Properties of Gases
advertisement

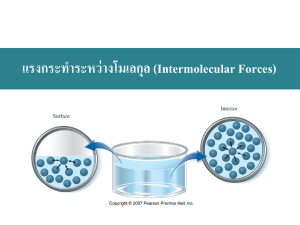
Liquids and Solids H/Chemistry/1213 Liquids and Solids Forces of Attraction Forces within Compounds Intermolecular forces (Forces between covalently-bonded compounds) Between molecules Dispersion (London) forces Dipole-dipole forces Hydrogen bonds Relatively weak compared to intramolecular forces Chemical Bonds: Ionic Metallic Covalent (intramolecular force) Intermolecular forces (van der Waals forces) 1. Dispersion or “London” forces (nonpolar molecules) (named after Fritz London, 1900-1954) weakest intermolecular force, present between all molecules results from an induced “temporary dipole” which induces a dipole in a nearby molecule. The constant motion of electrons uneven distribution of electrons at any particular moment: acts on all molecules all the time only intermolecular force acting among noble gas atoms and nonpolar molecules larger number of electrons larger temporary dipole stronger attractions between molecules higher m.p. and b.p. e.g. halogens F2, Cl2 gases at room T Br2 liquid at room T (larger than F2 and Cl2) I2 solid at room T (largest) -1- Liquids and Solids H/Chemistry/1213 2. 3. Dipole force (polar molecules) attractions between polar molecules, stronger than London dispersion forces (-) end of one polar molecule attracts the (+) end of another polar molecule more polar stronger dipole force closer together stronger dipole force Hydrogen bonding always involves H attached to an O, F or N (small, high electronegativity) strongest intermolecular force How strong? 5% of the strength of a covalent bond higher b.p. and higher viscosity accounts for high b.p. of H2O -2- Liquids and Solids H/Chemistry/1213 Liquids and Solids How are liquids and solids similar to and different from gases? (in terms of the KMT) Property Gases Liquids Solids Spacing Movement Average KE Attraction between Particles Disorder/order Volume Shape Fluidity Density Compressibility Diffusing ability -3- Liquids and Solids H/Chemistry/1213 Liquids Viscosity = a measure of the resistance to flow In liquids, viscosity is determined by the intermolecular forces – more attractive viscosity shape of the particles – longer chains viscosity temperature – colder viscosity Surface tension - result of attractive forces of particles in the liquid = E required to the SA of a liquid by a given amount intermolecular forces surface tension Surfactants = compounds that lower the surface tension of water (e.g. detergent or soap) Capillary action – the result of cohesion and adhesion Cohesion = force of attraction between identical molecules Adhesion = force of attraction between different types of molecules e.g. Water in capillary tube – adhesion between water molecules and glass > cohesion between water molecules Solids Density of solids is higher than density of liquids, and may be crystalline or amorphous Crystalline solids made of crystals – particles are arranged in an orderly, geometric, repeating pattern have definite geometric shape crystal lattice = total 3-D array of points that describe the arrangements of particles, smallest unit of which is the unit cell crystal has the same symmetry as its unit cell abrupt melting points – all bonds break at once -4- Liquids and Solids H/Chemistry/1213 7 shapes, based on arrangement of atoms in unit cell, cell lengths and cell angles (see textbook for details if you are particularly interested) Categories of Crystalline Solids 1. 2. 3. 4. 5. atomic (e.g. noble gases) molecular (e.g. table sugar) covalent network (e.g. diamond, quartz) ionic metallic Binding forces in crystals 1. Atomic and 2. Covalent molecular crystals Weak intermolecular forces low m.p., easily vaporized, relatively soft, good insulators 3. Covalent network (e.g. diamond, graphite) 3-D covalent bonds (giant covalent molecules) very hard, brittle, high m.p., nonconductors or semiconductors; some are planar, e.g. graphite – in sheets 4. Ionic (e.g. NaCl) strong positive and negative ions, electrostatically attracted to one another hard, brittle, high m.p., good insulators 5. Metallic (metals) positive ions surrounded by a cloud of electrons (electrons can move freely through lattice) high electrical conductivity, malleability, ductility Amorphous