Chapter 1 Introduction: Matter and Measurement
advertisement

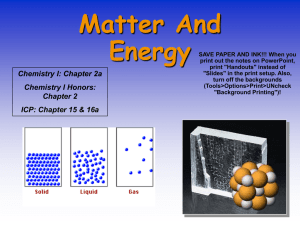
Page 1 of 6 Chapter 1 Introduction: Matter and Measurement What is chemistry? Chemistry – Immediately leads to several questions What is matter? States of matter: (fill in by class response) Definite volume (V)? Definite shape? Compressible/expandable? gas (g) liquid (l) solid (s) state depends on temperature (T) and pressure (P) How do we study matter and its changes? by making careful observations – this includes careful measurements, which leads us to discuss the units involved in making measurements these observations can yield both qualitative (what) and quantitative (how much) results example: qualitative – quantitative – These careful observations and measurements can lead to hypotheses, theories, and laws that allow scientists to categorize and explain observations. Law – ex. Law of conservation of matter: matter is neither created nor destroyed in chemical reactions ex. 4 g of hydrogen reacts with 32 g of oxygen to form 36 g of water Page 2 of 6 Theory – ex. why is matter neither created nor destroyed? Atomic Theory – atoms are neither created not destroyed in chemical reactions (they are just rearranged) example: draw reaction of 2 H2 + O2 2 H2O Laws usually precede theories because they do not have to explain Hypothesis – educated guess to explain observations or “baby” theory – followed by more experimentation Example of a theory – Kinetic Molecular Theory – atoms, molecules, ions in constant random motion unless constrained. Use Ar atom as an example. Draw diagrams of s, l, and g Ar and use them to show how this theory explains all of the observations in chart above describing properties of states of matter How do H2 and O2 molecules react to form water? Kinetic molecular theory explains this also. Molecules hit each other and break the bonds holding atoms together and then new bonds can form. Put equation on board. Page 3 of 6 Where did knowledge come from so laws and theories could be formulated? Demo #1 1. Craft Traditions – practical knowledge of matter was obtained mostly through trial and error (empirically). serendipity – accidental good fortune (discovery) ex. 2. Greek philosophers – observations and logic to study matter nature composed of 4 elements air, fire, water, and earth that have 4 properties – hotness, coldness, dryness, wetness (no atoms) 3. alchemists – furthered chemistry by trying to turn other metals into gold – discovered new compounds and elements 4. Robert Boyle -1661 rejected Greek concept of elements and defined element as a substance which could not be changed into a simpler substance 5. Lavoisier – 1772 – carefully controlled experiments and careful quantitative measurements 6. Dalton – 1808 atomic theory classification of matter Figure 1.9 instead of “uniform throughout” use “uniform at microscopic (atomic and molecular) level” heterogeneous mixture – not evenly distributed at microscopic level examples – homogeneous – uniform at the microscopic level Homogeneous matter is either: homogeneous mixture (solutions) or pure substances solutions have variable composition examples – pure substances have constant composition examples – compounds and elements Page 4 of 6 heterogeneous to homogeneous, and homogeneous mixture to pure substances by physical changes and differences in physical properties filtration, distillation, recrystallization, chromatography compounds to elements by chemical changes compound – molecule – element – need to know first 36 (names and symbols) plus some others (Table 1.2) Written name is not capitalized unless at start of sentence but first letter of symbol is always capitalized. What are these physical & chemical changes and chemical & physical properties? Physical change – does not result in the formation of a new substance examples Chemical change (chemical reaction) – results in the formation of new substance(s) Example: Demo #2, part A How to tell if a chemical change has occurred? Page 5 of 6 Demo #2, part B Demo #2, part C (if time) Open beakers of concentrated HCl and NH3 solutions put next to each other observe gas phase reaction Properties intensive – extensive – physical property – chemical property – Why the difference between physical & chemical changes & chemical and physical properties? Use water as example. Page 6 of 6 Atoms in a compound are held together by strong forces (intramolecular forces, bonds) can only be broken by chemical changes (reactions). Will study later this semester. Molecules attracted to each other by much weaker forces (intermolecular forces) that are much easier to break so components can be separated by physical changes. Will study first of next semester.