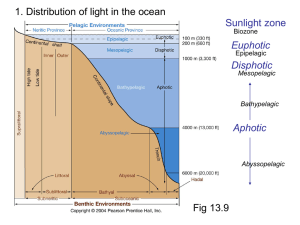
Structural Basis for Spectral Difference in Bioluminescence
advertisement

2005-09-10467B, Hiroaki Kato Supplementary Discussion Sequence analysis The click beetle, Pyrophorus plagiophthalamus (Ppl), is known to possess four types of luciferases distinguished by their luminescence color: green (546 nm), yellow-green (560 nm), yellow (578 nm) and orange (593 nm)1. The amino-acid sequences of the beetle luciferases are 95 to 99% identical with each other, but are only ca. 50% identical with those of the firefly luciferases. Interestingly, the Ppl luciferases that emit green (PplGR) and yellow-green light (PplYG) possess an Ile residue at the position corresponding to Ile288 of Lcr luciferase, but this position is substituted with a Val residue in the Ppl luciferases that emit light with longer wavelengths: yellow (PplYE) and orange (PplOR) (Fig. S2). We therefore believe that the translocation of the side chain of Ile288 generally controls the bioluminescence spectra by affecting the nature of the microenvironment for the excited state of oxyluciferin. The change in the hydrogen bonding at Ser286 might serve as a molecular switch that triggers the movement of Ile288. A luciferase from a railroad-worm Phrixothrix_vivianii (PvGR)2 emits green light (549 nm), but has a Val residue at the position corresponding to Ile288 of Lcr luciferase. In PvGR, however, two additional key residues corresponding to Ser286 and Ser349 of firefly luciferases are mutated to Thr and Cys residues, respectively. 1 The overall 2005-09-10467B, Hiroaki Kato effects of these mutations on bioluminescence color are not fully understood at the current level of our structural study, but do not exclude the possibility of general and integral role of Ile288 in the firefly luciferases. We therefore conclude that the conformational change involving the movement of Ile288 is a common, but not the sole catalytic machinery that is used widely by insect luciferases to control their bioluminescence spectra. 2 2005-09-10467B, Hiroaki Kato Supplementary Methods Synthesis of 5’-O-[N-(dehydroluciferyl)-sulfamoyl]adenosine (DLSA) In the molecular design of DLSA, we used dehydroluciferin, rather than luciferin itself, as the luciferyl moiety of the ligand to mimic the growing sp2-character at C-4 in the thiazoline ring predicted in the subsequent oxidation step of the luciferyl adenylate intermediate by molecular oxygen. Furthermore, the planar thiazole ring is analogous to oxyluciferin in a dianionic enolate form as a putative emitter for the yellow-green bioluminescence3, thereby allowing DLSA to mimic the late transition state for the activation of luciferin and/or the excited state of oxyluciferin. During the course of our study, however, the same molecule was synthesized as an inhibitor of Ppy luciferase4. The followings are our synthetic route. General methods. Compounds were purified by flash column chromatography on silica gel 60 (230–400 mesh, Merck, No. 9385) or by reversed-phase medium-pressure column chromatography on an ULTRA PACK™ octadecyl silica gel column (ODS-S-50B, 50 1 mm, 120 Å, Yamazen Co., Osaka, Japan). H and 13C NMR spectra were recorded on a Varian VXR-200 (200 MHz) or a JEOL JNM-AL400 (400 MHz) spectrometer using tetramethylsilane (for CDCl3 and DMSO-d6) or 3-(trimethylsilyl)propanesulfonic acid 3 2005-09-10467B, Hiroaki Kato sodium salt (for D2O) as an internal standard. Chemical shifts