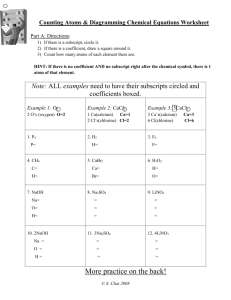
Balancing Chemical Equations Worksheet
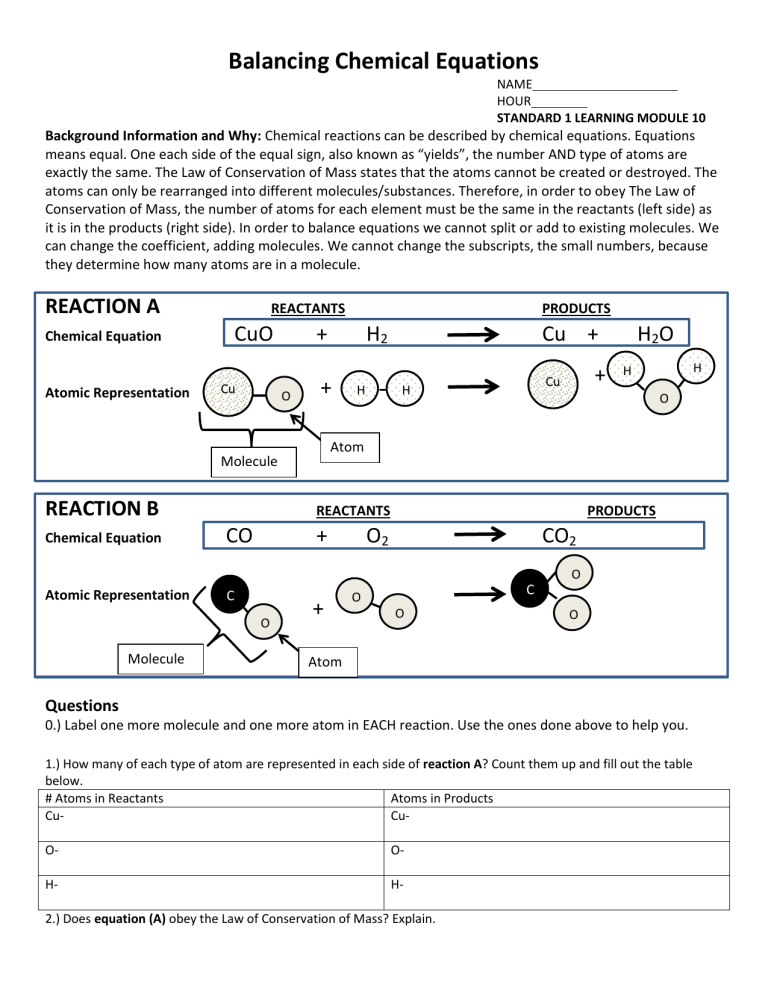
Balancing Chemical Equations
NAME
HOUR
STANDARD 1 LEARNING MODULE 10
Background Information and Why: Chemical reactions can be described by chemical equations. Equations means equal. One each side of the equal sign, also known as “yields”, the number AND type of atoms are exactly the same. The Law of Conservation of Mass states that the atoms cannot be created or destroyed. The atoms can only be rearranged into different molecules/substances. Therefore, in order to obey The Law of
Conservation of Mass, the number of atoms for each element must be the same in the reactants (left side) as it is in the products (right side). In order to balance equations we cannot split or add to existing molecules. We can change the coefficient, adding molecules. We cannot change the subscripts, the small numbers, because they determine how many atoms are in a molecule.
REACTION A
REACTANTS PRODUCTS
Chemical Equation
Atomic Representation
Cu
CuO + H
Molecule
O
+
H
Atom
2
H
Cu + H
Cu
+ H
2
O
O
REACTION B
REACTANTS PRODUCTS
Chemical Equation
CO + O
2
CO
2
Atomic Representation
Molecule
C h h
O
+
O
O
C h h
O
O s s
Atom s s s s
Questions
0.) Label one more molecule and one more atom in EACH reaction. Use the ones done above to help you.
1.) How many of each type of atom are represented in each side of reaction A? Count them up and fill out the table below.
# Atoms in Reactants Atoms in Products
Cu-
O-
Cu-
O-
HH-
2.) Does equation (A) obey the Law of Conservation of Mass? Explain.
H
3.) How many of each type of atom are represented in each side of reaction B? Count them up and fill out the table below.
# Atoms in Reactants Atoms in Products
C-
O-
C-
O-
4.) Does equation (B) obey the Law of Conservation of Mass? Explain.
5.) If either reaction/equation A or B does not obey the Law of Conservation of Mass, draw additional molecules on the reactant and/or product side(s) in order to make the number of all atoms equal on both sides of the equation. HINT: recall you cannot split existing molecules or add atoms to existing molecules. In other words EXISTING MOLECULES
MUST REMAIN INTACT AND YOU CAN ONLY USE REPLICAS OF WHAT YOU STARTED WITH.???????????????