solids e.g. glasses and plastics – particles are arranged randomly nearly any shape, depending on molding no definite melting point, gradually soften to thick, sticky liquids cool too fast for crystals to form -5- Liquids and Solids H/Chemistry/1213 Phase Changes Process Phases involved Endothermic or Exothermic? melting solid liquid -6- endothermic Liquids and Solids H/Chemistry/1213 Some Phase Change Terminology Melting point (m.p.) = T at which the forces holding the crystal lattice of a crystalline solid together are broken and it becomes a liquid Note: Amorphous solids act somewhat like liquids even when solid Vapor Pressure (VP)= P exerted by a vapor over a liquid Boiling point (b.p.)= T at which the VP of a liquid = atmospheric P Evaporation = vaporization only at the surface of liquid, below the b.p. Freezing point = T at which a liquid is converted into a crystalline solid Phase Diagrams Graph showing the relationships between solid, liquid and gaseous phases over a range of conditions, e.g. P vs. T Triple point = T and P conditions at which the solid, liquid and vapor of a substance can coexist at equilibrium Critical T = T above which the substance cannot exist in the liquid state Critical P = P at which a gas at its critical T is converted to a liquid Refer to diagram on next page for the following questions 1. What variables are plotted on a phase diagram? __________________________ 2. How many phases of water are represented in its phase diagram? _____ What are they? ___________________________ 3. What phases of water coexist at each point along the red curve? _________________ Along the yellow curve? _________________________ 4. Look at the phase diagram for carbon dioxide. Above which pressure and temperature is carbon dioxide unable to exist as a liquid? _______________ 5. At which pressure and temperature do the solid, liquid, and gaseous phases of carbon dioxide coexist? ____________________________ -7- Liquids and Solids H/Chemistry/1213 6. Use the phase diagram for water to complete the following table. Temperature (oC) Pressure (atm) 200 1 -2 1 150 100 -2 0.001 30 0.8 1 100.00 Phase liquid vapor -8- Liquids and Solids H/Chemistry/1213 Gas vs. Vapor Gas = state of particles at room temperature Vapor = a gas formed from a substance that normally exists as a solid or liquid at room T and P Vaporization = conversion of a liquid to gas or vapor Boiling point = T at which vapor pressure of liquid = atmospheric pressure Boiling point at 1 atm = “normal boiling point” H2O enters vapor state within liquid. Vapor is less dense, so rises to surface Needs constant energy (heat) to keep it boiling - cooling process Liquid never rises above its boiling point! (at constant P) If atm P , less energy is required for particles to escape atm P Mountains vs. pressure cooker Evaporation Evaporation occurs when particles have enough KE to overcome their I.M. forces In a contained vessel (closed): “dynamic equilibrium” occurs when rate of vaporization = rate of condensation vapor pressure = partial pressure of a vapor above a liquid depends on: 1. # gas particles: # particles vapor pressure 2. Temperature: as T v.p. because evaporation (particles have more energy to escape) 3. Intermolecular forces: the stronger the I.M. forces v.p. (fewer particles have enough energy to break the I.M. bonds and escape) -9- Liquids and Solids H/Chemistry/1213 In an uncontained vessel (open): evaporation as a cooling process: the most energetic particles leave so average KE of remaining particles is lower Rate of evaporation increases with increase in: 1. T (more particles have energy to escape the liquid) 2. air currents 3. surface area Evaporation vs. Boiling takes place at surface occurs throughout liquid below boiling T at boiling T Vapor Pressure 1. What is the VP of ethanol at 60oC? ________________________ 2. What is the VP of water at the same T (60oC)? ____________________ 3. Which compound boils at a lower T? How can you tell? ____________________ _________________________________________________________________ 4. Which exhibits stronger IMFs? Ethanol or water? Explain your answer. _________________________________________________________________ _________________________________________________________________ - 10 -