File
advertisement

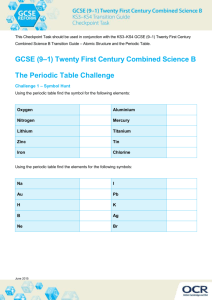
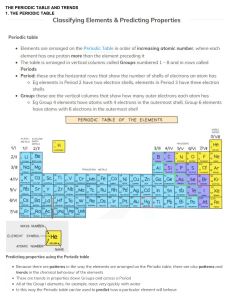
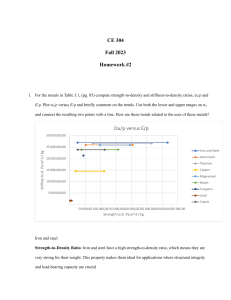
Extracting metals Alkaline Earth metals Know that Group 2 are the Alkaline Earth metals Know the electron configurations for Be, Mg and Ca Explain the trends in atomic radius, ionisation energy and reactivity down Group 2, related to shielding, atomic radius and nuclear charge Describe the trend in melting points down Group 2 (CARE: Magnesium) Write equations for the reactions of Group 2 elements with water to form a hydroxide State the trend in vigour of reaction of Group 2 with water State solubility trends for Group 2 hydroxides and sulfates Explain the trend in pH of the solution produced by a Group 2 hydroxide linked to solubility Describe a test for sulphate ions linked to solubility Identify what has been oxidised and reduced in the above reactions Discuss some uses of Group 2 compounds Understand the term ‘ore’ and relate the method of metal extraction to it’s reactivity Describe how to extract a metal from a sulphide ore Discuss the pro’s and con’s of oxidising sulfides Write equations for the reduction of iron (III) oxide and manganese (IV) oxide with carbon and carbon monoxide Write equations for the reduction of copper carbonate and copper oxide with carbon, and the thermal decomposition of copper carbonate Write an equation for reducing Tungsten oxide with hydrogen Discuss why carbon is not used to extract tungsten and some issues surrounding the use of hydrogen in this process Describe how aluminium is extracted in an electrolysis pot Discuss the economics of aluminium extraction linked to electricity, cryolite and graphite anodes Write symbol, ionic and half-equations for aluminium oxide electrolysis Discuss the properties of titanium Describe the three stages of extracting titanium from titanium (IV) oxide Write equations for the three stages discussed above Discuss the advantages and disadvantages of recycling metals Explain the role of scrap ion in copper extraction from low grade ores Exam ques Revised ): S: (: GROUP 2 AND METAL EXTRACTION Covered AS Chemistry – Unit 2