Photoelectron spectroscopy (PES)
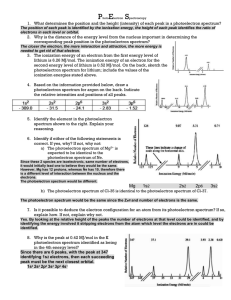
When photon ionizes…
The PES process
PES utilizes photo-ionization and analysis of the kinetic energy distribution of
the emitted photoelectrons to study the composition and electronic state of the
surface region of a sample.
PES measures the energy required to eject electrons from each occupied MO.
Traditionally, when the technique has been used for surface studies it has been
subdivided according to the source of exciting radiation into :
X-ray Photoelectron Spectroscopy (XPS)
- using soft x-ray (200-2000 eV) radiation to examine core-levels
Ultraviolet Photoelectron Spectroscopy (UPS)
- using vacuum UV (10-45 eV) radiation to examine valence-levels.
Calculation
What is the wavelength equivalent of 200 eV energy?
Using formula
λ = h·c/E
Data:
1 eV = 1.602 10-19 J
hc = 1.986 10-25 Jm,
λ = 1.986 10-25 Jm/(200·1.602 10-19 J) = 6.2 nm.
The photoionization process
In XPS the photon is absorbed by an atom in a molecule, leading to
ionization and the emission of a core (inner shell) electron.
By contrast, in UPS the photon interacts with valence levels of the
molecule, leading to ionisation by removal of one of these valence
electrons.
M + hν = M+ + e-
1.
Photoelectron spectroscopy is based upon a single photon in/electron out
process. The law of energy conservation says
1
h me ve2 E M E M
2
1
2.
The total energy of photon is the sum of the binding energy, BE
BE E M E M and the kinetic energy of electron.
BE is the difference in energy between the ionized and neutral atoms, it can be
identified the ionization energy (Koopman’s theorem).
The method
The kinetic energy distribution of the emitted photoelectrons (i.e. the number of
emitted photoelectrons as a function of their kinetic energy) can be measured
using any appropriate electron energy analyser and a photoelectron spectrum
can thus be recorded. PES uses monochromatic sources of radiation.
Figure 1. Schematic diagram of a photoelectron energy analyzer, detects the
number of emitted electrons per unit time.
Sample is placed in a high vacuum chamber (P ~ 10−8 millibar) for excluding other
ionization processes than the studied one. The preferred option for
photoemission experiments is a concentric hemispherical analyser (CHA) which
uses an electric field between two hemispherical surfaces to disperse the
electrons according to their kinetic energy.
The higher the velocity measured the lower the ionization energy of
photoelectron.
2
Examples
Fig.2. The ionization process of H atom: H + hν → H+ + eA single band observed: ionization energy of 1s1 electron is13.6 eV = 1312
kJ/mol
Fig.3. The ionization process of H2 molecule: H2 + hν → H2+ + eOne band spreading out from 15.45 eV to 18 eV, corresponding to ionization of
the (σ)2 electrons. Sharp peaks (“fine structure”) due to vibration in H 2 .
X-ray Photoelectron Spectroscopy (XPS)
The intensity of the peaks is related to the concentration of the element within
the sampled region. The most commonly employed x-ray sources are those
giving rise to:
Mg Kα radiation: hν = 1253.6 eV
Al Kα radiation: hν = 1486.6 eV
Each element will give rise to a characteristic set of peaks in the photoelectron
spectrum at kinetic energies determined by the photon energy and the respective
ionization energies.
Core ionization energies are characteristic of the individual atom rather than the
overall molecule.
The presence of peaks at particular energies therefore indicates the presence of a
specific element in the sample under study.
3
The XPS spectrum of Pd metal
The diagram below shows a real XPS spectrum obtained from a Pd metal
sample using Mg Kα radiation
Figure 4. Kinetic energy (KE) peaks
It is a trivial matter to transform the spectrum so that it is plotted against binding
energy (BE) as opposed to KE.
Figure 5. Binding energy peaks.
The most intense peak at ca. 335 eV is due to emission from the 3d levels of the
Pd atoms, whilst the 3p and 3s levels give rise to the peaks at ca. 534/561 eV
and 673 eV respectively.
4
The XPS can distinguish between chemically inequivalent but otherwise
identical atoms.
Example: Sodium azide
Fig. 6.The XPS spectrum of solid NaN3 excited by Al Kα radiation showing the
region of N core ionization and the assignment.
In the region of 400 eV the azide ion ( N 3 ) gives a doublet structure with
splitting 6 eV in XPS spectrum. This range is typical of N1s electrons. The
structure of the ion is characterized as,
-
form a:
-
NNN
or
form b:
NNN
and the XSP spectrum supports form a charge distribution. The peak position
and integrated area under the peak are the factors we use for explanation in
Figure. 6.
The negative charge on an atom lowers the ionization energy while positive
charge on the central atom raises it, because the repelling force lowers the
ionization energy.
Therefore, the higher intensity lower ionization energy peak belongs to the two
negative N atom. The integrated intensity ratio is 2:1 for N- / N+.
5
Ultraviolet Photoelectron Spectroscopy (UPS)
Koopman’s theorem ignores the fact that the electrons remaining rearrange their
distribution when ionization occurs.
Figure 7. The UPS spectrum of HBr.
The lowest ionization energy band corresponds to the ionization of bromine
lone-pair electron. The higher ionization energy band corresponds to the
ionization of a bonding σ-electron.
When ejected electron arises from bonding orbital, as in the case of HBr+ a
vibration term, E v should be added to energy balance equation (Eq.2.).
1
h me ve2 I Ev
2
3.
6
 0
0