were recorded in ppm with the internal standard set at H = 0.00. Elemental analyses were performed on a Yanaco MT-5 system. Mass spectra were obtained on a JEOL JMS700 spectrometer. Dehydroluciferin pentafluorophenyl ester (Compound 1) A solution of 2-(6-hydroxy-2-benzothiazolyl)thiazole-4-carboxylic acid (dehydroluciferin, DL)5,6 (111 mg, 0.4 mmol) and EDC (96 mg, 0.5 mmol) in pyridine (20 ml) was mixed with pentafluorophenol (92 mg, 0.5 mmol), and the mixture was stirred at ambient temperature for 16 h. Pyridine was removed by evaporation, and the residue was taken up in a mixture of AcOEt and MeOH (9:1). The solution was filtered, washed with 0.1 M HCl, and concentrated in vacuo to leave a brown oil. The residual oil was purified by flash column chromatography on silica gel (gradient of 0 to 100% AcOEt in hexane) to give Compound 1 as a yellow solid (250 mg, 70% purity) (84% yield). 1 H-NMR (200 MHz, DMSO-d6) H: 9.23 (s, 1H, thiazole H-5), 7.98 (d, 1H, J = 8.8 Hz, benzothiazole H-4), 7.50 (d, 1H, J = 2.4 Hz, H-7), 7.09 (dd, 1H, J = 9 and 2.4 Hz, H-5). 2’,3’-O-Isopropylidene-5’-O-[N-(dehydroluciferyl)-sulfamoyl]adenosine (2) A solution of DBU (91 mg, 0.6 mmol) was added at ambient temperature to a solution 4 2005-09-10467B, Hiroaki Kato of 2’, 3’-O-isopropylidene-5’-sulfamoyladenosine7 (160 mg, 0.41 mmol) in dry DMF (14 ml). After 10 min, a solution of Compound 1 (180 mg, 0.41 mmol) in dry DMF (1 ml) was added. The mixture was stirred under a nitrogen atmosphere for 16 h. Pyridine (1 ml) was added, and the mixture was stirred for additional 4 h. The reaction mixture was concentrated in vacuo, and the residual oil was purified by flash column chromatography on silica gel (gradient of 5 to 30% MeOH in AcOEt) to give Compound 2 as a bright yellow solid (55 mg, 21%). 1 H-NMR (200 MHz, DMSO-d6) H: 8.44 (s, 1H, thiazole H-5), 8.23 (s, 1H, adenine H-8), 8.13 (s, 1H, adenine H-2), 7.90 (d, 1H, J = 9.0 Hz, benzothiazole H-4), 7.48 (d, 1H, J = 2.2 Hz, H-7), 7.3 (br s, 2H, NH2), 7.07 (dd, 1H, J = 9.0 and 2.2 Hz, H-5), 6.19 (d, 1H, J = 3.0 Hz, ribose 1’-H), 5.41 (dd, 1H, J = 6.0 and 3.0 Hz, 2’-H), 5.08 (dd, 1H, J = 6.2 and 2.2 Hz, 3’-H), 4.5 (m, 1H, 4’-H), 4.1 (m, 2H, 5’-H). 5’-O-[N-(dehydroluciferyl)-sulfamoyl]adenosine (DLSA) Compound 2 (55 mg, 0.09 mmol) was dissolved in a mixture of trifluoroacetic acid and water (2:1 v/v, 3 ml). The mixture was stirred for 3 h and was evaporated. trifluoroacetic acid was removed by azeotrope with ethanol and toluene. A trace of The residual oil was taken up in water, filtered, and purified by a reversed-phase column chromatography on a Dianion HP20SS resin (linear gradient of 0 to 30% THF in water). 5 2005-09-10467B, Hiroaki Kato The fractions containing DLSA (Abs. 254 nm) was evaporated and lyophilized from water to afford DLSA as a bright yellow light solid (9 mg, 17%). 1 H-NMR (400 MHz, CD3OD) H: 8.51 (s, 1H, thiazole H-5), 8.32 (s, 1H, adenine H-8), 8.15 (s, 1H, adenine H-2), 7.83 (d, 1H, J = 8.9 Hz, benzothiazole H-4), 7.32 (d, 1H, J = 2.4 Hz, H-7), 7.02 (dd, 1H, J = 8.9 and 2.4 Hz, H-5), 6.08 (d, 1H, J = 5.5 Hz, ribose 1’-H), 4.72 (t, 1H, J = 5.3 Hz, 2’-H), 4.4-4.5 (m, 3H, 3’ and 5’-H), 4.3 (m, 1H, 4’-H). HRMS (FAB, 4-nitrobenzyl alcohol): Calcd. for C21H19N8O8S3 (M+H+), 607.0491; found, 607.0521; Calcd. for C21H18N8O8S3Na (M+Na+), 629.0307; found, 629.0303. DNA Techniques pTM18 contains the cDNA of the thermostable mutant luciferase (T217I) from the firefly Luciola cruciata, which