Leaf Design & Stomata: Red Oak Lab Exercise
advertisement

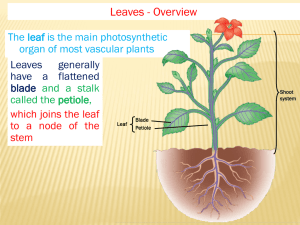
Biology of Woody Plants Leaf Design and Placement of Stomata Lab exercise written by Lindsey Colby and Judy Stone Introduction Living plants require a constant supply of available water for a variety of essential activities: water is a raw material in photosynthesis, it is the main component, by weight, of the plant body, and it acts as a solvent in which minerals and organic substances move within the plant and in which complex chemical reactions take place. Yet only a small fraction of the water absorbed by the plant is retained and metabolized. Most is lost from the leaves by a process called transpiration. The major function that most leaves perform is photosynthesis, which requires CO2 and water as well as light. Unfortunately for the plant, whenever stomata are open to admit CO2, water vapor is inevitably lost. Today in lab, we will focus on the placement and density of stomata in relation to the demands of leaves for carbon dioxide. Description of leaf veins Cells of the palisade and spongy layers must be close to their air supply for gas exchange and they must also be close to a leaf vein, with its xylem to supply water and minerals and phloem to remove synthesized food The photo shows the network of leaf veins in a maple leaf. Probably no cell in the spongy layer is more than two cells away from a vein. The xylem and phloem of veins is often surrounded by layers of sclerenchyma cells. These impart strength to the vein providing a stiff framework to support the soft tissues of the leaf blade. Fig 1. Surface of maple leaf showing veins. Description of Leaf Cross Section Mount a prepared slide of the cross section of a leaf from Syringa (Lilac). Find the following 4 distinct tissue layers: 1) Upper epidermis. This is a single layer of cells containing few or no chloroplasts. The cells are quite transparent and permit most of the light that strikes them to pass through to the underlying cells. The upper surface is covered with a waxy, waterproof cuticle, which serves to reduce water loss from the leaf. 2) Palisade layer. This consists of one or more layers of cylindrical cells oriented with their long axis perpendicular to the plane of the leaf. The cells are filled with chloroplasts (usually several dozen of them) and carry on most of the photosynthesis in the leaf. 1 Figure 2. Section of a typical leaf showing four layers of tissue and vascularization 3) Spongy layer. Lying beneath the palisade layer, its cells are irregular in shape and loosely packed. Although they contain a few chloroplasts, their main function seems to be the temporary storage of sugars and amino acids synthesized in the palisade layer. They also aid in the exchange of gases between the leaf and the environment. During the day, these cells give off oxygen and water vapor to the air spaces that surround them. They also pick up carbon dioxide from the air spaces. The air spaces are interconnected and eventually open to the outside through pores called stomata (sing., stoma). 4) Lower epidermis. Typically. most of the stomata (thousands per square centimeter) are located in the lower epidermis. Although most of the cells of the lower epidermis resemble those of the upper epidermis, each stoma is flanked by two sausage-shaped cells called guard cells. These differ from the other cells of the lower epidermis not only in their shape but also in having chloroplasts. The guard cells regulate the opening and closing of the stomata. Thus they control the exchange of gases between the leaf and the surrounding atmosphere. Detailed description of leaf epidermis The imprint of the leaf epidermis reveals a remarkable reconstruction of the original leaf. The epidermis constitutes the dermal tissue system of leaves, floral parts, fruits and seeds and of stems and roots until they undergo considerable secondary growth. Epidermal cells are quite variable both functionally and structurally. In addition to the relatively unspecialized cells, which form the bulk of the epidermis, the epidermis may contain guard cells, many types of appendages or trichomes, and other kinds of specialized cells. The bulk of epidermal cells is compactly arranged and gives considerable mechanical protection to the plant parts. The walls of the epidermal cells of the aerial parts are covered with a cuticle, which minimizes water loss. The cuticle consists mainly of cutin and wax. In many plants the 2 wax is exuded over the surface of the cuticles, either in smooth sheets or as rods or filaments extending upward from the surface, the so-called epicuticular wax responsible for the whitish or bluish "bloom" on the surface of some leaves and fruits (think of white and blue spruce). Interspersed among the flat, tightly packed epidermal cells, which typically lack chloroplasts, are the chloroplast-containing guard cells that regulate small pores or stomata (singular stoma), in the aerial parts of the plant and hence control the movement of gases, including water vapor, into and out of those parts. (The term "stoma" commonly is applied to the pore and the two guard cells.) Although stomata occur on all aerial parts, they are most abundant on leaves. Guard cells are often associated with epidermal cells that differ in shape from the ordinary epidermal cells. Such cells are termed subsidiary cells. Figure 3. Assorted leaf epidermal layers of leaves showing stomata, guard and subsidiary cells. Density of stomata The density of stomata on a leaf varies with such factors as the typical levels of temperature, humidity, and light intensity at the time the plant develops as well as the ambient concentration of carbon dioxide in the air around the leaves. The relationship is inverse; that is, as CO2 goes up, the number of stomata that form in the developing leaf epidermis goes down, and vice versa. Some evidence: Plants grown in an artificial atmosphere with a high level of CO2 have fewer stomata than normal. Herbarium specimens reveal that the number of stomata in a given species has been declining over the last 200 years - the time of the industrial revolution and rising levels of CO2 in the atmosphere. Stomatal density can be quantified by determining the stomatal index: the ratio of the number of stomata in a given area divided by the total number of stomata and other epidermal cells in that same area. How does the plant determine how many stomata to produce? It turns out that the mature leaves on the plant detect the conditions around them and send a signal (its nature still unknown) that adjusts the number of stomata that will form on the developing leaves. Two experiments (Lake, et al., 2001) revealed that when the mature leaves of the plant Arabidopsis are encased in glass tubes filled with high levels (720 ppm) of CO2, the developing leaves have fewer stomata than normal even though they are growing in normal air (360 ppm). Conversely, when the mature leaves are given normal air (360 ppm CO2) while the shoot is exposed to high CO2 (720 ppm), the new leaves develop with the normal stomatal index. 