Study Guide for Test
advertisement

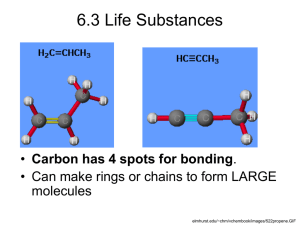
Study Guide for Test Name Molecules of Life The test on Molecules of Life is closed note, open brain . To prepare for this test, review the labs and activities, readings, notes, and returned work/comments. Terms to understand, identify in an image, and be able to apply to a variety of problems. Molecule Molecular formula Atom Bond Double bond Atomic Nucleus Proton Electron Monomer Polymer Carbohydrates Monosaccharide Disaccharide Polysaccharide Glycosidic Bond Starch Glycogen Cellulose Chitin Lipid Saturated Fat or Fatty Acid Unsaturated Fat or Fatty Acid Glycerol Fatty Acids (and examples) Ester Bond Hydrogenation Protein Polypeptide Amino Acids R-group (variable) Peptide Bond Primary Structure Secondary Structure Tertiary Structure Quaternary Structure Enzyme Substrate Active Site Hydrolysis Dehydration Synthesized Review Tasks: COMPLETE each task in your journal (or simply do the task) and check each box when you have completed the task. 1. Review/re-read journal entries pertinent to the molecules of life unit. These include all handouts and readings. Practice identifying different types of molecules (diagrams) from the lecture, worksheets, etc. Get yourself quizzed! Compare and contrast the various types of molecules, their functions, and their structures. Be able to infer what could happen to the cell and ultimately the organism if various molecules were in excess or absent. Be able to analyze data in a table and/or a graph that may pertain to experiment involving molecules. Connect at least 3 words (above) together into one sentence that explains something having to do with molecule structure, function, or interaction with the body. Examples: peptide bond, polysaccharide, cellulose. Try it! It’s hard, but doable! Revisit your quiz! Make sure to address all questions you missed. Why were wrong answers wrong and right answers right? Go through each possible answer and explain to a friend why ^^. TRY THIS! I highly recommend doing this! Visit the calendar/website and go through the review Jeopardy! when it is posted (usually the night before the test). Re-read a chapter in your summer reading book so you can discuss the molecules of life connection. Compare & contrast the following characteristics of lipids, proteins, and carbohydrates using a triple Venn Diagram like this – Molecular Structure – Functions – Monomers and Polymers – Sources (food) – Processes used to create monomers/polymers Need more space? Do it on a separate sheet. #sorrynotsorry 2. Explain and draw dehydration synthesis. 3. Explain and draw hydrolysis. 4. Write the molecular formula for a glycogen molecule consisting of 22 glucose molecules, and explain how you know. 5. Explain the 4 levels of protein structure and how they contribute to the overall 3D structure of the protein molecule. Here is a diagram that may be helpful to you. 6. Explain how proteins can be beneficial to living things using all of the following terms and underlining them when used: protein, enzyme, substrate, energy, structure, tertiary 7. What molecules or structures can be represented by each letter? If A is lactose, what are the rest? Label on the diagram or identify the: - Enzyme - Substrate - Products - Glycosidic bond (where it is) - Dehydration (where it happened) - Hydrolysis (where it happened) Give an example enzyme name and its associated substrate, and its products. Molecules of Life Review Carbohydrates Type of monomer Monomer examples Lipids Proteins Monosaccharides butyric acid, lauric acid Process to convert monomers into polymers Type of Polymer Polymer examples Process to convert polymers into monomers Type of bond formed between monomers Functions in the bodies of living organisms Rule to identify molecule by structure Sources (food) Where can we obtain this molecule? Image or rough sketch Saturated fat, steroid, wax Hemoglobin, insulin, amylase The Molecules of Life Crossword Puzzle Clues Across 6. the level of protein structure caused by repeated folding of the polypeptide backbone. The 2 types are the alpha helices and beta pleated sheets. 8. the level of protein structure determined by a unique sequence of amino acids 10. A sub-unit (single unit) of a polymer 13. lipids that exist as a liquid at room temp and has 1 or more double bond (C=C) 15. most common monosaccharide 16. the type of chemical bond that holds together amino acids. 17. lipids that serve as coatings for plant parts and as animal coverings to help prevent desiccation or drying out 19. a chemical reaction resulting in the breakdown of a polymer using water 20. a lipid that is composed of 1 glycerol + 2 fatty acids + phosphate group; also a component of the cell membrane 22. the type of chemical bond that holds together lipid monomers. Down 1. the monomers of protein. A molecule made of an asymmetric carbon covalently bonded to hydrogen, amino group, carboxyl, and R group 2. the type of chemical bond that holds together carbohydrate monomers 3. a relatively small carbohydrate that consists of 2 monosaccharides bonded together 4. the simplest form of a carbohydrate. Glucose is a good example of one. 5. a macromolecule that is involved with or is necessary for life. 7. A chemical reaction resulting in a bond and is created through the loss of water 9. the level of protein structure caused by the irregular folding of a polypeptide due to bonding between R groups. 11. the level of protein structure resulting from interactions among 2 or more polypeptides. 12. lipids composed of 4 carbon rings with no fatty acid tails. These act as components of membranes and for hormones. An example of one of them is cholesterol. 14. a chain of amino acids bonded together before the folding of the protein occurs. 18. a large molecule composed of monomers 20. a large carbohydrate; hundreds to thousands of monosaccharides bonded together 21. lipids that exist as a solid at room temp and has only single bonds (C-C) 23. a lipid that is composed of 1 glycerol + 3 fatty acids