produces the same yellow-green light as the wild-type enzyme does; pGLf37-CM19 contains the cDNA of the mutant luciferase (S286N), which displays an orange-shifted bioluminescence emission spectra. Nco I and BamH I sites were introduced by the polymerase chain reaction to 5’ and 3’ ends, respectively, of a 1.6-kb fragment that included the T217I cDNA; the ATG sequence in the restriction site of Nco I (CCATGG) was matched with the initiation codon of T217I luciferase. For construction of a T217I luciferase with the three N-terminal amino acids deleted, the restriction sequence for Nco I site was introduced in the region from 8 to 13 6 2005-09-10467B, Hiroaki Kato nucleotides downstream of the initiation codon. The synthetic mutagenic primers used were as follows (the restriction sites are underlined): forward primer, 5’-CATGCCATGGAAAACATGGAAAACGATGAA; forward primer for the deletion mutant, 5’-CCCGCCATGGAAAACGATGAAAATATTGTA; 5’-AAGGATCCTCACATCTTAGCAACTGG. reverse primer, The resultant DNA fragments were digested with Nco I and BamH I, and ligated to pTV118N (Takara, Shiga, Japan) digested with the same enzymes to yield pTVLt and pTVLt, respectively (t: thermostable). A 0.85-kb Hpa I (the restriction site was located at nucleotide position 789 to 794 from the initiation codon)–BamH I fragment isolated from pTVLt was replaced with a fragment derived from pGLf37-CM1 digested with the same enzymes to construct pTVLtS286N. method with Mutants, I288A and I288V, were constructed by PCR minor modification. Primers used were 5'-AAACAAGGTCGGTACCAGAGCAACACTTGTACATTT-3', 5'-CAAGATTATAAATGTACTAGTGTTGTTCTTGTACCGACCTTG-3', and primers used when constructing pTVLt, and pTVLt was used as template. The nucleotide sequences of all of the plasmid inserts were confirmed with an Applied Biosystems 310 DNA sequencer (Foster City, CA). Protein Expression and Purification 7 2005-09-10467B, Hiroaki Kato Wild-type (T217I) and mutant luciferases were produced in Escherichia coli HB101 cells. The recombinant E. coli cells producing the enzymes were grown aerobically at 30˚C for 18 h in LB medium containing 0.1 mg/ml ampicillin. The cells harvested from a 4-L culture were disrupted by sonication. The cell lysate was subjected to centrifugation at 75,600 x g for 20 min to remove cellular debris. The resulting cell-free extract was brought to 30% saturation of ammonium sulfate, and the precipitated protein was removed by centrifugation. The soluble supernatant was dialyzed against buffer 1A (10 mM potassium phosphate (pH 6.8), 5% (w/v) glycerol, 1 mM dithiothreitol (DTT), and 1 mM ethylenediaminetetraacetic acid (EDTA)). The enzyme was first purified at room temperature with an ÄKTA explorer 10S system (Amersham Biosciences) and a HiLoad 16/10 SP Sepharose FF cation exchanger column (column volume: 20 ml) employing a linear gradient of the buffers 1A and 1B (500 mM potassium phosphate (pH 6.8), 5% (w/v) glycerol, 1 mM DTT, and 1 mM EDTA); sample injection: 0% B, 1 CV; 0–18% B, 6 CV; 100% B, 1 CV (flow rate: 3 ml/min). The active fractions were pooled, dialyzed against buffer 2A (10 mM potassium phosphate (pH 6.8), 5% (w/v) glycerol, and 1 mM DTT), and applied to a Bio-Scale CHT5-I hydroxyapatite column (column volume: 5 ml) connected to the ÄKTA system. The enzyme was eluted employing a linear gradient of the buffers 2A and 2B (500 mM potassium phosphate (pH 6.8), 5% (w/v) glycerol, and 1 mM DTT); 8 2005-09-10467B, Hiroaki Kato sample