3 Study of the Density of Stomatal Apparati on Leaves of Red Oak (Quercus rubra) Motivation for experiment: We know that sun leaves are thicker than shade leaves, with more palisade cells and the potential for higher photosynthetic rates. It seems likely that more CO2 per cm leaf area would be required in sun leaves, so that CO2 concentration would not limit photosynthetic rate. For shade leaves, light rather than CO2 may limit photosynthesis, and excessive stomata may cause undue water loss. Does the development of sun and shade leaves allow different densities of stomata to be created? Objective: To determine whether position in the canopy affects stomatal density Procedure: 1) Go outside and sample oak leaves from the ground or off of trees. Your goal is to collect 10 sun and 10 shade leaves. Remember that shade leaves tend to be larger, with shallower lobes, and sun leaves tend to be smaller and more deeply lobed. Discuss with your team advantages/disadvantages of various sampling techniques and be prepared to describe your approach in your lab report. 2) Paint a thin layer of clear nail polish on both the adaxil (top) and abaxil (bottom) side of each leaf. Allow to dry for a few minutes then peel gently off with a pair of forceps. Make a wet mount using a slide, drop of water and cover slip and mount the slide on a microscope. Start at low magnification and work your way up to high dry (400X). Which side of the leaf would be best to use for this study to detect differences in density of stomata in sun versus shade leaves? 3) Use the nail polish again to create several impressions from each leaf. Mounting one per leaf, select random fields of view and count the number of stomata within that field at 400X magnification. Record the data for both sun and shade leaves in the table below. You may want to increase your replicate number. Replicate 1 2 3 4 5 6 7 8 9 10 Total Average Sun Leaves Shade Leaves 4 Statistical Analysis: A Statistical Test for Comparing Two Sets of Values You have generated two sets of stomatal densities, one for shade leaves and one for sun leaves. Are they different? You could compare their means and standard deviations, using a ttest. (The standard deviation is a measure of variability around the mean.) If the standard deviations are relatively small compared to the distance between the means, you would be able to say that the values are different. An introduction to the t-test can be found on the biology web site, and calculations can be done either by hand or using any statistical software. The t-test is a type of "parametric" test. This means that the test itself is based upon certain assumptions about the data. In particular, the values are assumed to approximate a normal distribution, and the standard deviations of both sets of numbers are assumed to be equal. Good statisticians test these assumptions before they conduct a parametric test. For many types of data, nonparametric tests are also available. These do not rely upon the data conforming to any particular distribution; that is, they are "robust". Many of them are also simple to calculate by hand. The Mann-Whitney U-test is appropriate for comparing two sets of numbers to see whether or not they are different. H0: The stomatal density is the same for shade leaves and sun leaves of the oaks we sampled. Procedure: 1. List the observations in each column from the smallest to the largest so that the two samples may easily be compared. 2. For each observation in one sample, count the number of observations in the other sample that are smaller. Count 1/2 for each tie. 3. Sum the counts (=C). The Mann-Whitney Statistic U is either C or (n1n2- C), whichever is greater. (n1 and n2 represent sample sizes.) 4. Determine the significance level of your statistic. If you were doing this for publication, you would need to adjust your statistic for ties. We will hope for no ties. If the sample sizes are 20, you can look up significance levels in a special table for the U-test. (See below.) If the sample sizes are > 20, calculate: (U - (n1n2)/2) t = n1n2(n1 + n2 + 1) 12 You can look this up in a table that gives values for the t statistic, using infinity for degrees of freedom. It is good for you to look at whole tables, but for now we have selected just the relevant row: p value t statistic .10 1.645 .05 1.960 .01 2.576 5 .001 3.291 How do you interpret this thing? Look again at the formula you used to calculate t. U is in the numerator. If U is large, t will be large. Under what conditions will U be large? It will be large if the values C and n1n2 were not close to each other in value; in other words, if the two distributions were quite different. (If the two distributions are similar, C will be close to n1n2.) A large t or U means that the distributions are different. Large compared to what? Large compared to the values in the table. You will notice that as the t or U statistics increase, the p value decreases. The smaller the p value, the more sure you are that the differences you see are real biological differences and are not just due to chance. If you have a sample size of 10 replicates each, here are critical values for the Mann-Whitney Utest: p value U statistic .10 68 .05 73 .01 81 .001 90 If you have sample sizes between 10 and 20, feel free to look up a significance table for the Mann-Whitney U test on line. For next week: Write a short (1 page) mini-paper telling about the observational experiment you did this week. The paper should include: introduction, methods, results, and discussion. Each section will be very short! You can look at guidelines for writing a scientific paper on the biology web page to get an idea of appropriate format. You do not need to include raw data. Means, sample sizes, and some estimate of variability in the data are necessary. (Standard deviation is probably easiest -- you can get it out of Excel.) You can report your statistics in the results section like this: The hedgehogs that were given warm milk went to sleep faster than the hedgehogs that received cold milk (U = 87, n = 10, p < 0.01). References Lake, JA, Quick WP, Beerling DJ, Woodward FI. 2001. Plant development – signals from mature to new leaves. Nature. 411:154. 6