Sovago_suppinfoC09.08.0052
advertisement

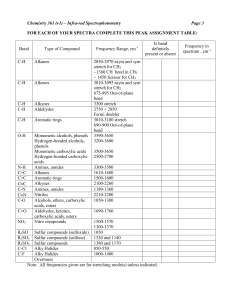
Observation of buried water molecules in phospholipid membranes by surface sum-frequency generation spectroscopy Maria Sovago,1 Erik Vartiainen,2 and Mischa Bonn1 1FOM 2 Institute for Atomic and Molecular Physics, Kruislaan 407, 1098 SJ, Amsterdam, The Netherlands. Lappeenranta University of Technology (LUT) Box20, Fin-53851 Lappeenranta, Finland. Supporting information: 1. SFG theory Conventional SFG yields an intensity spectrum (ISFG) proportional with the modulus squared of the second-order nonlinear polarization (P(2)): 2 I SFG P ( 2 ) ( 2 ) EVIS EIR , (1) Here, (2) represents the second-order susceptibility, EVIS and EIR are the VIS and IR electric fields, respectively. The second-order polarizability can be enhanced when molecules are aligned at the interface. This alignment can be due to the presence of a local electrostatic field, E0. This field will interact with the incident optical fields and can give rise to a third-order effect ((3)): 1-4 2 I SFG ( 2 ) EVIS EIR (3) EVIS EIR E0 NL EVIS EIR , 2 (2) With NL given by: NL ( 2) (3) E0 (3) The magnitude of (3) can have two distinct origins. One is the contribution of the third order electronic nonlinear polarizability (3) of a water molecules, related to the (3) as: (3) N B (3) , where NB is the water bulk density. The other contribution is due to the S1 alignment of water molecules by the electric field E0 created at the surface. As such, the (3) contribution to the SFG intensity can be written as: 4 (3) N B (3) ( 2) (4) bkT Where (2) and (3) are, respectively, the second- and the third-order polarizability, is the permanent dipole moment of the water and b is a constant determined by the particular susceptibility element under construction. Note that we (arbitrarily) include the effect of the charge-induced alignment of the water molecules in the 3) contribution (second term in equation (4)). The large SFG signal observed here for DPPC and DPPE when compared with neutral surfactants suggests 1, in fact, that the second term in equation (4) dominates over the first term. Our findings are in good agreements with the results from SHG experiments on charged solid-water interfaces, where the second term seems to dominate for liquid water. 4 S2 2. MEM analysis The application of MEM to VSFG spectra to extract to retrieve the phase and the Im[ ] spectra, respectively, was described in detail previously. 5 Briefly, the phase spectrum obtained from MEM analysis, (), is used to calculate the absolute phase spectrum, (), and the Im[] spectrum: ( ) ( ) ( ) Im[ ( 2) ] I SFG ( ) sin[ ( ) ( ) NR ] Here, , is the normalized frequency in the frequency interval (with []) and() represent the error phase. For the spectra analyzed here, the error phase is negligible. 5 Therefore, only a correction for the NR phase is necessary. The NR used to calculate the absolute phase for the surfactants and lipids is listed in Table S1. lipid NR LA 2.8 OTAB -2.5 DPTAP -2.3 DMPS -1.4 DPPC -1.2 DPPE -1.1 Table S1. The values of the NR phase used to correct the MEM phase spectra for the surfactants and lipids, respectively. The NR was chosen in the interval [0, 2] such that the following conditions are satisfied. First, the Im[ ] should be negative in the C-H stretch region, as has been shown previously. 6 Second, Im[] ≈ 0 in the regions without resonances. 7 Third, the C-H S3 resonances must appear as (negative) peaks in the Im[] at frequencies known with an accuracy of a few cm-1, e.g. for CH3 symmetric stretch (ss) the peak position is around 2875 cm-1. 8 Furthermore, for the VSFG spectra analyzed here we used an additional restriction for NR. For the surfactants we used the NR phase value close to the values found from the phasesensitive measurements for the cationic and anionic surfactants. 6 There, NR ≈ -2 for cationic surfactant and NR ≈ 2 for anionic surfactant, respectively. Hence, the NR phase value changes by a little over , as the local electric field E0 changes sign as going from a positively charged to a negatively charged interface. The obtained Im[ ] has negative values in the C-H stretch region, as expected, 6 while the values in the O-D stretch region are opposite for OTAB and all lipids. This observation indicates that for these systems the water molecules are oriented oppositely to the CH3 groups. To illustrate the sensitivity of the obtained Im[ ] spectrum to the NR phase correction, we calculated the Im[] for DPTAP monolayers for NR in the interval [-1.1, -3.0], around the chosen NR phase value NR = -2.3 . The results are displayed in Figure S1. For NR = -1.1, the peaks in the C-H stretch region have positive amplitudes and have a dispersive shape. On the contrary, those peaks have negative values for NR = -3.0, and the Im[ ] spectrum is very similar to the one obtained for NR = -2.3. Therefore we conclude that we can determine NR with an accuracy of ~ 0.7. Within this error in the NR, there is significant uncertainty regarding the shape of the spectrum, but we can indisputably determine the sign of the O-D stretch vibrations and determine the orientation of the water dipoles. As the shape of the Im[(2)] spectrum depends on the nonresonant phase, direct conclusions about the change in the water structure near the different lipid headgroups based on changes in the vibrational response are not fully warranted. In principle, the change of the water structure can be determined from the change of the central frequencies of the peaks (or the S4 first moment of the OD intensity distribution) appearing in the Im[(2)] spectrum for the different monolayers studied here: as the frequency of the O-D stretch increases, the strength of the H-bonds decreases. Here, we use the first moment of the SFG intensity distribution in the O-D stretch region of the Im[(2)] spectra to determine the change in the water structure near the different lipids, as described in the main text. Figure S1. Top panel: The recalculated Im[] spectra from MEM analyses for DPTAP monolayer with various NR phase around the chosen NR phase, NR = -2.3. The NR was chosen in such a way that the C-H modes have negative values and Im[] ≈ 0 in the regions without resonances. Bottom panel: the same curves in the C-H stretch region. The vertical dotted line shows the position of the CH3 symmetric stretch (2875 cm-1). S5 References: (1) (2) 113, 4269. (3) (4) (5) (6) 204704. (7) 8, 279. (8) Gragson, D. E.; Richmond, G. L. J. Phys. Chem. B 1998, 102, 3847. Hayes, P. L.; Chen, E. H.; Achtyl, J. L.; Geiger, F. M. J. Phys. Chem. A 2009, Zhao, X. L.; Ong, S. W.; Eisenthal, K. B. Chem. Phys. Lett. 1993, 202, 513. Ong, S. W.; Zhao, X. L.; Eisenthal, K. B. Chem. Phys. Lett. 1992, 191, 327. Sovago, M.; Vartiainen, E.; Bonn, M. J. Phys. Chem. C 2009, 113, 6100. Nihonyanagi, S.; Yamaguchi, S.; Tahara, T. J. Chem. Phys. 2009, 130, Rinia, H. A.; Bonn, M.; Muller, M.; Vartiainen, E. M. Chem. Phys. Chem. 2007, Guyot-Sionnest, P.; Hunt, J. H.; R., S. Y. Phys. Rev. Lett. 1987, 59, 1597. S6