Formulas and Equations
advertisement

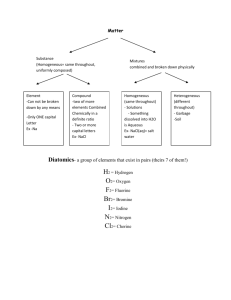
Topic 2 Formulas and Equations Chemical Symbols and Formulas Each elements has been assigned a unique 1, 2, or 3-letter Symbol for its identification, and these name/symbols are given by the International Union of Pure and Applied Chemists (IUPAC) Formulas: show both symbol and numbers of the atoms present Qualitative: show the symbol of the element Quantitative: show numbers of the element Coefficient: tells how many units of the formula are present, and it applies to th entire formula Monatomic:1 atom Diatomic: 2 atoms Polyatomic:multiple atoms Ex: H2O = 2 hydrogen atoms, 1 oxygen atom (read the subscripts) Ex: CO = 1 carbon atom, 1 oxygen atom (no subscript means 1 atom) Ex: 3 H2O = 6 hydrogen atoms, 3 oxygen atom (subscript x coefficient) Ex: 3 Mg (C2H3O2)2 Atoms: = 3 Mg, 12 C, 18 H, 12 O (subscript x parenthesis x coefficient) -neutral charge, monatomic element or diatomic element Ex: Au, O2 Compound:-neutral charge, 2 or more elements bonded together Ex: H2O, NaCl, (NH4)3PO4 Ions: -either positive (lose electron) or negative (gain electron) charged Ex: Na+, NO32- Writing Formulas and Naming Compound (use oxidation number, (charges)) Ionic compound: metal (positive charge) + nonmetal (negative charge) (Binary compound: change ending to –ide) Ex: NaCl Sodium Chloride MgS Magnesium Sulfide CaBr2 Calcium Bromide K2S With polyatomic ions (Charges), use Table E Ex: KOH Potassium Hydroxide NH4Cl NaCN Sodium cyanide SrClO4 Potassium Sulfide Ammonium Chloride Strotium Perchlorate Covalent / molecular compound: nonmetal + nonmetal (No Charges) Use prefixes (1=mon, 2=di, 3=tri, 4=tetr, 5=pent, 6=hex, 7=hept, 8=oct, 9=non, 10=dec) *note: If only one atom of the first element is present, the prefix mono is not used. Ex: CO Carbon monoxide N2O5 Dinitrogent pentoxide The Stock System (Transition metals): have more than one oxidation number Use Roman numerals (1=I, 2=II, 3=III, 4=IV, 5=V, 6=VI, 7=VII) Ex: FeCl2 Iron (II) Chloride Fe+2 Cl-1 FeCl3 Iron (III) Chloride Fe+3 Cl-1 Polyatomic ions: [Table E] Hydrates: compound with water Anhydrates: compound without water Ex: Al(ClO3)3 ● 6 H2O Ex: Al(ClO3)3 Physical changes: anything that you can measure, such as phase change = melting or boiling of water, sizes, temperatures, textures, etc. Chemical changes: forming a new substance, such as burning a piece of paper or gasoline, reacting with other chemical Endothermic Processes [Table I]: processes that require energy, ∆H is positive, energy is on the left side of the equation. Exothermic Processes [Table I]: processes that release thermal energy, ∆H is negative, energy is on the right side of the equation. Type of reaction: 1. Synthesis (combining together) A + B → AB Ex: H2(g) + O2(g) → 2H2O(g) 2. Decomposition (breaking down) AB → A + B Ex: H2O(l) → H2(g) + O2(g) 3. Single Replacement A + BC → AB + C (One element and one compound on each side) Ex: Zn + Cu(NO3)2 →Cu + Zn (NO3)2 4. Double Replacement AB + CD → AD + BC (Two different compound on each side) Ex:AgNO3(aq) + NaCl(aq) → AgCl(s) + NaNO3(aq) Balancing Chemical Equations: left side of the equation is equal to the right side of the equation Balanced equation Ex: C + O2 → CO2 1C, 2O = 1C, 2O Unbalanced equation H2 + O2 → H2O 2H, 2O =2H, 1O