b: Compare and contrast the trends in the chemical and physical properties of elements and their placement on the
Periodic Table.
Objectives:
Identify the locations and properties of assigned families of elements
Apply concepts from last unit to periodic table
Thursday
Essential Question: How is the periodic table organized?
Standard: SC4b: Compare and contrast the trends in the chemical and physical properties of elements and their placement on the
Periodic Table.
SC4a: Use the Periodic Table to predict periodic trends including atomic radii, ionic radii, ionization energy, and electronegativity of various elements.
Objectives:
Investigate properties of ionization energy, electronegativity, and atomic radius
Unit Two Lab
Practical
Unit Two
Cumulative
Test
Visual/Kinesthetic Activity:
Color-code and label a periodic table to identify families of elements
Visual/Auditory Activity:
Discuss physical and chemical properties of elements based on periodic table location
[Alkali metals, alkaline earth metals, transition metals, inner transition metals, halogens, noble gases, metals, nonmetals, metalloids, diatomic elements]
WEEK 5 Feb. 4 - 8
Self-Assessment Activity:
Reflect on Unit Two Test and
Lab Practical
Collaborative Activity: Chalk
Talk – Classify elements by family, chemical and physical properties
Visual/Auditory Activity:
Learn the trends of atomic radius and ionic radius
Friday
Essential Question: How is the periodic table organized?
Standard: SC4b: Compare and contrast the trends in the chemical and physical properties of elements and their placement on the
Periodic Table.
SC4a: Use the Periodic Table to predict periodic trends including atomic radii, ionic radii, ionization energy, and electronegativity of various elements.
Objectives:
Identify the locations and properties of assigned families of elements
Investigate properties of ionization energy, electronegativity, and atomic radius
Apply concepts from last unit to periodic table
Formative Assessment:
Placement and Properties of
Elements Quiz
Visual/Auditory Activity:
Learn the trends of ionization energy and electronegativity
Collaborative Activity: Chalk
Talk – Predict properties and arrange elements in order of increasing or decreasing atomic radius, ionization energy, and electronegativity
Honors Chemistry Week-At-A-Glance
WEEK 6 Feb. 11-14
Goal: SC4 Periodicity https://sites.google.com/site/harrisonhighchemistrytarvin/
Monday
Essential Question: How is the periodic table organized?
Standard: SC4b: Compare and contrast the trends in the chemical and physical properties of elements and their placement on the
Periodic Table.
SC4a: Use the Periodic Table to predict periodic trends including atomic radii, ionic radii, ionization energy, and electronegativity of various elements.
Objectives:
Identify the locations and properties of assigned families of elements
Investigate properties of ionization energy, electronegativity, and atomic radius
Apply concepts from last unit to periodic table
Formative Assessment:
Periodic Trends Quiz
Kinesthetic Activity:
Reactivity of the Halogen
Family Lab
Tuesday
Essential Question: How is the periodic table organized?
Standard: SC4b: Compare and contrast the trends in the chemical and physical properties of elements and their placement on the
Periodic Table.
SC4a: Use the Periodic Table to predict periodic trends including atomic radii, ionic radii, ionization energy, and electronegativity of various elements.
Objectives:
Identify the locations and properties of assigned families of elements
Investigate properties of ionization energy, electronegativity, and atomic radius
Apply concepts from last unit to periodic table
Self-Assessment: Review
Periodic Trends Quiz results
Collaborative Activity:
Answer sample short answer test questions and score answers with rubric
Wednesday
Essential Question: How is the periodic table organized?
Standard: SC4b: Compare and contrast the trends in the chemical and physical properties of elements and their placement on the
Periodic Table.
SC4a: Use the Periodic Table to predict periodic trends including atomic radii, ionic radii, ionization energy, and electronegativity of various elements.
Objectives:
Identify the locations and properties of assigned families of elements
Investigate properties of ionization energy, electronegativity, and atomic radius
Apply concepts from last unit to periodic table
UNIT THREE LAB
PRACTICAL
&
CUMULATIVE QUIZ
Thursday
Essential Question: How is the periodic table organized?
Standard: SC4b: Compare and contrast the trends in the chemical and physical properties of elements and their placement on the
Periodic Table.
SC4a: Use the Periodic Table to predict periodic trends including atomic radii, ionic radii, ionization energy, and electronegativity of various elements.
Objectives:
Identify the locations and properties of assigned families of elements
Investigate properties of ionization energy, electronegativity, and atomic radius
Apply concepts from last unit to periodic table
UNIT THREE TEST
Friday