Chemistry Exercise
advertisement

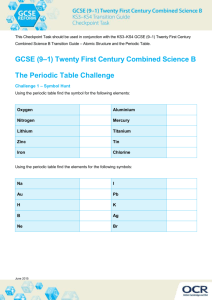
Form 4 ( ) 1. Complete the table Name of the compound T.W.G..Hs. Chen Zao Men College Chemistry Exercise Name : ________________( Formula Name of compound ) formula Sodium bromide K2O Calcium nitrate FeCl3 Zinc chloride FeSO4 Copper (II) hydroxide PbCl2 Magnesium oxide Mg(NO3)2 2. Chlorine is an element of group VII , third period. The natural abundance of chlorine contains 30% chlorine-37 and 70 % chlorine-35. a) b) c) d) Write the atomic symbol for Chlorine-37 . How many neutrons are there in Chlorine-37 What is the relationship between Chlorine-37 and chlorine-35 ? Calculate the atomic mass of chlorine . 3) X, Y and Z are three elements with atomic number 11, 16, and 17 respectively. Using dots and crosses to represent electrons, show how the following compounds are any 2 elements a) Ionic bond b) covalent bond c) How do you test that the compound formed is an ionic compound ? 2. State the observations and write equations for all chemical reaction that have occurred. a). Adding a piece of magnesium to copper(II) sulphate b) Adding a piece of calcium into a test tube containing water c) 4ex_metals.doc Page 1 formed by c) A piece of zinc is added to hydrochloric acid d) copper(II) oxide is heated strongly with carbon . e) lead(II) oxide heated with carbon is 3. Draw a labelled diagram to show how you obtain test tubes of hydrogen gas by reacting steam and iron filings. a) Write equation for the formation of hydrogen gas. b) How do you test the gas. 5) X ,Y and Z are three metals . The oxide of Y can be reduced by using carbon but Z cannot . Z is displaced from its solution by X.. a) Which is the most reactive metal ? b) Arrange the metals in the descending order of reactivity. c) 6) State 4 physical properties of metals ? a) b) c) d) 4ex_metals.doc Page 2