6.) In reaction (A) how many H
2
O molecules are produced for every H
2
molecule that is needed in the reactants?
7.) In reaction (B) how many CO
2
molecules are produced for every O
2
molecule that is needed in the reactants?
8.) In reaction (B) how many CO molecules react with every O
2
molecule?
9.) In order to answer question 15 you should see that reaction (a) is already drawn correctly, but reaction B does not have the same number of oxygen atoms on each side. It should be drawn like this….. GO BACK AND CHECK YOURS.
REACTANTS PRODUCTS
C
C
+
C
C
O
O
O
O
O
O
We needed to add this carbon dioxide(CO
2
) because we
O O already had carbon dioxide. A total of 2 carbon
We needed to add this carbon monoxide (CO) because we dioxide. already had carbon monoxide. A total of 2 molecules
10.) Recall we can only change the coefficients in a chemical equation. Below are the blanks where coefficients can go.
Look at the new atomic representation for Reaction (B) above and write the correct coefficient in the correct blank.
Yes, you can
REACTANTS change these
PRODUCTS
2 CO + O
2
2 CO
2
NO, you CANNOT change these
11.) This drawing and equation now show how many oxygens in the reactants? How many oxygens in the products?
12.) This drawing and equation now show how many carbons in the reactants? How many carbons in the products?
13.) Is this equation balanced? Explain?
Strategy to Balance A Chemical Equation
Background: Now you understand that the atoms in the reactants and products have to be the same for each element.
This is because of the Law of Conservation of Mass. You should also recall that we cannot split any given atoms or add any new ones to those already given. We do this by changing ONLY the coefficients and NEVER the subscripts. We will go through an example that will show you how to make a prediction, change a coefficient, recount the number of atoms, update a table, and determine next step.
Reaction (C)
Na + O
2
Na
2
O
2
Step 1: First you would draw a table and make a list of the atoms in the reactants and products, count them up and update your table. This one is done for you
Reactants
Na-1
Products
Na-2
O-1 O-2
Step 2: Do the Na’s (sodium) and O’s (oxygen) balance? Are they the same in as out? No!! Determine what coefficient
(only positive integers) could be put in front of atoms and molecules to get them to balance. Let’s try a 2 in front of Na.
Step 3: Write the coefficient in the correct spot and update the table. Because the Na and O are separated by a + te 2 affects only the Na. Notice the change in the table.
2 Na + O
2
Reactants
Na-
O-
Na
2
O
2
Products
Na-
O-
Step 4: Continue placing coefficients in the correct places until the equation is balanced. You may be wrong occasionally
and have to cross out and try again. Check out below. This person put a 2 for the coefficient in the product. Because
Na
2
O
2 is all one molecule the Na atoms AND the O atoms will go up. 2 times 2 s 4 for each. But this gets us further from balanced! Erase as try another.
2 Na + O
2
Reactants
Na-1 2
O-2
2 Na
2
O
2
Products
Na-4
O-4
Step 5: Continue placing coefficients in the correct places until the equation is balanced. A coefficient of 2 in front of the
Na does the trick. Update your table EVERY TIME!!!!!
This is your final answer. The
2 Na + O
2
Na
2
O
2 balanced equation. CIRCLE IT
WHEN YOU GO IT!!
Reactants
Na- 1 2
O- 2
14.) Why do we have to balance chemical equations?
Products
Na-4 2
O-4 2
15.) Balance these equations. USE EXTRA PAPER IF YOU NEED THEN DRAW THE BALANCED MODEL a.) H
2
+ O
2
H
2
O Reactants Products
H- H-
O- O- b.) H
2
O
2
H
2
O + O
2
Reactants
H-
O-
Products
H-
O- c.) N
2
+ H
2
NH
3
Reactants Products
Reactants Products
d.) Al
2
O
3
O
2
+ Al e.) Fe + H
2
O Fe
3
O
2
+ H
2 f.) Na
2
SO
4
+ CaCl
2
CaSO
4
+ NaCl
Reactants
Reactants
BALANCE THESE EQUATIONS: make your own table and start from scratch.
CO
2
+ H
2
O
2 g.) C
2
H
6
+ O
2 h.) Na + O
2
Na
2
O
2
Products
Products
EXTRA CREDIT BALANCE THESE EQUATIONS THEN DRAW THE
MODEL. GET TEACHER SIGNATURE FOR EXTRA CREDIT-USE SEPARATE PAPER IF NEEDED
EC1.) __ Ca(OH)
2
+ __ H
2
SO
4
__HOH + __ CaSO
4
EC2.) __ Cu + __ AgNO
3
__ Cu(NO
3
)
2
+ __ Ag
EC1.) __ Ca(OH)
2
+ __ H
2
SO
4
__HOH + __ CaSO
4
EC2.) __ Cu + __ AgNO
3
__ Cu(NO
3
)
2
+ __ Ag