injection: 0% B, 3 CV; 0–40% B, 16 CV; 100% B, 1 CV (flow rate: 4 ml/min). The active fractions were concentrated, dialyzed against 20 mM Tris-HCl (pH 8.0), 10% (w/v) glycerol, and 2 mM 2-mercaptoethanol, and used as a purified preparation of the enzyme. Seeding techniques for crystallization In order to produce a seeding solution, the clusters of crystals were placed in 60 l of the reservoir solution and crushed. Tenfold serial dilutions of the crushed crystals were made in the reservoir solution. Hanging drops were formed with the enzyme solution and the diluted seeding solution. Several single crystals were obtained from approximately 104-fold dilutions of seeds after 2–3 days. Measurement of bioluminescence emission spectra Emission spectra were recorded by multichannel spectrophotometer MCPD-7000 (Otsuka Electronics, Japan). Substrate solution (500 L) containing 100 mM HEPES-Na (pH 7.8), 0.6 mM luciferin, 3.2 mM ATP, and 10 mM MgSO4 was mixed with the wild-type or mutant (S286N, I288V, or I288A) luciferases solution (500 L) in a quartz cell. The concentration of the wild-type and mutant are 70 and 140 g/ml, respectively. 9 2005-09-10467B, Hiroaki Kato References 1. 2. 3. 4. 5. 6. 7. 8. 9. Wood, K. V., Lam, Y. A., Seliger, H. H. & McElroy, W. D. Complementary DNA coding click beetle luciferases can elicit bioluminescence of different colors. Science 244, 700-2 (1989). Viviani, V. R., Bechara, E. J. & Ohmiya, Y. Cloning, sequence analysis, and expression of active Phrixothrix railroad-worms luciferases: relationship between bioluminescence spectra and primary structures. Biochemistry 38, 8271-9 (1999). White, E. H., Rapaport, E., Hopkins, T. A. & Seliger, H. H. Chemi- and bioluminescence of firefly luciferin. J Am Chem Soc 91, 2178-80 (1969). Branchini, B. R., Murtiashaw, M. H., Carmody, J. N., Mygatt, E. E. & Southworth, T. L. Synthesis of an N-acyl sulfamate analog of luciferyl-AMP: a stable and potent inhibitor of firefly luciferase. Bioorg Med Chem Lett 15, 3860-4 (2005). White, E. H., McCapra, F. & Field, G. F. The structure and synthesis of firefly luciferin. 85, 337-343 (1963). Bowie, L. Synthesis of firefly luciferin and structural analogs. Methods Enzymol 57, 15-28 (1978). Heacock, D., Forsyth, C. J., Shiba, K. & Musier.-Forsyth, K. Synthesis and Aminoacyl-tRNA Synthetase Inhibitory Activity of Prolyl Adenylate Analogs. Bioorg. Chem. 24, 273-289 (1996). Kajiyama, N. & Nakano, E. Thermostabilization of firefly luciferase by a single amino acid substitution at position 217. Biochemistry 32, 13795-9 (1993). Kajiyama, N. & Nakano, E. Isolation and characterization of mutants of firefly luciferase which produce different colors of light. Protein Eng 4, 691-3 (1991). 10 2005-09-10467B, Hiroaki Kato Supplementary Figure Legends Figure S1 a, Stereo view of the luciferin binding site of LcrLuc (WT) in complex with DLSA. The polar interactions are shown as dashed lines. The sulfamoyladenosine moiety of DLSA is omitted from the figure for clarity. b, Superposition of the structures of LcrLuc (WT):MgATP (light blue) and LcrLuc (WT):AMP:oxyluciferin (white) complexes. dashed lines. The polar interactions are shown as The - and - phosphates of ATP are not visible. c, Superposition of the LcrLuc (WT) : DLSA (green) and LcrLuc (S286N) : DLSA (pink) complexes. The polar interactions are shown as dashed lines. Figure S2 Sequence alignment of firefly and click beetle luciferases. Firefly luciferases: Lcr, Luciola