FLORIDA INTERNATIONAL UNIVERSITY
advertisement

CHM 3410 – Problem Set 11
Due date: NONE. The final exam is Monday, December 5 th, from noon to 2pm. It is comprehensive. You may
bring your book, all of the class handouts, a calculator, and one 8.5 x 11 inch sheet of paper (a formula page) to use
on the exam.
Do all of the following problems. Show your work.
“Sometimes men come by the name of genius in the same way that certain bugs come by the name of centipede; not
because they have a hundred feet, but because most people cannot count above fourteen.” G. C. Lichtenberg
1) Consider a system consisting of 20 diatomic molecules with a total of 4 “packets” of energy. Find all of the
possible distributions of energy packets among these 20 molecules, and the weight of each distribution.
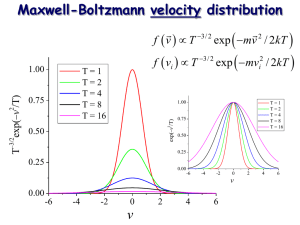
2) The Boltzmann distribution can be used as the starting point for a simple derivation of the Maxwell-Boltzmann
distribution of molecular speeds in an ideal gas. Starting with the relationship
p(|v|) ~ v2 exp(-/kt) = v2 exp(-mv2/2kT)
(2.1)
and the normalization condition
0 p(|v|) dv = 1
(2.2)
derive the Maxwell-Boltzmann distribution. (Note that v2 is proportional to the degeneracy of a translational state
with energy , and that = mv2/2).
3) The internal energy (relative to the value at T = 0) and heat capacity of a system are given by the expressions
U(T) – U(0) = (NkT2/q) (q/T)V = (nRT2/q) (q/T)V
(3.1)
CV = (U/T)V
(3.2)
q = i gi exp(- i/kT)
(3.3)
Consider a system that has three energy levels, with energies 0, , and 2 (a real example would be a nucleus with
spin = 1 in a magnetic field).
a) Find a general expression for q and U(T) – U(0) for the above system.
b) While your general expression for U(T) – U(0) is somewhat complicated, it takes on a more simple form
for particular limiting conditions. Find the value for U(T) – U(0) for the following two limiting conditions
(1) T (high temperature limit)
(2) T 0 (low temperature limit). Obviously U(T) – U(0) = 0 when T = 0, so what I am asking is the
value for U(T) – U(0) for small values of T.
c) Use your answers in part b to find expressions for CV,m, the constant volume molar heat capacity, for the
same two limiting cases.
4) The lowest energy electronic state for an oxygen atom consists of three distinct states with degeneracy and energy
as listed below
state
3
P2
P1
3
P0
3
degeneracy
5
3
1
energy (cm-1)
0.0
158.3
227.0
Find the population of each of these states and the average energy of an oxygen atom at equilibrium at T = 300.0 K.
Note that the Boltzmann constant can be written as k = 0.69503 cm-1/K.
5) The partition function for the vibrational energy of a collection of diatomic molecules in the gas phase, at
equilibrium, in the harmonic oscillator approximation, is (see Example 15.2 in the text of Atkins).
q = ( 1 – e-/kT )–1
(5.1)
where is the vibrational constant for the molecule.
a) Starting with the above expression for q, find an expression for U(T) – U(0), the average vibrational
energy of a diatomic molecule at equilibrium at temperature T. (Hint: See equn 3.1).
b) Show that in the limit T the contribution to the constant volume molar heat capacity of a diatomic
molecule from vibrational energy is equal to R. (Hint: One way to do this problem is to look at your answer in part a
in the limit T , and then use equn 3.2 to find the contribution to CV,m from molecular vibration).
Solutions.
1) The possible distributions are listed below. The weight is calculated using the relationship
W = N!/(N0!N1!N2!N3!N4!)
where N = 20, N0 is the number of states with zero packets of energy, N1 is the number of states with one packet of
energy, and so forth.
distribution
weight
4, 0, 0, ..., 0
20
3, 1, 0, 0, ..., 0
N0 = 19, N4 = 1
380
N0 = 18, N1 = 1, N3 = 1
2, 2, 0, 0, ..., 0
2, 1, 1, 0, 0, ..., 0
190
3420
N0 = 18, N2 = 2
N0 = 17, N1 = 2, N2 = 1
1, 1, 1, 1, 0, 0, ..., 0
4845
N0 = 16, N1 = 4
2) We can write the normalized Maxwell-Boltzmann distribution as
p(v) dv = N v2 exp(-mv2/2kT) dv
where 0 v < , and N is the normalization factor. Normalization requires
1 = 0 Nv2 exp(-mv2/2kT) dv
= N 0 v2 exp(-mv2/2kT) dv
The above definite integral has the form
0 x2n exp(-ax2) dx = (a)1/2 (1.3.5.....2n-1)/(2a)n+1 ; n = 0, 1, 2, ...
where n = 1, a = m/2kT
So
1 = N (m/2kT)1/2 (kT/m)2 = (/2)1/2 (kT/m)3/2
and so
N = (2/)1/2 (m/kT)3/2
This is the same result as given in the Chapter 20 handout (note that (kT/m) = (RT/M) ).
3)
a)
q = 1 + e-/kT + e-2/kT
U(T) - U(0) = (NkT2/q) (q/T)V
= (NkT2/q)
/T)V { 1 + e-/kT + e-2/kT }
= (NkT2/q) { (/kT2) e-/kT + (2/kT2) e-2/kT }
= (N) e-/kT
(1 + 2 e-/kT )
1 + e-/kT + e-2/kT
b) For T , e-/kT e0 = 1, e-2/kT e0 = 1
U(T) - U(0) N
(1 + 2)
= N
(1 + 1 + 1)
For T 0, e-/kT e- = 0, e-2/kT e- = 0
U(T) - U(0) N e-/kT (1/1) = N e-/kT
c) For T , U(T) - U(0) N, a constant
and so CV /T)V N = 0
For T 0, U(T) - U(0) N e-/kT
and so CV /T)V N e-/kT = N (/kT2) e-/kT = (N2/kT2) e-/kT
Notice that the exponential term approaches zero more rapidly that (N2/kT2) approaches infinity, and so
the heat capacity approaches zero in the limit T 0.
4) q = 5 exp(0) + 3 exp[ - 158.3/(0.69503)(300.0)] + 1 exp[ - 227.0/(0.69503)(300.0)]
= 5 + 1.404 + 0.337 = 6.741
The probability of being in a particular state is
pi = gi exp(i/kT)
q
state
p
3
0.7418
0.2083
0.0499
P2
P1
3
P0
3
The average energy is then
Eave = (0.7418) (0.0 cm-1) + (0.2083) (158.3 cm-1) + (0.0499) (227.0 cm-1) = 44.3 cm-1
5)
a) U(T) – U(0) = (NkT2/q) (q/T)V = (NkT2/q) /T)V (1 - e-/kT)-1
= (NkT2/q) (-1) (1 - e-/kT)-2 (- /kT2)
= N (1 - e-/kT)-1
b) In the limit T , we may use the power series expansion for e-x
e-x = 1 - x + x2/2 - x3/6 +... 1 - x.
And so U(T) – U(0) N [ 1 - (1 - /kT) ]-1 = N (kT/) = NkT
For one mole of molecules in this limit
U(T) – U(0) NAkT = RT
The contribution to the constant volume molar heat capacity for molecular vibrations is then
CV,m = (/T)V (RT) = R
This is in fact the classical result, which applies to harmonic oscillators at high temperatures.