DETERMINATION OF THE CHARGE ON AN ELECTRON
advertisement

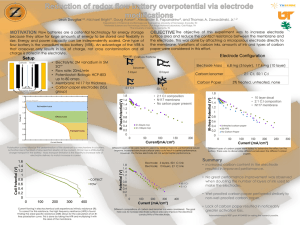
DETERMINATION OF THE CHARGE ON AN ELECTRON! BACKGROUND INFORMATION: Robert Millikan, an American scientist, determined the charge on a single electron in 1909. You will be collecting data to make the same determination of the charge on an electron. Although your procedure will be different than his, your calculations should give you the same charge. You will be collecting zinc atoms on a zinc electrode to carry out this activity. Zinc ions, or Zn+2 atoms, will each gain 2 electrons to become neutral zinc atoms that “electroplate” on one of two zinc electrodes. The other zinc electrode will lose the same number of zinc atoms as they become zinc ions, or Zn+2 atoms. PURPOSE: To experimentally determine the exact charge of an electron in coulombs by plating zinc MATERIALS: 2 zinc strips 125 mL beaker alligator clips steel wool 1.0M ZnSO4 power source digital balance multimeter squirt bottle with acetone PROCEDURE: 1. GOGGLES ARE MANDATORY DURING THIS ENTIRE ACTIVITY. 2. Lay the zinc electrodes on paper towels, and polish both of them on both sides with steel wool. Using a pencil, label one electrode as “+” and the other electrode as “-“. 3. Find the mass of both electrodes and record. 4. Place the electrodes in a 125 mL beaker, so that the labeled ends are at the top of the beaker. Bend the tops over the rim of the beaker. 5. Connect the red wire into the 10A port on the multimeter. Connect the black wire into the COM port on the multimeter. Set the blue knob to 20m/10A” on the “A” option. Make sure the black switch is set to DC and not to AC. 6. Connect the “+” end of the power source to the “+” electrode with an alligator clip of a wire lead. 7. Connect the “-” end of the power source to the red wire on the multimeter with an alligator clip of a wire lead. 8. Connect the black wire on the multimeter to the “-” electrode with an alligator clip of a wire lead. 9. Make sure your instructor inspects your set-up, and consult the diagram above. 10. Pour ZnSO4 solution into the 125 mL beaker until it is about an inch or so from the rim. 11. Turn on the power source, and adjust the power to 6 volts. Allow the current to flow for 20-25 minutes. RECORD THE EXACT TIME THE CURRENT FLOWS THROUGH THE SOLUTION – THIS IS IMPORTANT! 12. Record the amperage reading off of the multimeter every minute for the entire 20-25 minutes. At the conclusion of your experiment, average the amperage reading for your entire experiment. Record your time elapsed. 13. Disconnect all wire leads, remove your electrodes, and LET THEM DRY COMPLETELY FOR 10 MINUTES. Wipe down the electrode that lost mass, but do not wipe down the electrode that gained mass – simply allow it to dry completely. You can rinse it with acetone to speed the drying process. Mass both electrodes after 10 minutes. 14. Pour the remaining ZnSO4 into the waste container designated by Mr. Jauss. MEASURED DATA: Create a table in your final lab report that shows the following data: Mass of both electrodes before and after electrolysis, Voltage setting, Amp readings every minute for 20-25 minutes, time elapsed in minutes CALCULATIONS: 1. Calculate the total time elapsed in seconds (s). 2. Calculate your average amp (A) reading by adding and dividing by total number of readings. 3. An amp (A) is a measurement of how many coulombs of charge flow through a solution in one second. Multiply the average amps used in the experiment by the number of seconds, to obtain the total charge used in the experiment in coulombs. 4. Calculate the change in mass of each electrode. Both electrodes should have close to the same mass = the “+” electrode should lose the same mass that the “-” electrode gained. Do calculations for both electrodes! 5. Calculate the total number of zinc atoms that were gained at the “-” electrode AND lost at the “+” electrode (the mass of a Zn atom is 1.09 x 10-22 grams). 6. Calculate how many total electrons were required to plate the zinc atoms. Remember – every Zn+2 ion must gain two electrons in order to become a Zn atom, and every Zn atom must lose two electrons in order to become a Zn+2 ion. Do this for both electrodes. 7. Now, calculate the charge that one electron possesses. Take the total charge in coulombs that you flowed into your solution (#3) and divide by the total number of electrons that you gained at the “-” electrode (#6). Do this for the number of electrons you lost at the “+” electrode as well. 8. How does your calculation for the charge on an electron compare to the accepted value as determined by Millikan of 1.60 x 10-19 coulombs? Calculate your % error for your values at both the “-” and “+” electrodes. DERIVED DATA: Create a second data table in your final lab report the shows the following data: Time elapsed in seconds, average amp reading, total charge in coulombs, change in mass at each electrode, number of zinc atoms gained at “-” electrode, number of zinc atoms gained at “+” electrode, total number of electrons required at “-” electrode, total number of electrons required at “+” electrode charge on an electron at “” electrode, charge on an electron at “+” electrode, % error for both electrodes. CONCLUSION: 1. List and describe at least two sources of error when conducting this lab, and how it could affect your results. 2. Why could we not have used sodium sulfate solution instead of zinc sulfate solution? 3. Discuss in detail why we can use either the mass lost from the “+” electrode or the mass gained from the “-” electrode for our calculations!