cruciata; Lla, Luciola lateralis; Lmi, Luciola mingrelica; Hpa, Hotaria parvula; Pmi, Pyrocoelia miyako; Pru, Pyrocoelia rufa; Lno, Lampyris noctiluca; Ppy, Photinus pyralis; Ppe, Photuris pennsylvanica. Click beetle luciferases: Ppl, Pyrophorus plagiophthalamus; Pyt, Pyrearinus termitilluminans. The four Ppl luciferases with differently colored emission spectra are indicated as PplYE (yellow), PplOR (orange), PplYG (yellow-green), and PplGR (green). The secondary structures involved in the luciferin-binding site are drawn in red. The position of Ile 288 and the conserved key residues 11 2005-09-10467B, Hiroaki Kato are highlighted in black and red, respectively. Figure S3 Synthesis of 5’ -O-[N-(dehydroluciferyl)-sulfamoyl] adenosine (DLSA). (i) pentafluorophenol, EDC, pyridine, 25˚C, 16 hr, 84%; (ii) 2’, 3’-O-isopropylidene-5’-sulfamoyladenosine, 1, DBU, DMF, 25˚C, 20 hr, 21%; (iii) TFA - water (2:1), 25˚C, 3 hr, Dianion HP20SS (0 to 30% water in THF), 17%. 12 2005-09-10467B, Hiroaki Kato Supplementary Movie Legends Movie S1 Movements involved in the luminescence reaction of wild-type luciferase. The movie is composed of 4 steps; (1) before reaction (MgATP complex, The -, and - phosphates of ATP are not visible.), (2) intermediate (mimicked by DLSA complex), (3) excited state (model) and (4) after reaction (AMP:oxyluciferin complex). Excited state is a speculative model predicted by DLSA and AMP:oxyluciferin complexes. The excited oxyluciferin is drawn in green. All ligands are shown in ball and stick model. Ile288 (blue) and the other amino acid residues are drawn in space-filing and stick models, respectively. The polar interactions are shown as dashed lines. Movie S2 Movements involved in the luminescence reaction of S286N luciferase. The movie is composed of 4 steps; (1) before reaction (calculated with the wild-type MgATP complex), (2) intermediate (mimicked by DLSA complex), (3) excited state (model) and (4) after reaction (AMP:oxyluciferin complex; unpublished data). Excitation state is a speculative model predicted by DLSA and AMP:oxyluciferin complexes. The excited oxyluciferin is drawn in red. The other settings are the same as in Movie S1. 13 2005-09-10467B, Hiroaki Kato Supplementary Table Table S1 Data collection and refinement statistics Wild:MgATP Wild:DLSA Wild:AMP S286N:DLSA :Oxyluciferin Data collection Space group P 212121 P 212121 P 212121 P 212121 57.8, 182.0, 53.6 57.6,181.3,52.0 57.6,181.4,52.7 57.4,181.1,52.2 () 90, 90, 90 90, 90, 90 90, 90, 90 90, 90, 90 Resolution (Å) 2.30 1.30 1.60 1.45 (2.38 – 2.30) * (1.35 – 1.30) (1.66 – 1.60) (1.53 – 1.45) Rsym or Rmerge 0.058 (0.150) 0.054 (0.209) 0.054 (0.210) 0.090 (0.283) I/I 10.7 (4.0) 9.0 (2.6) 10.1 (3.0) 6.0 (2.6) Completeness (%) 97.9 (91.1) 99.2 (95.8) 99.7 (97.4) 97.6 (95.0) Redundancy 3.11 (2.55) 3.96 (3.36) 6.63 (3.58) 6.91 (7.47) Resolution (Å) 2.30 1.30 1.60 1.45 No. reflections 25394 133611 73719 94838 Rwork/ Rfree 0.179 / 0.228 0.180 / 0.202 0.184 / 0.210 0.165 / 0.182 Protein 3976 4127 4111 4126 Ligand/ion 23 42 39 42 Water 283 794 626 843 Protein 23.5 10.0 15.1 9.9 Ligand/ion 13.6 5.4 7.6 5.0 Water 29.3 21.8 26.4 22.8 Bond lengths (Å) 0.011 0.009 0.011 0.010 Bond angles (º) 1.27 1.38 1.33 1.39 Cell dimensions a, b, c (Å) Refinement No. atoms B-factors R.m.s deviations *Highest resolution shell is shown in parenthesis. Rmerge = |Ii-<Ii>| / Ii, where Ii = observed intensity, <Ii> = average intensity of multiple measurements 14